Содержание
- 3. Refinery gas C1-C4 Light gasoline (bp: 25-95 °C) C5-C12 Naphtha (bp 95-150 °C) Kerosene (bp: 150-230
- 4. Petroleum refining Cracking converts high molecular weight hydrocarbons to more useful, low molecular weight ones Reforming
- 5. 2.14 Physical Properties of Alkanes and Cycloalkanes
- 6. Boiling Points of Alkanes governed by strength of intermolecular attractive forces alkanes are nonpolar, so dipole-dipole
- 7. Induced dipole-Induced dipole attractive forces + – + – two nonpolar molecules center of positive charge
- 8. Induced dipole-Induced dipole attractive forces + – + – movement of electrons creates an instantaneous dipole
- 9. Induced dipole-Induced dipole attractive forces + – + – temporary dipole in one molecule (left) induces
- 10. Induced dipole-Induced dipole attractive forces + – + – temporary dipole in one molecule (left) induces
- 11. Induced dipole-Induced dipole attractive forces + – + – the result is a small attractive force
- 12. Induced dipole-Induced dipole attractive forces + – + – the result is a small attractive force
- 13. increase with increasing number of carbons more atoms, more electrons, more opportunities for induced dipole-induced dipole
- 14. increase with increasing number of carbons more atoms, more electrons, more opportunities for induced dipole-induced dipole
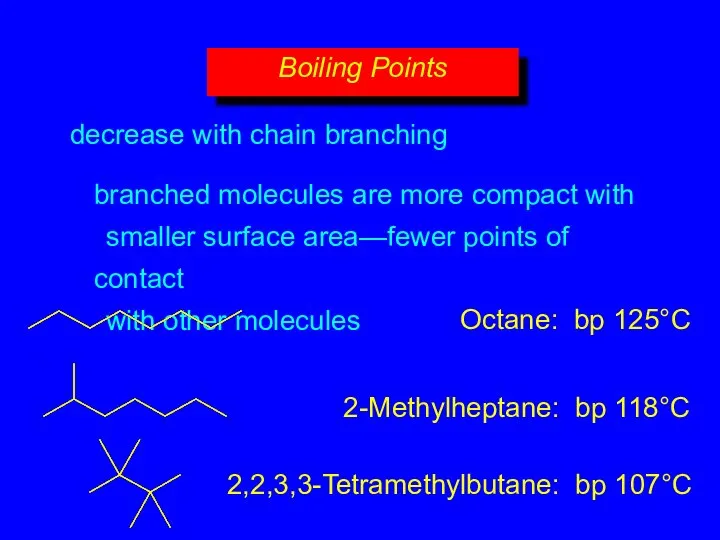
- 15. decrease with chain branching branched molecules are more compact with smaller surface area—fewer points of contact
- 16. 2.15 Chemical Properties. Combustion of Alkanes All alkanes burn in air to give carbon dioxide and
- 17. increase with increasing number of carbons more moles of O2 consumed, more moles of CO2 and
- 18. Heats of Combustion 4817 kJ/mol 5471 kJ/mol 6125 kJ/mol 654 kJ/mol 654 kJ/mol Heptane Octane Nonane
- 19. increase with increasing number of carbons more moles of O2 consumed, more moles of CO2 and
- 20. Heats of Combustion 5471 kJ/mol 5466 kJ/mol 5458 kJ/mol 5452 kJ/mol
- 21. Isomers can differ in respect to their stability. Equivalent statement: Isomers differ in respect to their
- 22. 8CO2 + 9H2O 5452 kJ/mol 5458 kJ/mol 5471 kJ/mol 5466 kJ/mol Figure 2.5
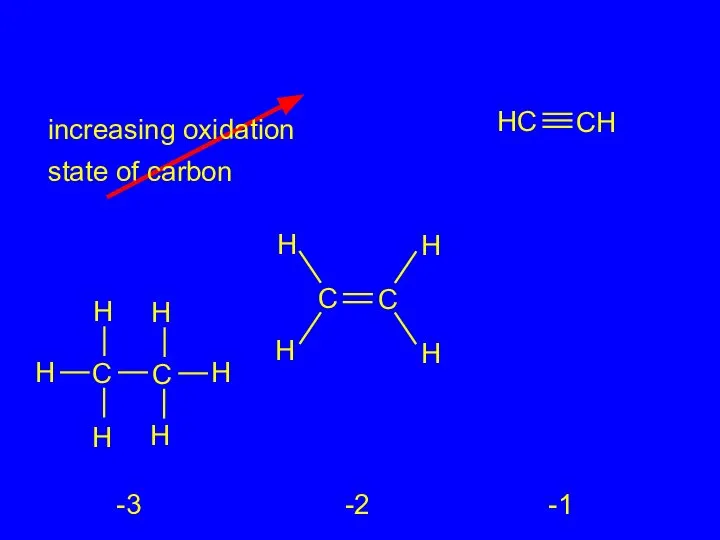
- 23. 2.16 Oxidation-Reduction in Organic Chemistry Oxidation of carbon corresponds to an increase in the number of
- 24. increasing oxidation state of carbon -4 -2 0 +2 +4
- 25. increasing oxidation state of carbon -3 -2 -1
- 26. But most compounds contain several (or many) carbons, and these can be in different oxidation states.
- 27. Fortunately, we rarely need to calculate the oxidation state of individual carbons in a molecule .
- 28. Generalization Oxidation of carbon occurs when a bond between carbon and an atom which is less
- 30. Скачать презентацию

























 Биологическое окисление
Биологическое окисление Симметрия кристаллических решеток
Симметрия кристаллических решеток Нітрати в продуктах харчування
Нітрати в продуктах харчування  Аллотропные модификации
Аллотропные модификации Проектная деятельность школьников в процессе обучения химии
Проектная деятельность школьников в процессе обучения химии Мінеральні добрива Хімія, 10 клас Розробила вчитель сзш №213 Деснянського р-ну м. Києва Мартиненко О.А.
Мінеральні добрива Хімія, 10 клас Розробила вчитель сзш №213 Деснянського р-ну м. Києва Мартиненко О.А.  Уран. Применение. Ядерное топливо
Уран. Применение. Ядерное топливо Презентация к уроку химии по теме «Спирты» 10 класс. УМК Габриеляна О.С. Базовый уровень
Презентация к уроку химии по теме «Спирты» 10 класс. УМК Габриеляна О.С. Базовый уровень Соли. Классификация сложных неорганических веществ
Соли. Классификация сложных неорганических веществ Epoxy-Based Fibre Reinforced Nanocomposites
Epoxy-Based Fibre Reinforced Nanocomposites Сера, сероводород, сульфиды
Сера, сероводород, сульфиды Химия элементов. Лекция 6 Комплексные соединения: типы и классификация. Методы получения и разрушения. Решение задач.
Химия элементов. Лекция 6 Комплексные соединения: типы и классификация. Методы получения и разрушения. Решение задач.  Радиофармацевтическая химия
Радиофармацевтическая химия Век медный, бронзовый, железный. Химические элементы металлы
Век медный, бронзовый, железный. Химические элементы металлы Характеристика химического элемента
Характеристика химического элемента Мыло. Мылящие вещества в природе
Мыло. Мылящие вещества в природе Ферменты. Размеры ферментов и их строение
Ферменты. Размеры ферментов и их строение Vodík
Vodík Скло, його різновиди та застосування Робота Учениці 10-А класу Баранюк Надії
Скло, його різновиди та застосування Робота Учениці 10-А класу Баранюк Надії  Проектная работа. Косметика своими руками
Проектная работа. Косметика своими руками Серная кислота и её свойства
Серная кислота и её свойства Презентация по Химии "Полімери" - скачать смотреть бесплатно
Презентация по Химии "Полімери" - скачать смотреть бесплатно Хімічні зв’язки Алмакаева 8-Б
Хімічні зв’язки Алмакаева 8-Б  Золото и серебро
Золото и серебро Химические волокна. Полиэфирные волокна. Лавсан
Химические волокна. Полиэфирные волокна. Лавсан Химические методы получения наночастиц
Химические методы получения наночастиц Выполнил: Лизунов Иван, 9Т
Выполнил: Лизунов Иван, 9Т Окислительно – восстановительные реакции. Раева Анна Семеновна Учитель химии МОБУ СОШ № 24 имени С. И. Климакова, г. Якутск
Окислительно – восстановительные реакции. Раева Анна Семеновна Учитель химии МОБУ СОШ № 24 имени С. И. Климакова, г. Якутск