Содержание
- 2. HISTORY OF THE ATOM 460 BC Democritus develops the idea of atoms he pounded up materials
- 3. HISTORY OF THE ATOM 1808 John Dalton suggested that all matter was made up of tiny
- 4. HISTORY OF THE ATOM 1898 Joseph John Thompson found that atoms could sometimes eject a far
- 5. HISTORY OF THE ATOM Thompson develops the idea that an atom was made up of electrons
- 6. HISTORY OF THE ATOM 1910 Ernest Rutherford oversaw Geiger and Marsden carrying out his famous experiment.
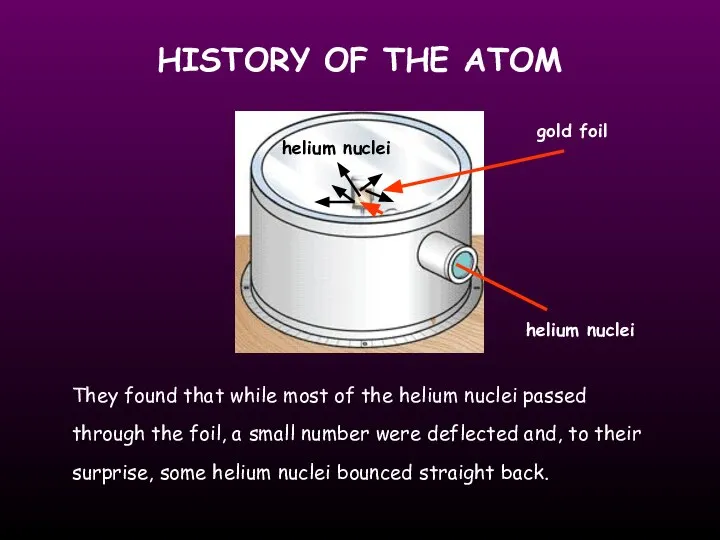
- 7. HISTORY OF THE ATOM gold foil helium nuclei They found that while most of the helium
- 8. HISTORY OF THE ATOM Rutherford’s new evidence allowed him to propose a more detailed model with
- 9. HISTORY OF THE ATOM 1913 Niels Bohr studied under Rutherford at the Victoria University in Manchester.
- 10. Bohr’s Atom electrons in orbits nucleus
- 11. HELIUM ATOM + N N + - - proton electron neutron Shell What do these particles
- 12. ATOMIC STRUCTURE Particle proton neutron electron Charge + ve charge -ve charge No charge 1 1
- 13. ATOMIC STRUCTURE the number of protons in an atom the number of protons and neutrons in
- 14. ATOMIC STRUCTURE Electrons are arranged in Energy Levels or Shells around the nucleus of an atom.
- 15. ATOMIC STRUCTURE There are two ways to represent the atomic structure of an element or compound;
- 16. ELECTRONIC CONFIGURATION With electronic configuration elements are represented numerically by the number of electrons in their
- 17. ELECTRONIC CONFIGURATION Write the electronic configuration for the following elements; Ca O Cl Si Na 20
- 18. DOT & CROSS DIAGRAMS With Dot & Cross diagrams elements and compounds are represented by Dots
- 19. DOT & CROSS DIAGRAMS Draw the Dot & Cross diagrams for the following elements; O Cl
- 20. SUMMARY The Atomic Number of an atom = number of protons in the nucleus. The Atomic
- 22. Скачать презентацию


















 Волоконно-оптичні лінії зв'язку. (Лекція 8)
Волоконно-оптичні лінії зв'язку. (Лекція 8) Упругие волны. (Тема 5)
Упругие волны. (Тема 5) Геометрия в физике
Геометрия в физике Урок физики 7 класс
Урок физики 7 класс Законы Ньютона
Законы Ньютона Презентация по физике Курс лекций по общей физике
Презентация по физике Курс лекций по общей физике Презентация на тему: Трансформатор
Презентация на тему: Трансформатор Механічні коливання та хвилі
Механічні коливання та хвилі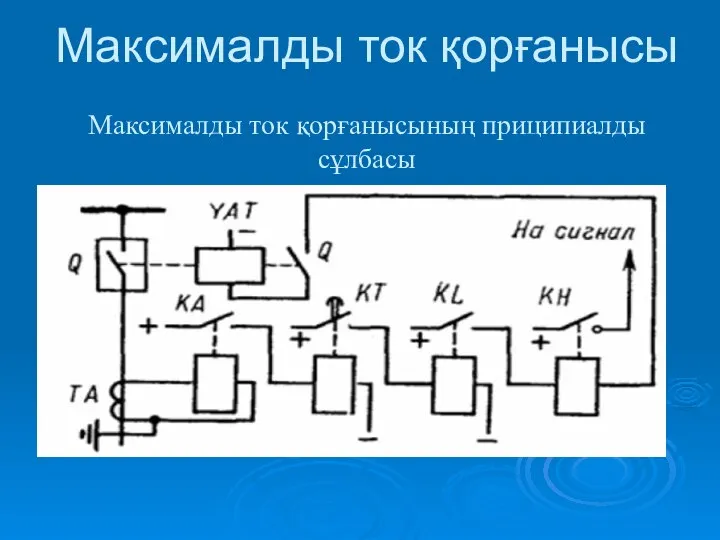 Максималды ток қорғанысы
Максималды ток қорғанысы Теория вероятностей. Треугольник Паскаля.
Теория вероятностей. Треугольник Паскаля.  Знаменитые лаборатории мира
Знаменитые лаборатории мира Моделерование микрополоскового фильтра
Моделерование микрополоскового фильтра Тестовые вопросы по физике 1-35
Тестовые вопросы по физике 1-35 Спонтанное и вынужденное излучение. Инверсия заселенности энергетических уровней. Принцип работы лазера
Спонтанное и вынужденное излучение. Инверсия заселенности энергетических уровней. Принцип работы лазера Автомобіль і люди
Автомобіль і люди Физика колебаний и волн. Спектральное разложение света. (Лекция 4)
Физика колебаний и волн. Спектральное разложение света. (Лекция 4) Спектроскопія ядерного магнітного резонансу
Спектроскопія ядерного магнітного резонансу Магнитное поле
Магнитное поле Zakon_Oma_dlya_uchastka_tsepi
Zakon_Oma_dlya_uchastka_tsepi Моделирование траектории самолёта
Моделирование траектории самолёта Электромагнитные волны Выполнила Жаркова С.В.
Электромагнитные волны Выполнила Жаркова С.В. Энергетическая характеристика электростатического поля Работа электрического поля по перемещению электрического заряда. Поте
Энергетическая характеристика электростатического поля Работа электрического поля по перемещению электрического заряда. Поте Специальная теория относительности. Постулаты теории относительности
Специальная теория относительности. Постулаты теории относительности Электромагниттік өріс көздерін санитарлық қадағалаудың әдістемелері
Электромагниттік өріс көздерін санитарлық қадағалаудың әдістемелері Ультрадисперсные (наноструктурные и ультрамелкозернистые) материалы
Ультрадисперсные (наноструктурные и ультрамелкозернистые) материалы Звук как физическое явление
Звук как физическое явление Основные положения термодинамики и теплотехники. Энтропия
Основные положения термодинамики и теплотехники. Энтропия Электроемкость. Конденсаторы. Энергия заряженного конденсатора
Электроемкость. Конденсаторы. Энергия заряженного конденсатора