Содержание
- 2. Irreversibility of processes There exist many processes that are irreversible: the net transfer of energy by
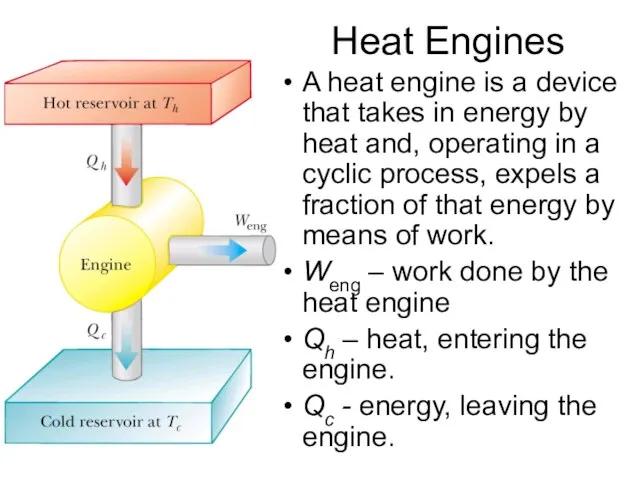
- 3. Heat Engines A heat engine is a device that takes in energy by heat and, operating
- 4. Thermal Efficiency of a Heat Engine
- 5. Heat Pumps or Refrigerators In a heat engine a fraction of heat from the hot reservoir
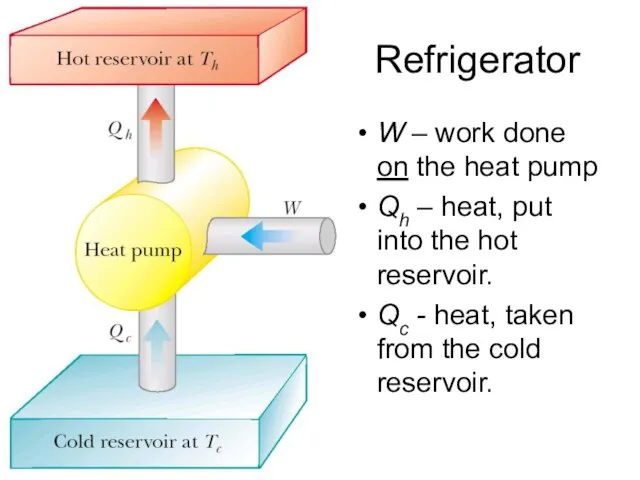
- 6. Refrigerator W – work done on the heat pump Qh – heat, put into the hot
- 7. Coefficient of performance of a refrigerator The effectiveness of a refrigerator is described in terms of
- 8. The Second Law of Thermodynamics The Kelvin form: It is impossible to construct a cyclic engine
- 9. The Second Law of Thermodynamics The Clausius form: It is impossible to construct a cyclic engine
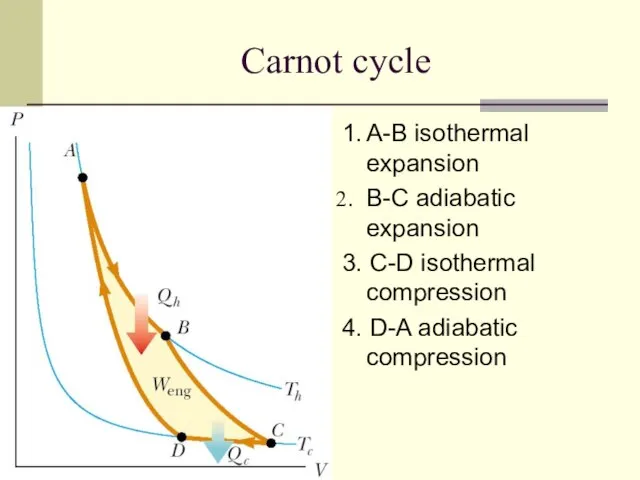
- 10. Carnot cycle 1. A-B isothermal expansion B-C adiabatic expansion 3. C-D isothermal compression 4. D-A adiabatic
- 11. Carnot Efficiency Using the equation of state and the first law of thermodynamics we can easily
- 12. So, the work done on a gas during an isothermal process A → B is: (1)
- 13. For adiabatic processes: So, statement (3) gives us:
- 14. So, using the last expression and the expression for efficiency: Thus we have proved that the
- 15. Carnot theorem The Carnot engine is the most efficient engine possible that operates between any two
- 16. Carnot Theorem Proof Let’s prove it from the contrary: let’s have Carnot engine A to be
- 17. Entropy Measures the amount of disorder in thermal system. It is a function of state, and
- 18. Entropy change calculations Entropy is a state variable, the change in entropy during a process depends
- 19. So for infinitesimal changes: The subscript r on the quantity dQr means that the transferred energy
- 20. Change of Entropy in a Carnot Cycle Carnot engine operates between the temperatures Tc and Th.
- 21. Reversibility of Carno Cycle Using equality, proved for the Carnot Cycle (slide N13): We eventually find
- 22. Reversible Cycle Now consider a system taken through an arbitrary (non-Carnot) reversible cycle. Because entropy is
- 23. Ideal Gas Reversible Process Suppose that an ideal gas undergoes a quasi-static, reversible process from an
- 24. - This expression demonstrates that ΔS depends only on the initial and final states and is
- 25. The Second Law of Thermodynamics The total entropy of an isolated system that undergoes a change
- 26. Microscopic States Every macrostate can be realized by a number of microstates. Each molecule occupies some
- 27. Entropy on a Microscopic Scale Let’s have an ideal gas expanding from Vi to Vf. Then
- 28. After further transformations: n – number of moles, R=kbNa. Then we use the equation for isothermal
- 29. Entropy is a measure of Disorder The more microstates there are that correspond to a given
- 31. Скачать презентацию

























 Поняття про полімери. Будова та їх властивості
Поняття про полімери. Будова та їх властивості  Червячная зубчатая передача Кулачковый механизм (кулачок)
Червячная зубчатая передача Кулачковый механизм (кулачок) Примеры решения задач по теме Движение с постоянным ускорением свободного падения
Примеры решения задач по теме Движение с постоянным ускорением свободного падения Исследование личности Ильи Давидовича Усыскина
Исследование личности Ильи Давидовича Усыскина Многократное рассеяние волн
Многократное рассеяние волн Законы Ньютона
Законы Ньютона СУЩЕСТВОВАНИЕ НА ГЛУБИНЕ ПОДГОТОВИЛА: СМАГЛЮК ПОЛИНА
СУЩЕСТВОВАНИЕ НА ГЛУБИНЕ ПОДГОТОВИЛА: СМАГЛЮК ПОЛИНА Техническое обслуживание и ремонт кузовов и кабин автомобиля
Техническое обслуживание и ремонт кузовов и кабин автомобиля Действия электрического тока
Действия электрического тока Презентация по физике "Реактивное движение" - скачать бесплатно_
Презентация по физике "Реактивное движение" - скачать бесплатно_ МАРИЯ СКЛОДОВСКАЯ- КЮРИ
МАРИЯ СКЛОДОВСКАЯ- КЮРИ Презентация по физике "Физика и космическая еда" - скачать
Презентация по физике "Физика и космическая еда" - скачать  Световые явления в живой и неживой природе
Световые явления в живой и неживой природе Измерение объема
Измерение объема Электротехника и электроника. Однофазные электрические цепи синусоидального тока. (Лекция 2)
Электротехника и электроника. Однофазные электрические цепи синусоидального тока. (Лекция 2) Самоиндукция Презентация по физике
Самоиндукция Презентация по физике Частотный метод синтеза корректирующего звена по ЛАЧХ разомкнутой системы
Частотный метод синтеза корректирующего звена по ЛАЧХ разомкнутой системы Техническая термодинамика. Термодинамичекие потенциалы. Эффект Джоуля-Томсона. (Лекция 5)
Техническая термодинамика. Термодинамичекие потенциалы. Эффект Джоуля-Томсона. (Лекция 5) Теорія псевдопотенціалів
Теорія псевдопотенціалів История развития электрического освещения
История развития электрического освещения Мастер-класс: Поиск новых решений с помощью морфологической матрицы
Мастер-класс: Поиск новых решений с помощью морфологической матрицы Технологические процессы и операции монтажа
Технологические процессы и операции монтажа Презентация Ядерная энергетика
Презентация Ядерная энергетика Конструирование подшипниковых узлов, механизмов подач и суппортов
Конструирование подшипниковых узлов, механизмов подач и суппортов Статика и плотность сжимаемой среды
Статика и плотность сжимаемой среды Презентация по физике "Оптика 11 класс" - скачать
Презентация по физике "Оптика 11 класс" - скачать  Электромагнитное поле. Электромагнитные волны
Электромагнитное поле. Электромагнитные волны Мао Цзедун
Мао Цзедун