Содержание
- 2. Physical change These are physical changes where there is no change in particles, just their arrangement
- 3. Chemical change These are examples of chemical changes where a chemical reaction takes place and a
- 4. Chemical reactions During chemical reactions the atoms (particles) rearrange to form a new substance. The signs
- 5. Activity 2.1: What changes are taking place in chemical reactions? Results are on the next slide.
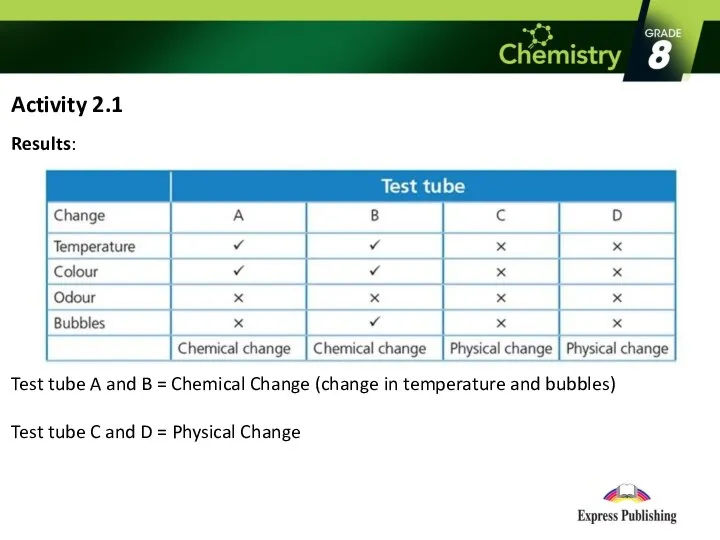
- 6. Activity 2.1 Results: Test tube A and B = Chemical Change (change in temperature and bubbles)
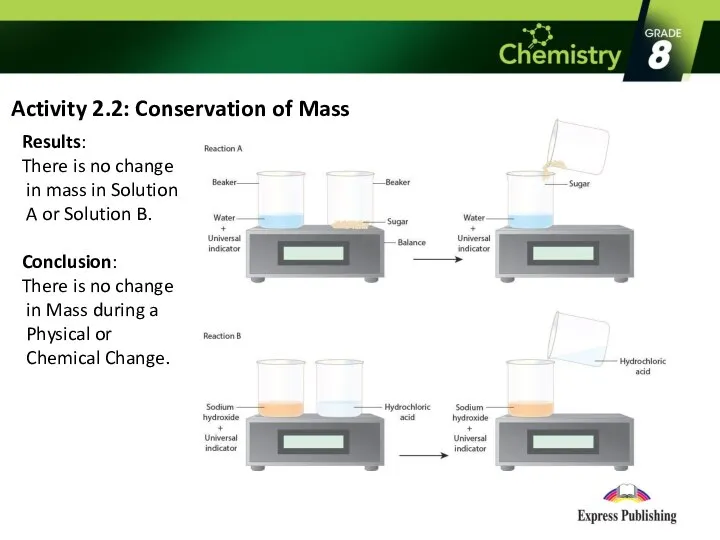
- 7. Activity 2.2: Conservation of Mass Results: There is no change in mass in Solution A or
- 8. Law of Conservation of Mass Antoine Lavoisier discovered that the mass of a substance cannot be
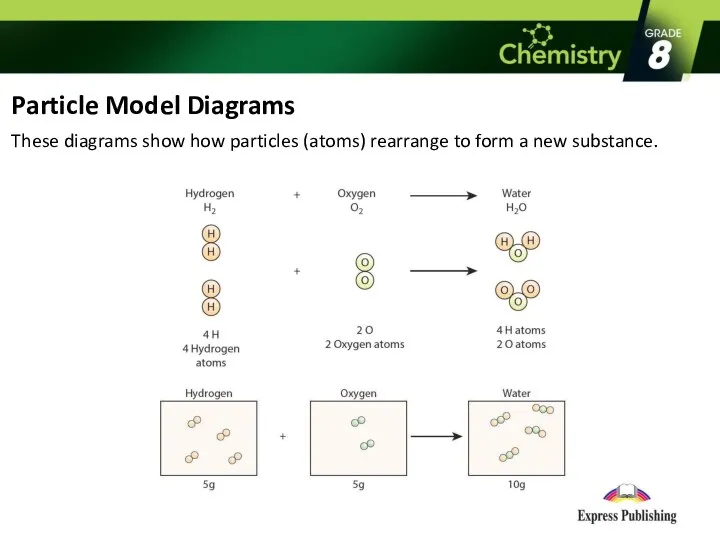
- 9. Particle Model Diagrams These diagrams show how particles (atoms) rearrange to form a new substance.
- 10. Particle Model Diagrams Draw a particle model diagram for below: Copper + Oxygen → Copper oxide
- 12. Скачать презентацию






 Дисперсные системы (продолжение)
Дисперсные системы (продолжение) Короткі відомості з історії хімії. Становлення хімії як науки
Короткі відомості з історії хімії. Становлення хімії як науки Солнечные батареи в космосе. (Задача 2)
Солнечные батареи в космосе. (Задача 2)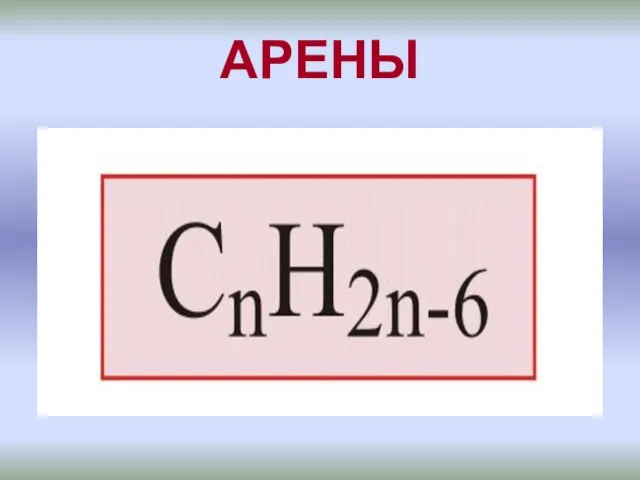 Бензоидные ароматические углеводороды - арены
Бензоидные ароматические углеводороды - арены Молекулярная биология. Белки нервной системы
Молекулярная биология. Белки нервной системы Катарометрический метод контроля
Катарометрический метод контроля Производство стекла
Производство стекла Процессы комплексообразования
Процессы комплексообразования Нефть. Состав и свойства
Нефть. Состав и свойства ФГОУ СПО «ЧПК» Выполнила: Гараева Оксана, студент группы №43 Руководители: Хасанова В.К., Чеснокова П.В.
ФГОУ СПО «ЧПК» Выполнила: Гараева Оксана, студент группы №43 Руководители: Хасанова В.К., Чеснокова П.В. Производные пурина
Производные пурина СЕРА S
СЕРА S Как осуществить цепочку превращений на основании положений теории электролитической диссоциации?
Как осуществить цепочку превращений на основании положений теории электролитической диссоциации? Применение этанола
Применение этанола Классификация химических реакций
Классификация химических реакций Великие химики России
Великие химики России Оксид азота(2) NO
Оксид азота(2) NO Повторение и обобщение знаний по темам "Металлы и сплавы."
Повторение и обобщение знаний по темам "Металлы и сплавы." Значение окислительно-восстановительных реакций в жизни человека
Значение окислительно-восстановительных реакций в жизни человека Аминокислоты Модели молекул
Аминокислоты Модели молекул Презентацию составила учитель МОУ «Ягринская гимназия» г.Северодвинска Шапошникова Т.С.
Презентацию составила учитель МОУ «Ягринская гимназия» г.Северодвинска Шапошникова Т.С.  Отрицательное влияние азотных минеральных удобрений на почву
Отрицательное влияние азотных минеральных удобрений на почву Green chemistry application to intermolecular forces
Green chemistry application to intermolecular forces Химическая связь
Химическая связь Неорганические соединения галогенов
Неорганические соединения галогенов Особенности формирования естественно-научной картины мира на уроках географии и химии
Особенности формирования естественно-научной картины мира на уроках географии и химии Камни и Козерог
Камни и Козерог Хімія і медицина
Хімія і медицина