Содержание
- 3. Discovery and naming In the early 1800s, Wollaston was studying an ore of platinum. Although scientists
- 4. Physical properties Rhodium is a silver-white metal. It has a melting point of 1,966°C (3,571°F) and
- 5. Chemical properties Rhodium is a relatively inactive metal. It is not attacked by strong acids. When
- 6. Occurrence in nature Rhodium is one of the rarest elements on Earth. Its abundance is estimated
- 7. Isotopes Only one naturally occurring isotope of rhodium is known, rhodium-103. Rhodium also has a number
- 8. Extraction Rhodium is usually obtained as a by-product in the recovery of platinum from its ores.
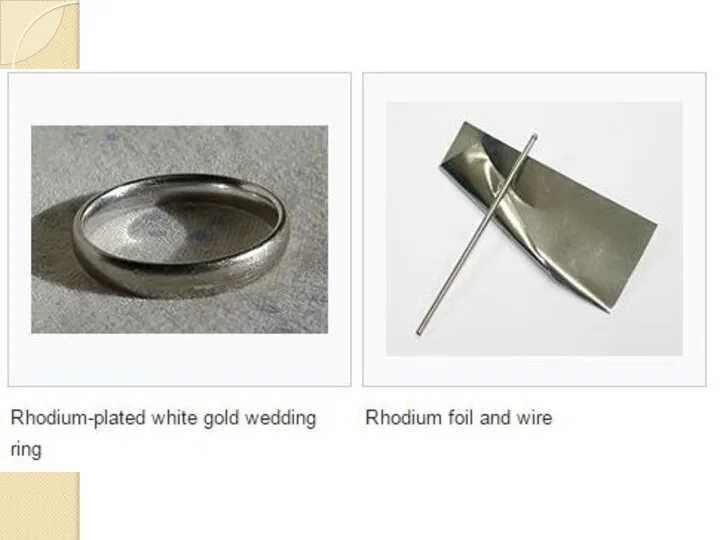
- 9. Uses Most of the rhodium metal sold in the United States is used to make alloys.
- 12. Compounds Compounds of rhodium are used as catalysts. A catalyst is a substance used to speed
- 13. Fluorides Rhodium trifluoride: RhF3 Rhodium hexafluoride: RhF6 Rhodium tetrafluoride: RhF4 Tetrarhodium eicosafluoride: [RhF5]4 Chlorides Rhodium trichloride:
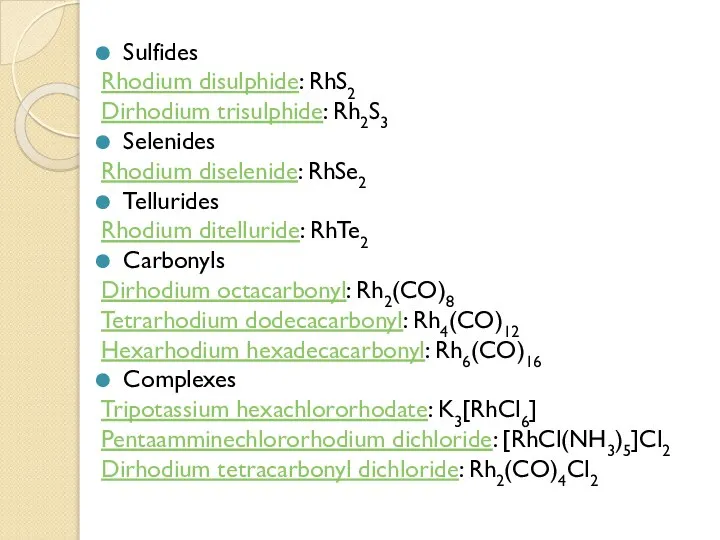
- 14. Sulfides Rhodium disulphide: RhS2 Dirhodium trisulphide: Rh2S3 Selenides Rhodium diselenide: RhSe2 Tellurides Rhodium ditelluride: RhTe2 Carbonyls
- 15. Health effects There are no studies of the health effects from rhodium or its common compounds.
- 17. Скачать презентацию












 Спортивная игра волейбол
Спортивная игра волейбол Теория врожденных знаний Выполнили : Елена Данильченко и Ольга Ярош 2 курс, лого-1
Теория врожденных знаний Выполнили : Елена Данильченко и Ольга Ярош 2 курс, лого-1  Конкурс силачей - презентация для начальной школы_
Конкурс силачей - презентация для начальной школы_ Народна медицина і лікувальна магія українців
Народна медицина і лікувальна магія українців Организация модульного обучения
Организация модульного обучения Презентация по МХК Художественная культура Среднего Востока
Презентация по МХК Художественная культура Среднего Востока  «Золотой век» русской культуры. Первая половина XIX века. Архитектура, скульптура, живопись
«Золотой век» русской культуры. Первая половина XIX века. Архитектура, скульптура, живопись ИНТЕРМОДАЛЬНЫЕ ПЕРЕВОЗКИ ГРУЗОВ Контейнерные перевозки грузов; Операторы смешанной перевозки грузов; Особенности взаимодей
ИНТЕРМОДАЛЬНЫЕ ПЕРЕВОЗКИ ГРУЗОВ Контейнерные перевозки грузов; Операторы смешанной перевозки грузов; Особенности взаимодей Апаратура обчислювальних засобів. Обчислювальні засоби КЗА 86Ж6. (Тема 7.1)
Апаратура обчислювальних засобів. Обчислювальні засоби КЗА 86Ж6. (Тема 7.1) ГангренаТкани
ГангренаТкани Объекты культурного наследия Европы
Объекты культурного наследия Европы Презентация Юридические лица,как субъекты гражданского права
Презентация Юридические лица,как субъекты гражданского права  Патология слизистой оболочки полости рта 2015
Патология слизистой оболочки полости рта 2015 Иридодиагностика-4
Иридодиагностика-4 Безопасное поведение детей за компьютером
Безопасное поведение детей за компьютером  Лекция по теме: «Металлы»
Лекция по теме: «Металлы» Древесина и изделия из нее
Древесина и изделия из нее  Стимулирование свободным временем
Стимулирование свободным временем Шалаш Ленина в разливе
Шалаш Ленина в разливе Обращение болельщиков сборной Беларуси
Обращение болельщиков сборной Беларуси Николай Николаевич Раевский (1771—1829)
Николай Николаевич Раевский (1771—1829) Смертная казнь как вид уголовного наказания
Смертная казнь как вид уголовного наказания Презентация "ЖИВОПИСЬ ГОЛЛАНДИИ" - скачать презентации по МХК
Презентация "ЖИВОПИСЬ ГОЛЛАНДИИ" - скачать презентации по МХК Медиатеория и медиаистория
Медиатеория и медиаистория Плитні матеріали
Плитні матеріали Иволгинский дацан
Иволгинский дацан День славянской письменности и культуры
День славянской письменности и культуры DIASOFT Российский разработчик программного обеспечения для автоматизации банков (АБС), инвестиционных и страховых компаний
DIASOFT Российский разработчик программного обеспечения для автоматизации банков (АБС), инвестиционных и страховых компаний