Содержание
- 2. PLAN Classification of carbohydrates. Nomenclature. Structural representations be Fisher and Haworth. Chirality. Optical isomers. Tautomerism. Mutarotation.
- 3. Carbohydrates The term "carbohydrate" was proposed by K.G. Shmidt in 1844. Cn(H2O)m (n=3-9) A carbohydrate is
- 4. Carbohydrates. Classification. There are two classes of carbohydrates: simple carbohydrates and complex carbohydrates. Simple carbohydrates are
- 5. Monosaccharide‘s classification. type of the carbonyl group Carbon chain length 3 Carbon - Trioses are not
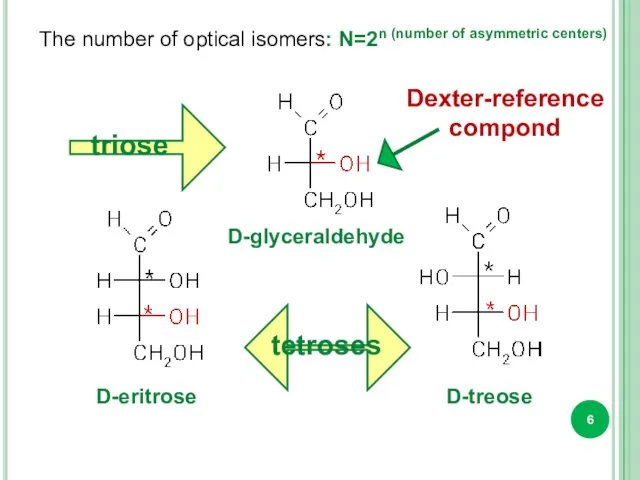
- 6. The number of optical isomers: N=2n (number of asymmetric centers) triose tetroses D-glyceraldehyde D-eritrose D-treose Dexter-reference
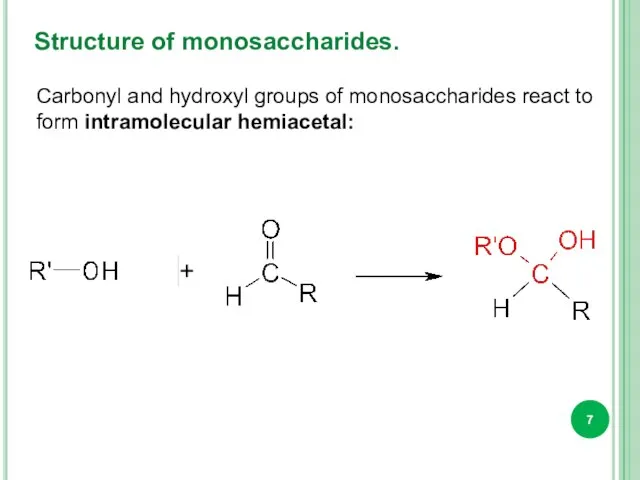
- 7. Structure of monosaccharides. Carbonyl and hydroxyl groups of monosaccharides react to form intramolecular hemiacetal:
- 8. The structure of monosaccharides is presented in three forms: Fisher projection: straight chain representation. 2. Haworth
- 9. Structure of monosaccharides. β-Hemiacetal hydroxyl α-Hemiacetal hydroxyl Anomeric center Anomeric center D-Glucose β,D-glucopyranose α,D-glucopyranose Dexter OH
- 10. Hydroxyl group at the anomeric center is called a hemiacetal or glycoside. Diastereomers - stereoisomers that
- 11. α-Hemiacetal hydroxyl Prospective Haworth formula (1929)
- 12. Prospective Haworth formula One can remember that the β anomer is cis by the mnemonic, "It's
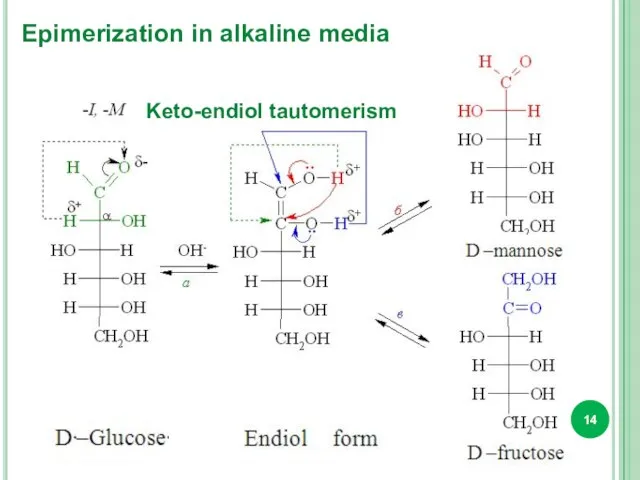
- 13. Isomeric transformation of monosaccharides by the action of alkalis is called epimerization. Epimers are called diastereoisomers,
- 14. Keto-endiol tautomerism Epimerization in alkaline media
- 15. 1. Intermolecular dehydration Chemical properties Reaction to distinguish pentoses from hexoses.
- 16. 1. Intermolecular dehydration Chemical properties Reaction to distinguish pentoses from hexoses.
- 17. Molish test α-naphtol
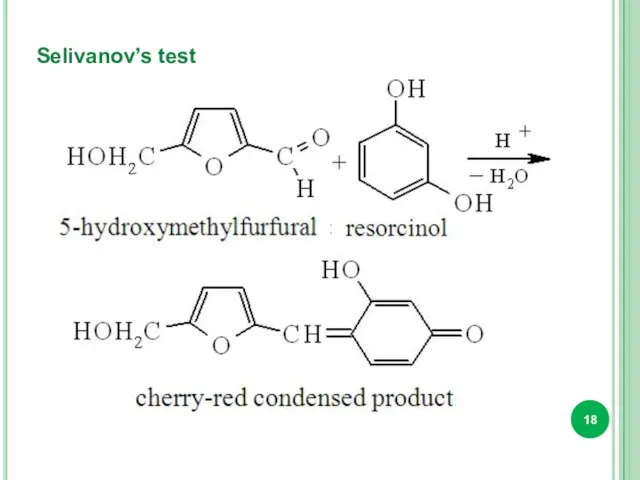
- 18. Selivanov’s test
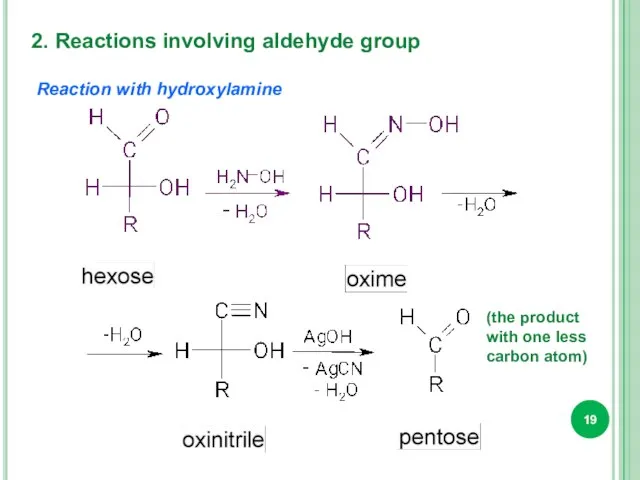
- 19. 2. Reactions involving aldehyde group Reaction with hydroxylamine (the product with one less carbon atom)
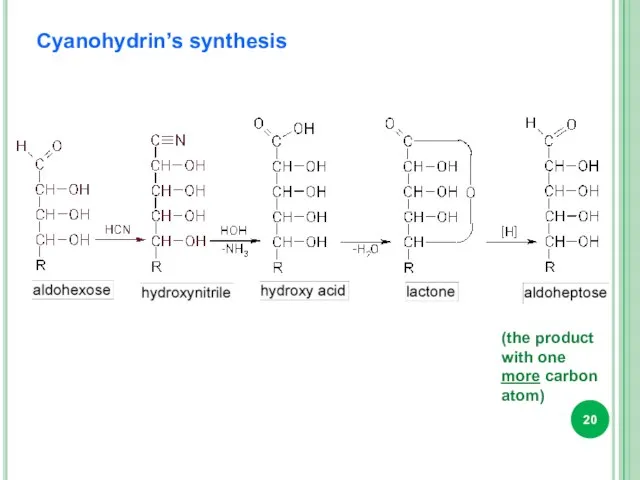
- 20. Cyanohydrin’s synthesis (the product with one more carbon atom)
- 21. 3. Reduction diabetic sweeteners
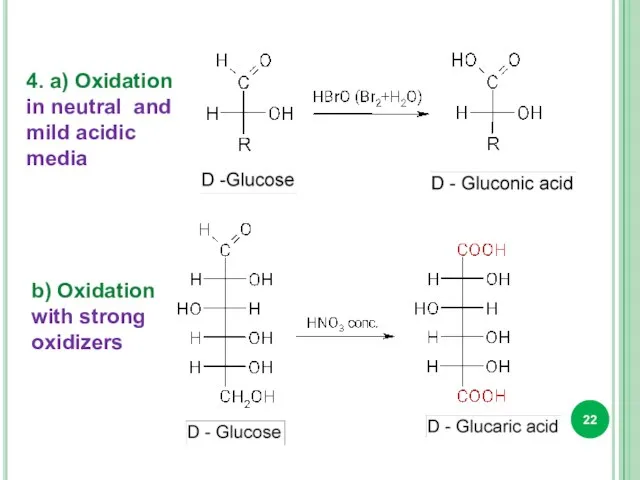
- 22. 4. a) Oxidation in neutral and mild acidic media b) Oxidation with strong oxidizers
- 23. c) Oxidation by enzymes
- 24. Identification of aldehyde group with:
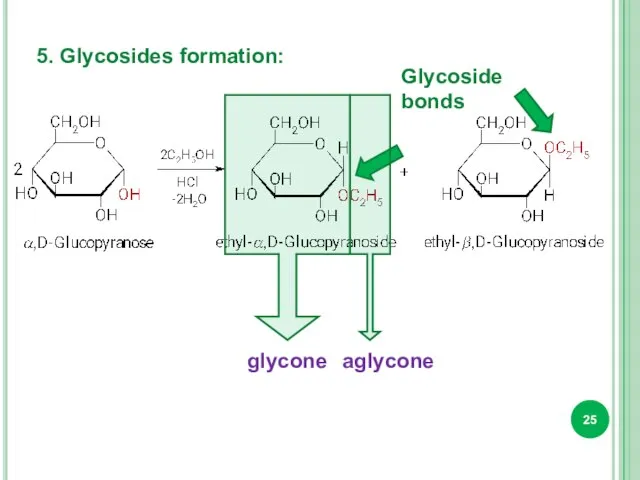
- 25. 5. Glycosides formation: Glycoside bonds glycone aglycone
- 26. 6. Acylation: 7. Alkylation:
- 27. Disaccharides (bioses) Depending on the method of the glycosidic bond formation reducing nonreducing In reducing disaccharide
- 28. Maltose
- 29. Cellobiose
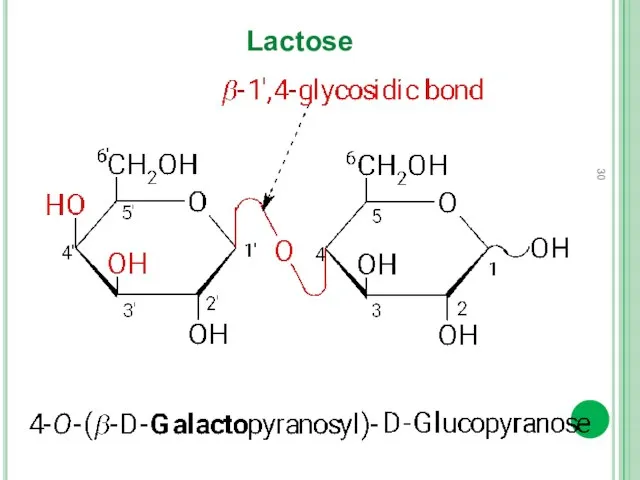
- 30. Lactose
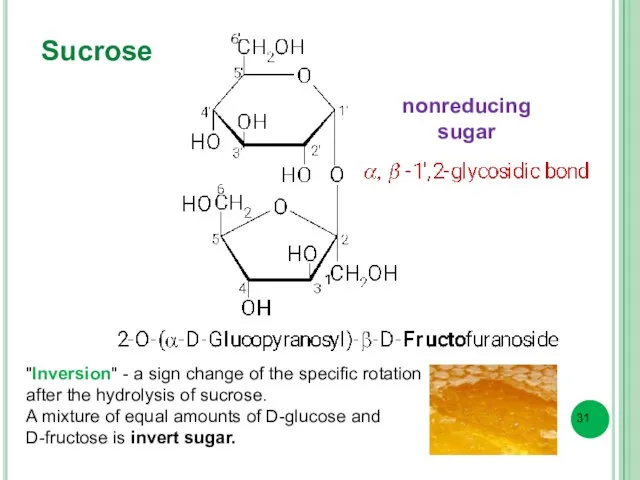
- 31. Sucrose "Inversion" - a sign change of the specific rotation after the hydrolysis of sucrose. A
- 32. Sucrose. chemical properties. Doesn’t mutorotate No silver mirror reaction No reactions by aldehyde group Hydrolysing to
- 33. Amylose 20% Soluble in Н2О Amylopectine 80% Insoluble Н2О Starch hydrolysis Starch. Structure
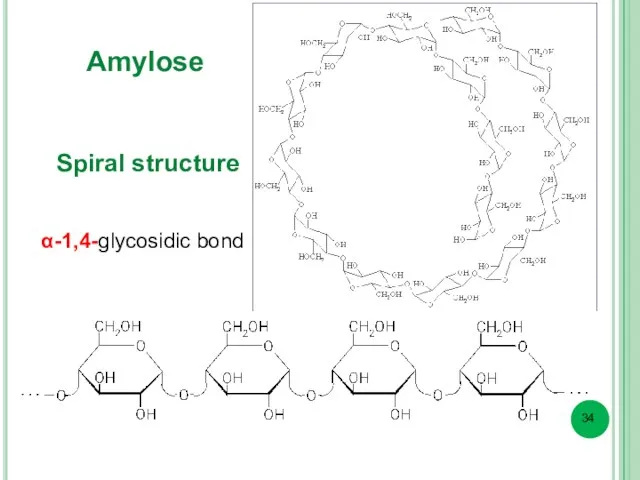
- 34. Amylose α-1,4-glycosidic bond Spiral structure
- 35. Amylose with iodine forms clastrates of dark blue color.
- 36. Amylopectin α-1,6’-glycosidic bond α-1,4’-glycosidic bond branched structure
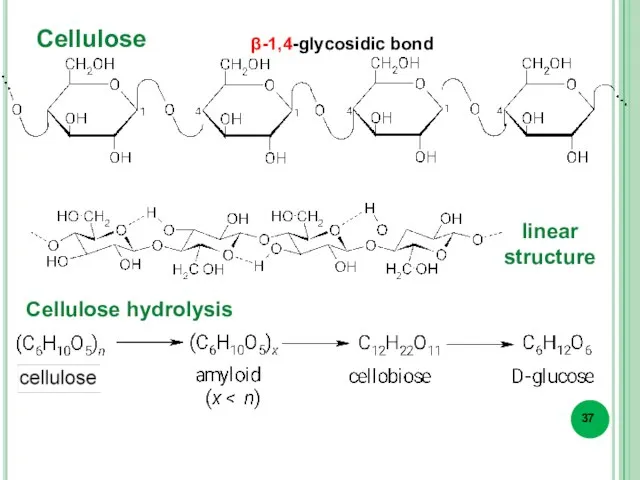
- 37. Cellulose Cellulose hydrolysis β-1,4-glycosidic bond linear structure
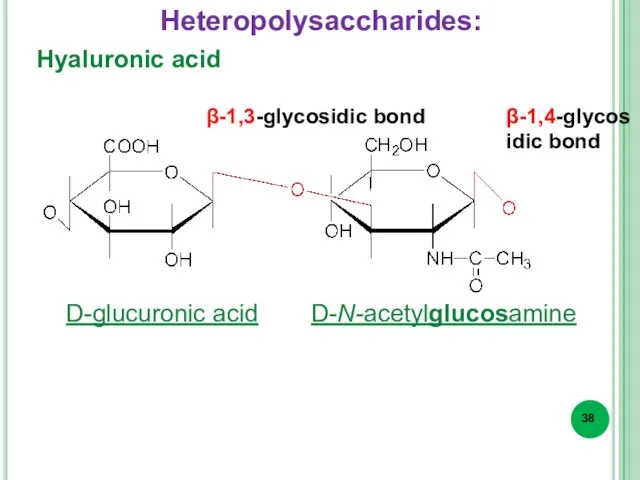
- 38. Hyaluronic acid β-1,3-glycosidic bond β-1,4-glycosidic bond D-glucuronic acid D-N-acetylglucosamine Heteropolysaccharides:
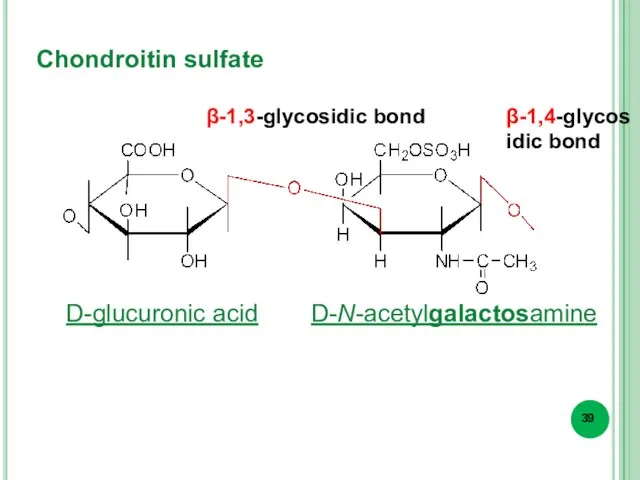
- 39. Chondroitin sulfate β-1,3-glycosidic bond β-1,4-glycosidic bond D-glucuronic acid D-N-acetylgalactosamine
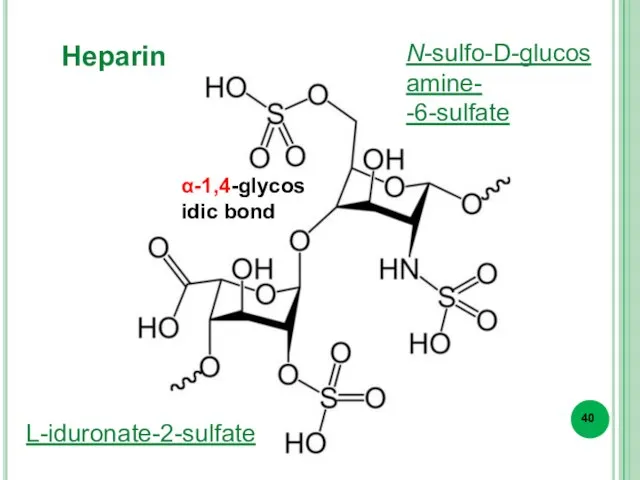
- 40. Heparin L-iduronate-2-sulfate N-sulfo-D-glucosamine- -6-sulfate α-1,4-glycosidic bond
- 42. Скачать презентацию
























 Введение в титриметрический анализ. Кислотно-основное титрование
Введение в титриметрический анализ. Кислотно-основное титрование Соединения алюминия
Соединения алюминия Свойства океанической воды
Свойства океанической воды Классификация элементов. III тип. Концентрационные элементы
Классификация элементов. III тип. Концентрационные элементы Основные понятия и законы химии
Основные понятия и законы химии VI группа периодической системы. VIA-группа
VI группа периодической системы. VIA-группа Правила безопасности в кабинете химии. Приёмы обращения с лабораторным оборудованием
Правила безопасности в кабинете химии. Приёмы обращения с лабораторным оборудованием
 Лабораторные, микробиологические и иммунологические исследования. Окраска препаратов по методу Грама
Лабораторные, микробиологические и иммунологические исследования. Окраска препаратов по методу Грама Неорганические соединения. Вода. Роль воды в клетке
Неорганические соединения. Вода. Роль воды в клетке Адсорбционные взаимодействия
Адсорбционные взаимодействия Бинарный интегрированный урок (география + химия) . 8 класс
Бинарный интегрированный урок (география + химия) . 8 класс Жесткость воды и способы ее устранения
Жесткость воды и способы ее устранения Аттестационная работа. Исследование влияния питательных веществ на качество выгонки древесно-кустарниковых растений
Аттестационная работа. Исследование влияния питательных веществ на качество выгонки древесно-кустарниковых растений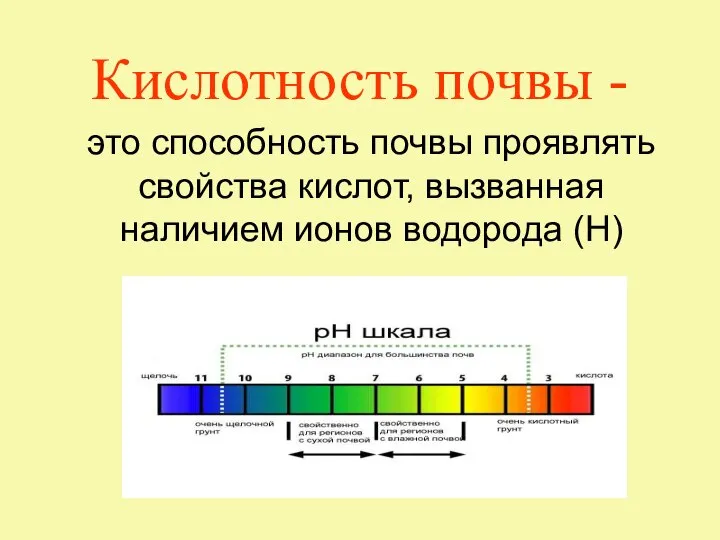 Кислотность почвы
Кислотность почвы Валентность. Порядок действий при составлении химической формулы
Валентность. Порядок действий при составлении химической формулы Производство азотной кислоты
Производство азотной кислоты Химия в сельском хозяйстве
Химия в сельском хозяйстве Крахма́л (C6H10O5)n
Крахма́л (C6H10O5)n Электрохимические цепи
Электрохимические цепи Кислоты и соли
Кислоты и соли Окислительно-восстановительные реакции
Окислительно-восстановительные реакции Металлы. Атомы металлов
Металлы. Атомы металлов History of radioactivity
History of radioactivity Гипер-гипофосфатемия
Гипер-гипофосфатемия Аттестационная работа. Рабочая программа по курсу внеурочной деятельности для обучающихся 7 класса «Химия в профессиях»
Аттестационная работа. Рабочая программа по курсу внеурочной деятельности для обучающихся 7 класса «Химия в профессиях» Презентация по Химии "Кристаллы вокруг нас" - скачать смотреть
Презентация по Химии "Кристаллы вокруг нас" - скачать смотреть  Вода. Химические и физические свойства воды
Вода. Химические и физические свойства воды