Содержание
- 2. 1H NMR SPECTROSCOPY
- 3. Метод я́дерного магни́тного резона́нса (ЯМР) основан на взаимодействии внешнего магнитного поля) основан на взаимодействии внешнего магнитного
- 4. Сабақтың мақсаты: Ядролық магнитті резонанс әдісімен танысу ЯМР қарапайым спектрлерімен танысу
- 5. Тілдік терминология Ядролық магнитті резонанс -magnetic nuclear resonance- ядерно магнитный резонанс
- 6. Samples are dissolved in solvents free of 1H atoms, e.g. CCl4, CDCl3. A small amount of
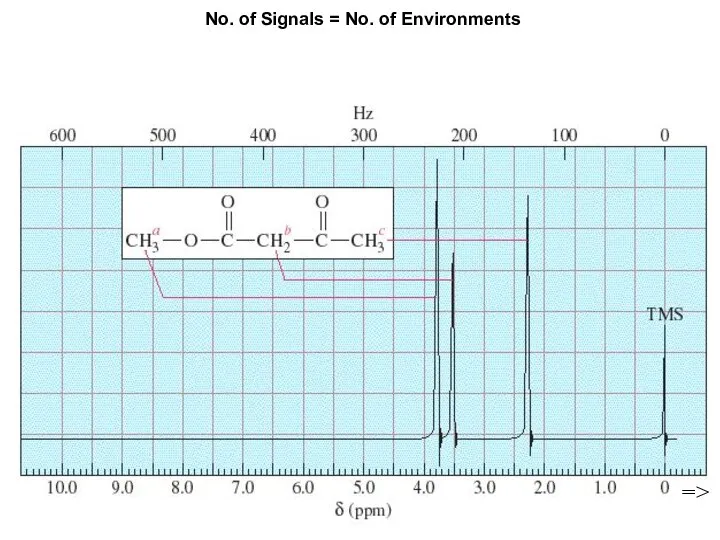
- 7. Chapter 13 No. of Signals = No. of Environments =>
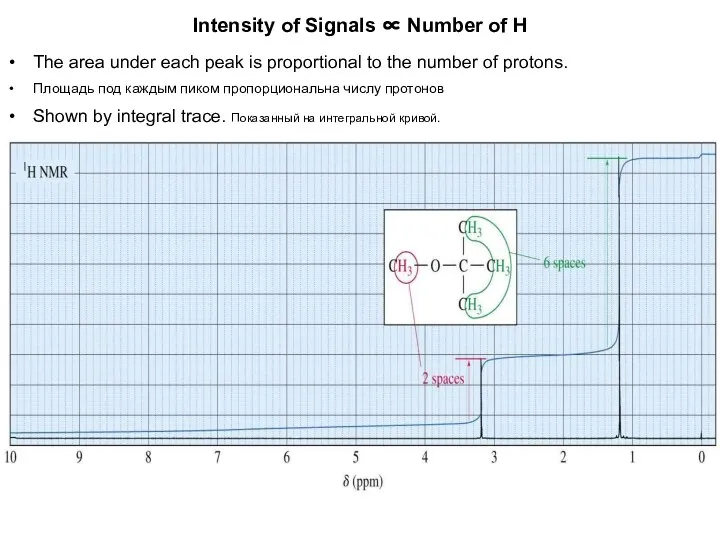
- 8. Intensity of Signals ∝ Number of H The area under each peak is proportional to the
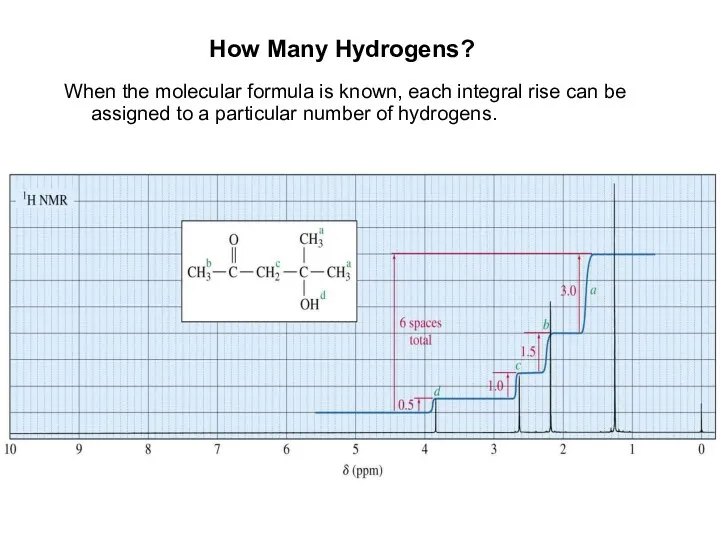
- 9. How Many Hydrogens? When the molecular formula is known, each integral rise can be assigned to
- 10. In a spectrum, there is one signal for each set of equivalent H atoms. В спектре
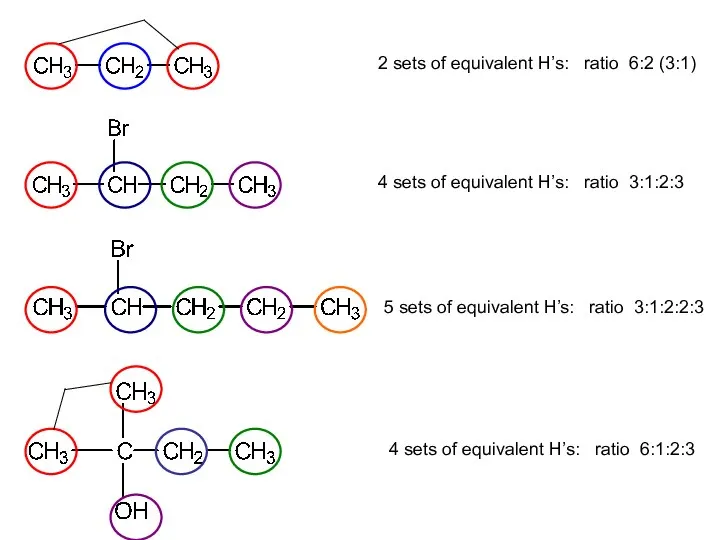
- 11. 2 sets of equivalent H’s: ratio 6:2 (3:1) 4 sets of equivalent H’s: ratio 3:1:2:3 5
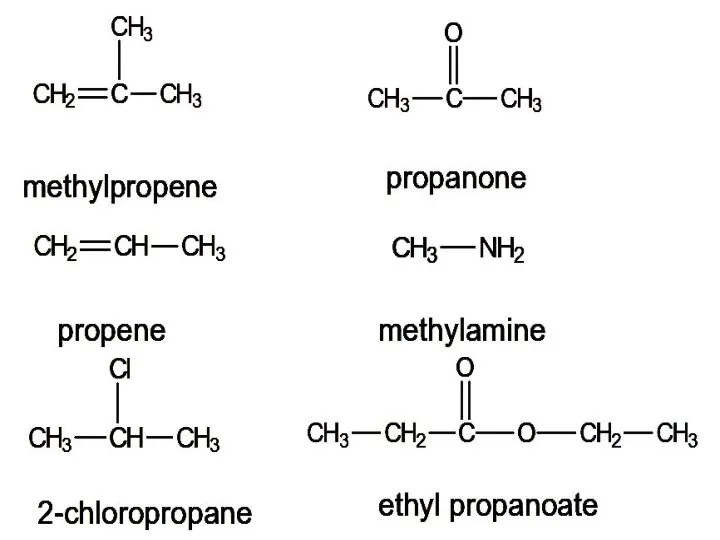
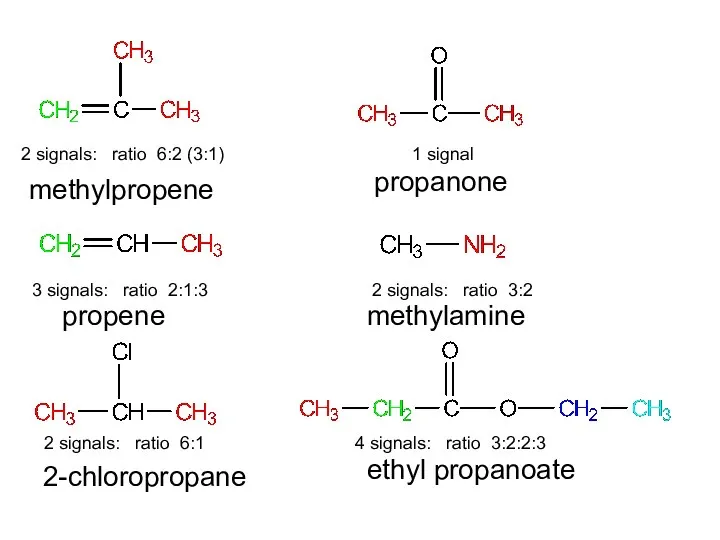
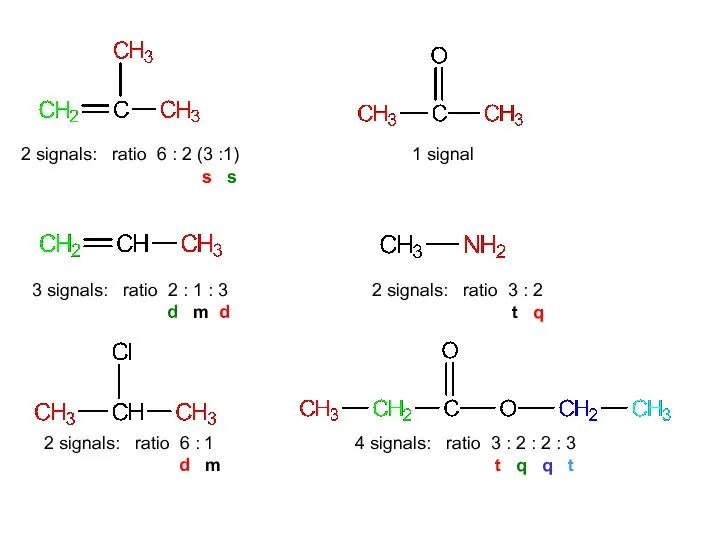
- 12. For each of the following compounds, predict the number of signals and the relative intensity of
- 13. methylpropene propene 2-chloropropane propanone methylamine ethyl propanoate
- 14. 2 signals: ratio 6:2 (3:1) 3 signals: ratio 2:1:3 2 signals: ratio 6:1 1 signal 2
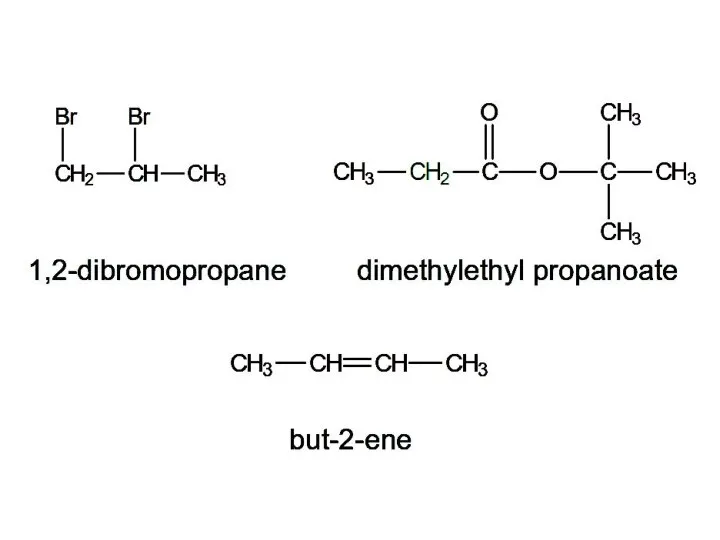
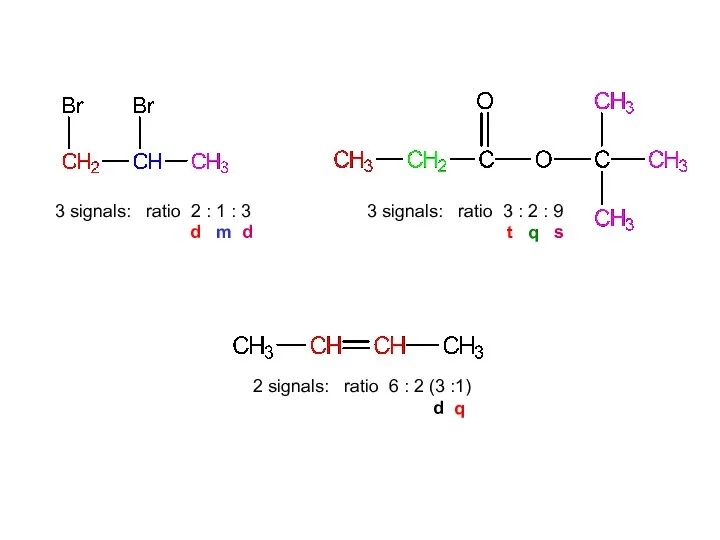
- 15. 1,2-dibromopropane dimethylethyl propanoate but-2-ene
- 16. 3 signals: ratio 2:1:3 2 signals: ratio 6:2 (3:1) 3 signals: ratio 3:2:9 1,2-dibromopropane dimethylethyl propanoate
- 17. RELATIVE INTENSITY There are four signals here – each has the same area and so represents
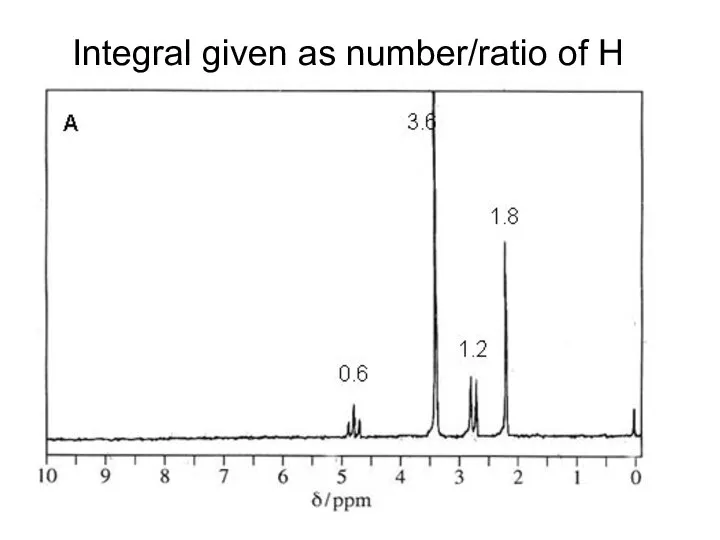
- 18. Integral given as number/ratio of H
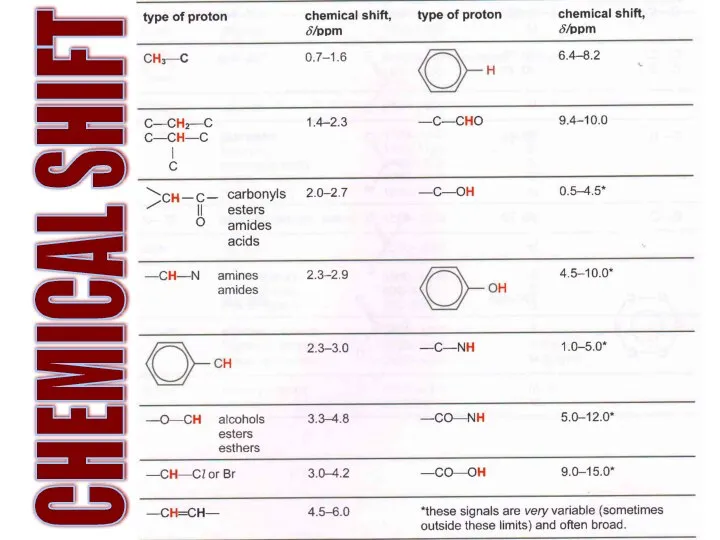
- 19. CHEMICAL SHIFT
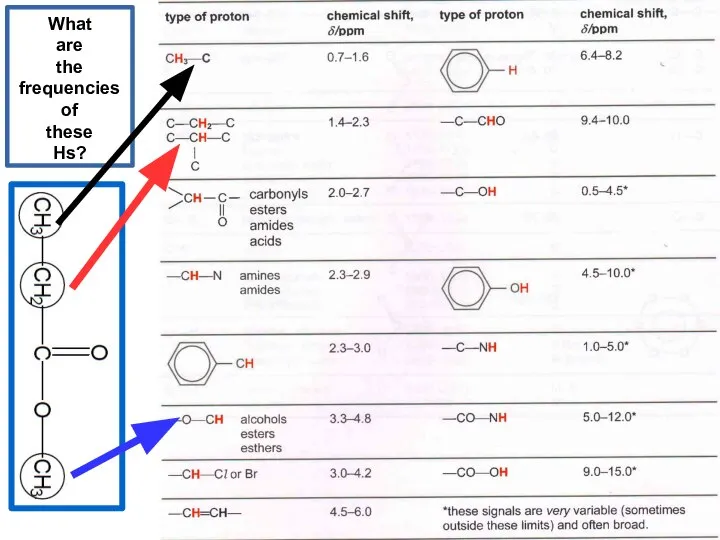
- 20. What are the frequencies of these Hs?
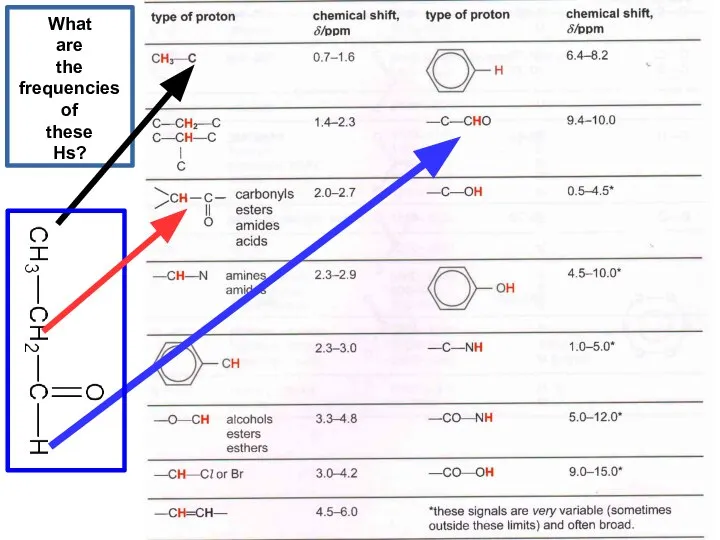
- 21. What are the frequencies of these Hs?
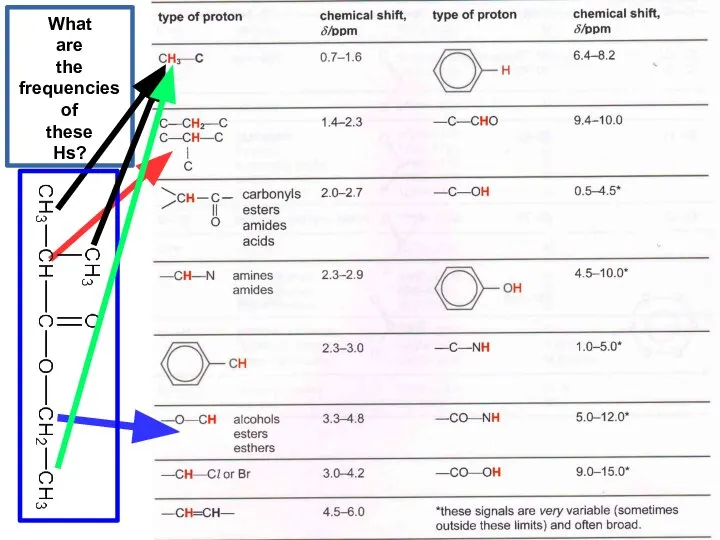
- 22. What are the frequencies of these Hs?
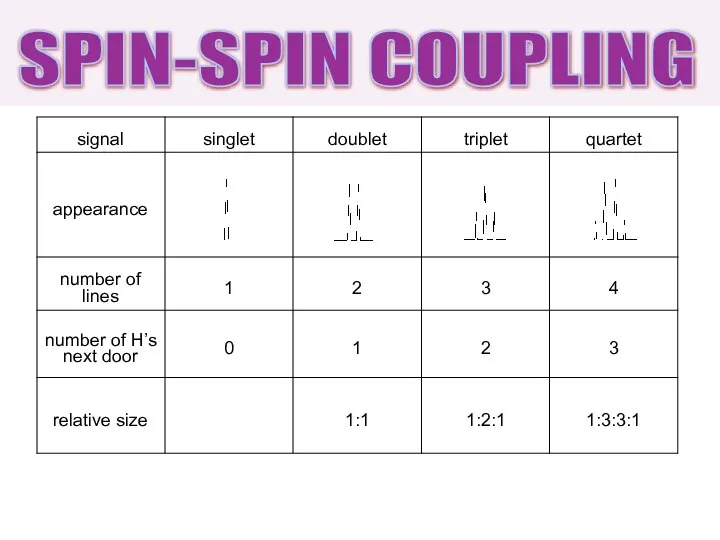
- 23. 0 H next door singlet (s) 1 H next door doublet (d) 2 H next door
- 24. SPIN-SPIN COUPLING
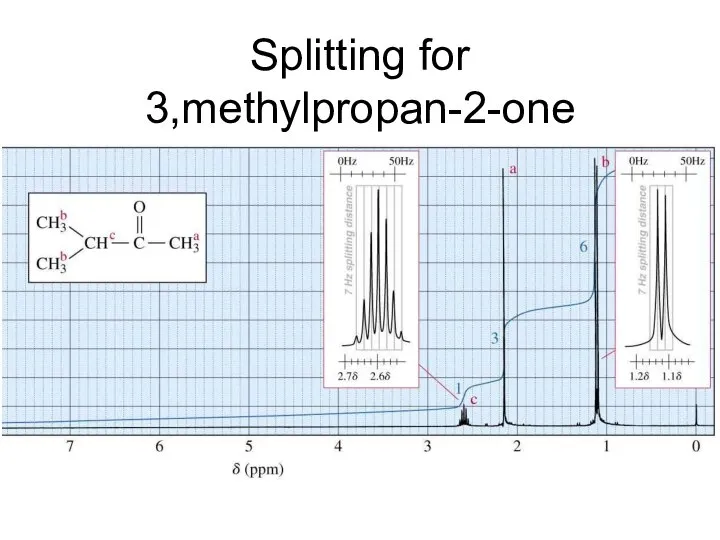
- 25. Splitting for 3,methylpropan-2-one
- 26. Number of H’s next door +1 But you don’t couple to H’s that are equivalent H’s
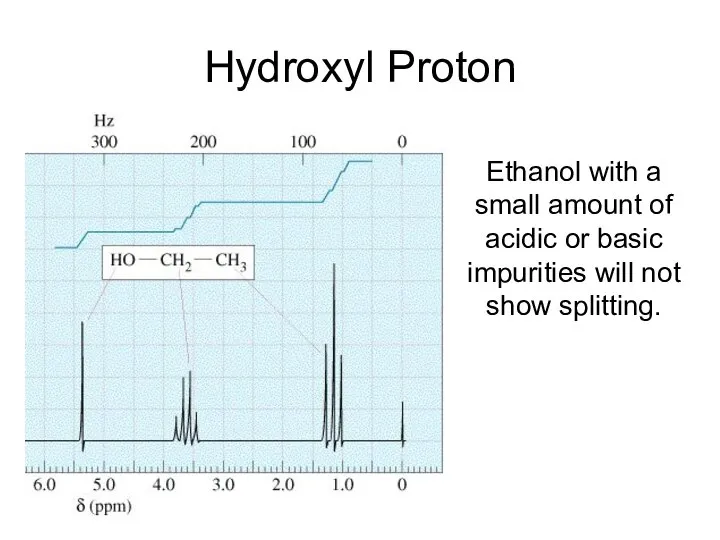
- 27. Hydroxyl Proton Ethanol with a small amount of acidic or basic impurities will not show splitting.
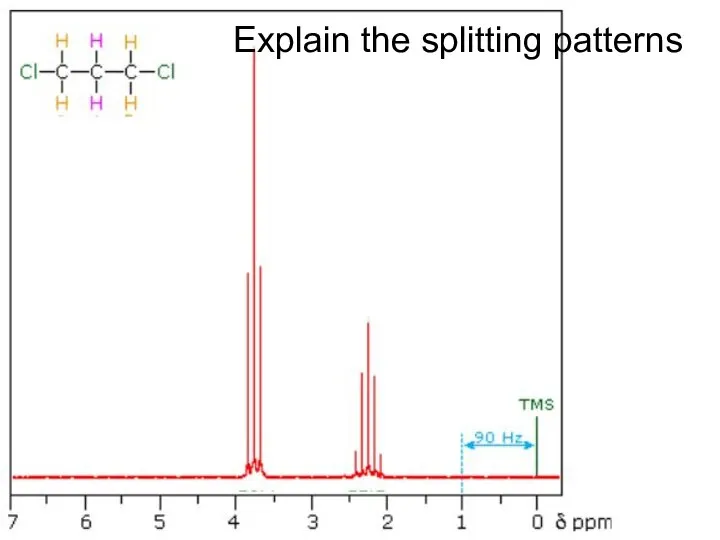
- 28. Explain the splitting patterns
- 30. Explain the splitting patterns
- 32. Explain the splitting patterns
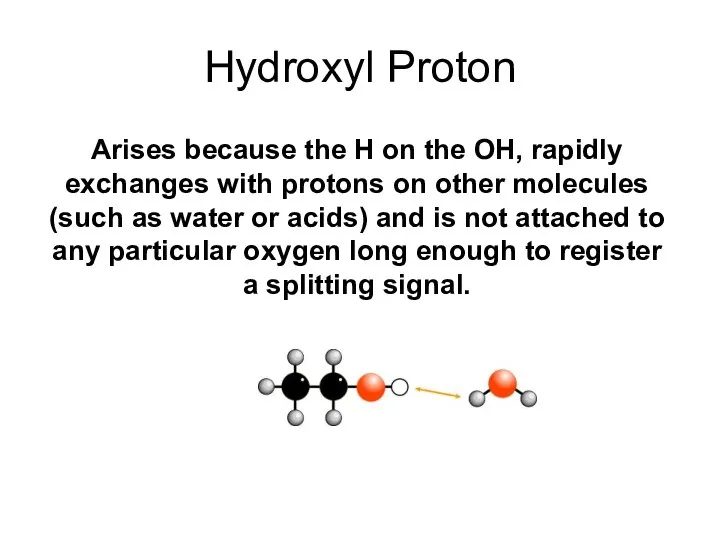
- 34. Hydroxyl Proton Arises because the H on the OH, rapidly exchanges with protons on other molecules
- 36. Identifying the O-H or N-H Peak Chemical shift will depend on concentration and solvent. To verify
- 38. Number of signals Position of signals Relative intensities Splitting how many different sets of equivalent H
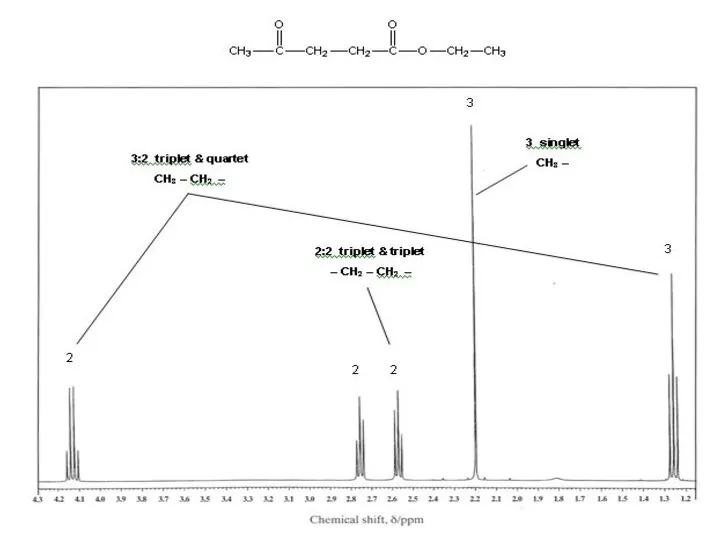
- 39. For each of the following compounds, predict the number of signals, the relative intensity of the
- 40. 2 signals: ratio 6 : 2 (3 :1) s s 3 signals: ratio 2 : 1
- 41. 3 signals: ratio 2 : 1 : 3 d m d 2 signals: ratio 6 :
- 43. 13C NMR SPECTROSCOPY
- 44. 13C NMR spectra are often simpler than 1H NMR spectra. They give a lot of valuable
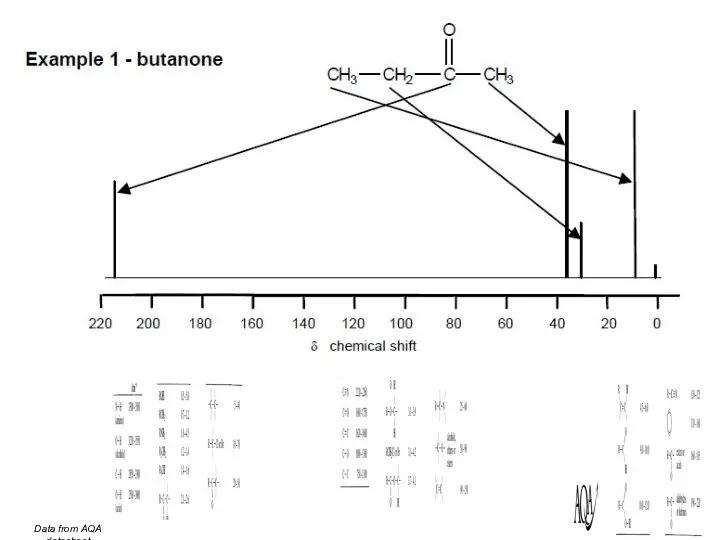
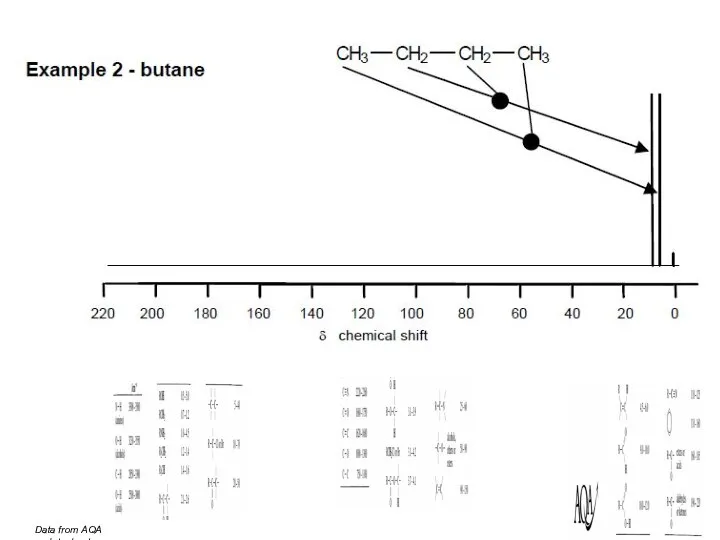
- 45. Data from AQA datasheet
- 46. Data from AQA datasheet
- 48. Скачать презентацию























 Гравитация. Всемирное тяготение
Гравитация. Всемирное тяготение Закон всемирного тяготения
Закон всемирного тяготения Колебания
Колебания Презентация Гальванический элемент
Презентация Гальванический элемент Георг Ом
Георг Ом Создание платформы для изучения формул по физике
Создание платформы для изучения формул по физике Презентация по физике "Основные сведения о строении атомов" - скачать
Презентация по физике "Основные сведения о строении атомов" - скачать  Квантовая теория поля
Квантовая теория поля Вплив електричного поля на живі організми
Вплив електричного поля на живі організми  Проект по физике на тему: «Экология, энергетика, человек»
Проект по физике на тему: «Экология, энергетика, человек» Презентацию готовили: Цыбенко Наташа и Ксенофонтова Юля
Презентацию готовили: Цыбенко Наташа и Ксенофонтова Юля  Архимедова
Архимедова История люминесцентной лампы
История люминесцентной лампы Распространение звука. Скорость звука. Физика 9 класс
Распространение звука. Скорость звука. Физика 9 класс  Транзисторы
Транзисторы Жылу энергетикада және жылу техникада жылу энергиясын қолдану
Жылу энергетикада және жылу техникада жылу энергиясын қолдану Физические и химические явления
Физические и химические явления Аттестационная работа. Тематическое планирование элективного курса «Физические законы вокруг нас»
Аттестационная работа. Тематическое планирование элективного курса «Физические законы вокруг нас» Проект: Вычисление реагентов расходуемых за год. год.
Проект: Вычисление реагентов расходуемых за год. год.  Основы медицинской акустики
Основы медицинской акустики Катушки индуктивности
Катушки индуктивности Элективный курс по теме: «Как делаются открытия в физике» мини-проект «Сборник задач по физике: Физика в быту»
Элективный курс по теме: «Как делаются открытия в физике» мини-проект «Сборник задач по физике: Физика в быту»  Плоскошлифовальные станки
Плоскошлифовальные станки Специальные методы микроскопии
Специальные методы микроскопии Лампа ДРВ, ДРЛ, ДРИ и ДНаТ
Лампа ДРВ, ДРЛ, ДРИ и ДНаТ Тема: «Магнитные бури и их влияние на здоровье человека и успеваемость школьников» Исследовательскую работу выполнил ученик 8 кла
Тема: «Магнитные бури и их влияние на здоровье человека и успеваемость школьников» Исследовательскую работу выполнил ученик 8 кла Ширина запрещенной зоны в полупроводниках и методы её расчета
Ширина запрещенной зоны в полупроводниках и методы её расчета Сделайте свое открытие в физике. Знаки препинания в предложениях с обособленными дополнениями. Контрольная работа №1
Сделайте свое открытие в физике. Знаки препинания в предложениях с обособленными дополнениями. Контрольная работа №1