Содержание
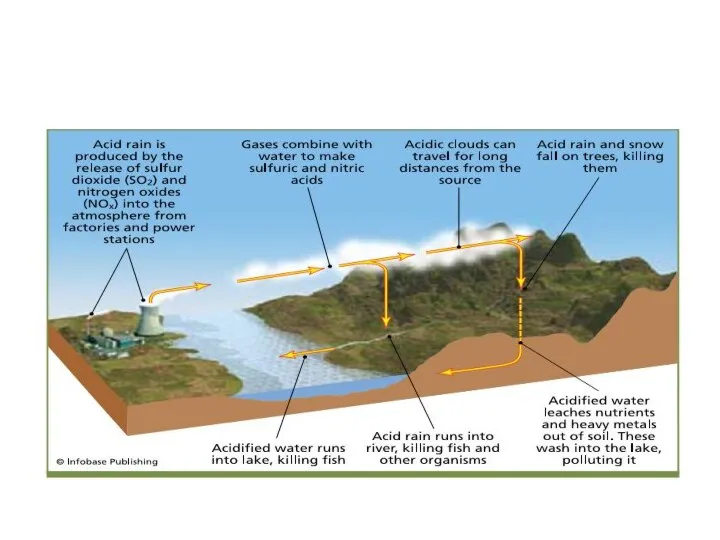
- 2. General Question about Acidification Аcid deposition occurs when sulfuric acid, nitric acid, or hydrochloric acid, emitted
- 3. General Question about Acidification Acid deposition is caused by the emission or atmospheric formation of gas-
- 4. History of the problem Historically, coal was the first and largest source of anthropogenically produced atmospheric
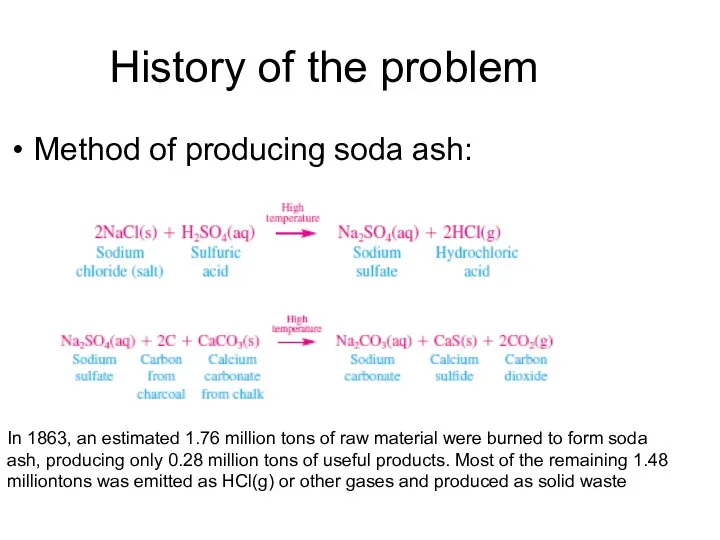
- 5. History of the problem Method of producing soda ash: In 1863, an estimated 1.76 million tons
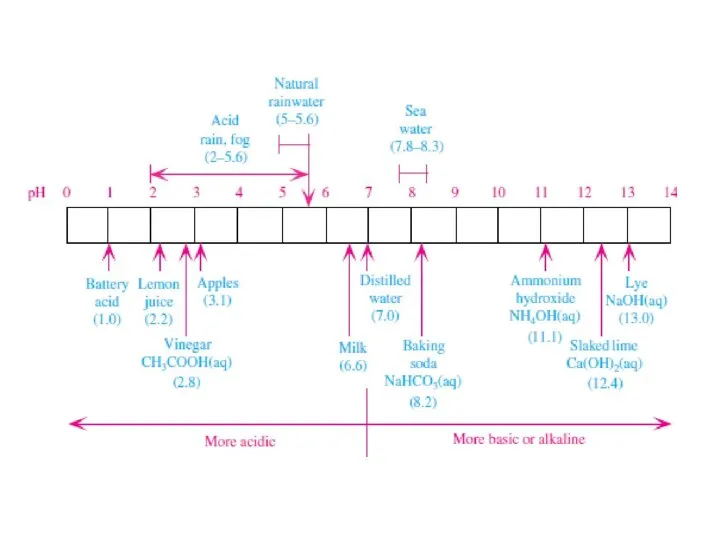
- 6. рН pH was defined as pH = - log10[H+], where [H+] is the molarity (moles per
- 8. Carbonic Acid Water can be acidified in one of several ways. When gas-phase carbon dioxide dissolves
- 9. A fraction of CO2(g) always dissolves in rainwater. Thus, rainwater, even in the cleanest environment on
- 10. Sulfuric Acid When gas-phase sulfuric acid condenses onto rain drops, the resulting aqueous-phase sulfuric acid [H2SO4(aq)],
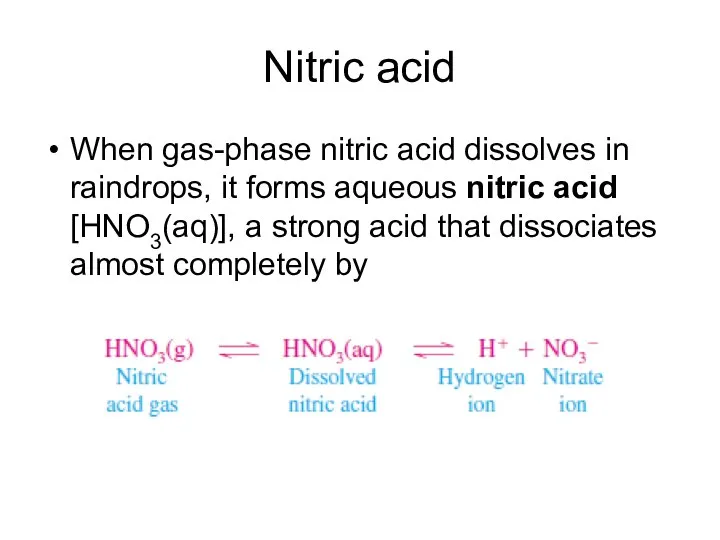
- 11. Nitric acid When gas-phase nitric acid dissolves in raindrops, it forms aqueous nitric acid [HNO3(aq)], a
- 12. Hydrochloric Acid When gas-phase hydrochloric acid dissolves in raindrops, it forms aqueous hydrochloric acid [HCl(aq)], a
- 13. Sources of Acids Some of the enhanced acidity of rainwater from sulfuric acid, nitric acid, and
- 14. Sources of Acids Acid deposition occurs when anthropogenically produced acids are deposited to the ground, plants,
- 15. Sulfuric Acid Deposition The most abundant acid in the air is usually sulfuric acid [H2SO4(aq)], whose
- 16. Power plants usually emit SO2(g) from high stacks so that the pollutant is not easily downwashed
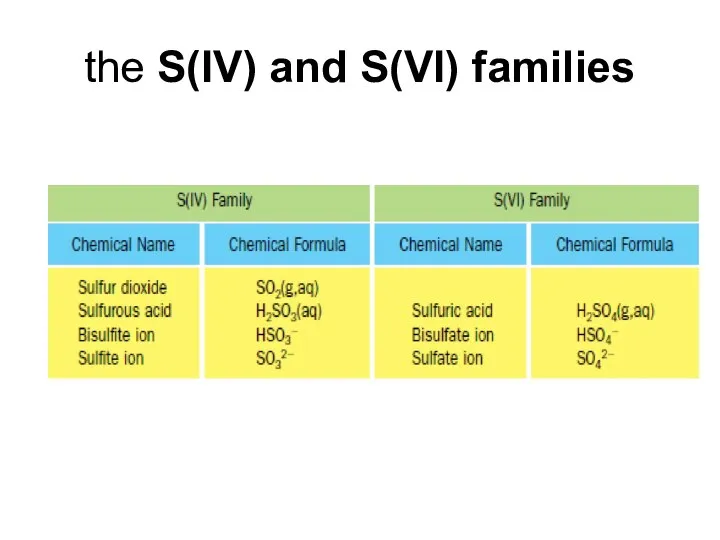
- 17. the S(IV) and S(VI) families
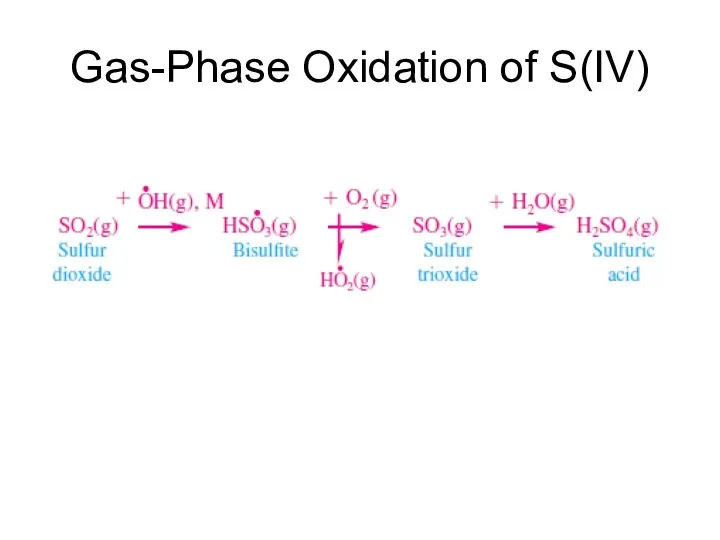
- 18. Gas-Phase Oxidation of S(IV)
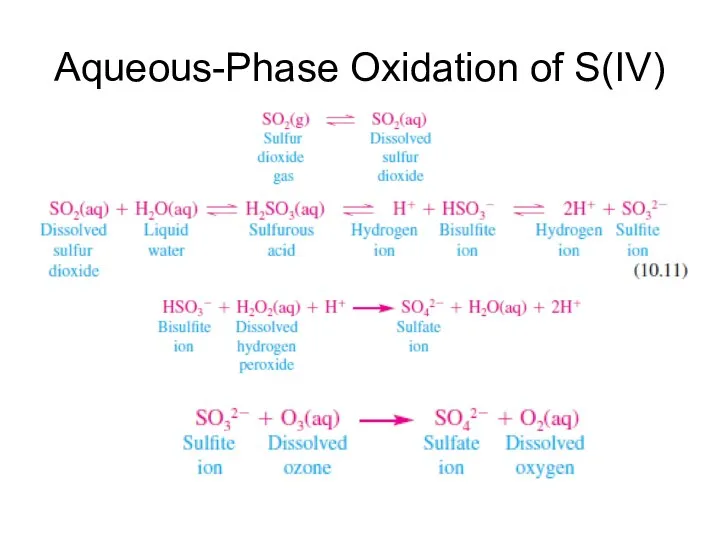
- 19. Aqueous-Phase Oxidation of S(IV)
- 20. Nitric acid deposition
- 21. Effect of acid deposition. London-type smog were recorded in London in the IX and XX centuries.
- 22. Effect of acid deposition. Effects on Lakes and Streams Effects on Biomass a) Acidified forest, Oberwiesenthal,
- 23. Effect of acid deposition Effects on Buildings and Sculptures Sandstone figure over the portal of a
- 24. Acidification in the Arctic
- 25. Sources Industrial areas farther south contribute to Arctic air pollution Most sulfur in Arctic air comes
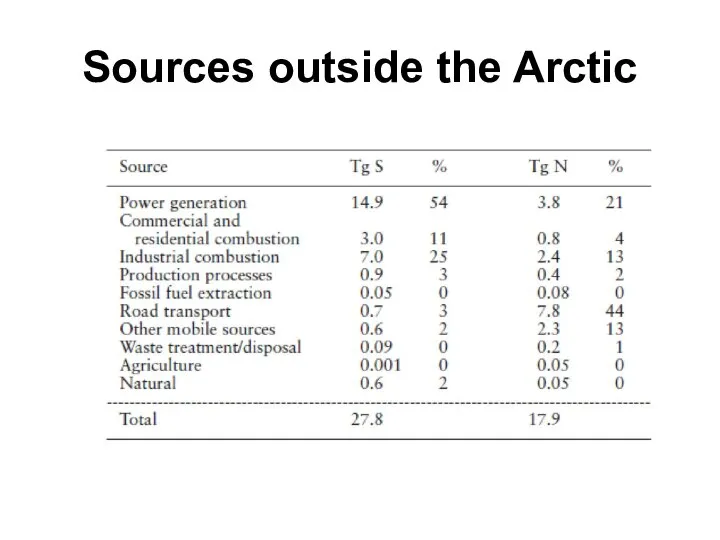
- 26. Sources outside the Arctic
- 27. Natural sources The algae in ocean surface waters are a source of sulfur to the atmosphere
- 28. Ammonia (NH3) is also involved in acidification processes; it is a neutralizing compound in the atmosphere,
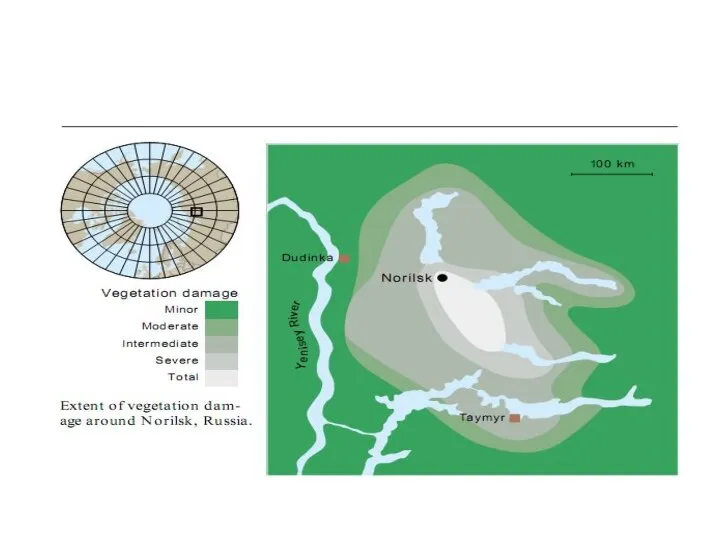
- 29. Sources within the Arctic Metal smelters have the largest emissions within the Arctic Production of copper,
- 30. Sources within the Arctic Exploitation and usage of fossil fuels Within the Arctic, there is coal
- 31. Sources within the Arctic Shipping and fishing activities are also sources of air pollutants in the
- 32. Natural emissions There are areas of volcanic activity in the North Atlantic and Bering Sea regions,
- 33. Natural emissions In winter, anthropogenic sources account for almost all of the sulfur in the Arctic
- 34. Local energy production is a small source Emissions from energy production in the Arctic are generally
- 35. Nitrogen emissions are less important Burning of fossils fuels also creates nitrogen oxides. In more densely
- 36. Atmospheric processes The fate of sulfur and nitrogen emissions depends on what happens in the atmosphere.
- 37. Sulfur dioxide turns into haze and acid precipitation Fossil fuels with high sulfur content produce sulfur
- 38. The atmospheric chemistry of the sulfur cycle The atmospheric chemistry of the sulfur cycle is dominated
- 39. Nitrogen chemistry Nitric oxide (NO) and nitrogen dioxide (N02) are the two most important nitrogen oxide
- 40. the Zeldovich mechanism. The first reaction above has a relatively high activation energy, due to the
- 42. Effect in the Arctic Loss of soil fertility contributes to tree death Most Arctic mineral soils
- 45. Скачать презентацию



![рН pH was defined as pH = - log10[H+], where [H+]](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/643134/slide-5.jpg)





























 Водные ресурсы Земли
Водные ресурсы Земли Экологическое исследование почвы на территории Белоярского района - копия
Экологическое исследование почвы на территории Белоярского района - копия Су – тіршілік көзі
Су – тіршілік көзі Природа Заполярья – наша забота
Природа Заполярья – наша забота Экологические проблемы Краснодарского края
Экологические проблемы Краснодарского края Автомобиль и экология
Автомобиль и экология Окружающая среда и здоровье человека
Окружающая среда и здоровье человека Бытовые отходы человечества
Бытовые отходы человечества Структура и свойства экосистемы
Структура и свойства экосистемы Что такое экология? Тест №4
Что такое экология? Тест №4 Про воздух. Окружающий мир. 2 класс
Про воздух. Окружающий мир. 2 класс Природа в опасности
Природа в опасности Чырвоная кніга Рэспублікі Беларусь
Чырвоная кніга Рэспублікі Беларусь Аутэкология. Факторная экология. Общие закономерности действия факторов среды на живые организмы
Аутэкология. Факторная экология. Общие закономерности действия факторов среды на живые организмы Химическое загрязнение окружающей среды и его последствия
Химическое загрязнение окружающей среды и его последствия В Почепе нет хорошего хозяина
В Почепе нет хорошего хозяина Санитарная классификация промышленных предприятий
Санитарная классификация промышленных предприятий Игра День экологии
Игра День экологии Инженерная защита окружающей среды
Инженерная защита окружающей среды Природоохранные организации
Природоохранные организации Современные экологические проблемы
Современные экологические проблемы Основные экологические проблемы современности
Основные экологические проблемы современности Помощь водным объектам
Помощь водным объектам презентация по ИЗО
презентация по ИЗО Состояние природы
Состояние природы Аттестационная работа. Дополнительная общеобразовательная программа. Школьное лесничество
Аттестационная работа. Дополнительная общеобразовательная программа. Школьное лесничество Путешествие по природным паркам Югры. Заповедник Малая Сосьва
Путешествие по природным паркам Югры. Заповедник Малая Сосьва Ecology problems of metropolis
Ecology problems of metropolis