Содержание
- 2. Thermodynamics Thermodynamics is the study of the effects of work, heat, and energy on a system
- 3. Getting Started All of thermodynamics can be expressed in terms of four quantities Temperature (T) Internal
- 4. Classical vs Statistical Classical thermodynamics concerns the relationships between bulk properties of matter. Nothing is examined
- 5. Introduction According to British scientist C. P. Snow, the three laws of thermodynamics can be (humorously)
- 6. 1.0 You can’t win (1st law) The first law of thermodynamics is an extension of the
- 7. Slide courtesy of NASA
- 8. 1.1 Process Terminology Adiabatic – no heat transferred Isothermal – constant temperature Isobaric – constant pressure
- 9. 1.1.1 Adiabatic Process An adiabatic process transfers no heat therefore Q = 0 ΔU = Q
- 10. 1.1.2 Isothermal Process An isothermal process is a constant temperature process. Any heat flow into or
- 11. 1.1.3 Isobaric Process An isobaric process is a constant pressure process. ΔU, W, and Q are
- 12. 1.1.4 Isochoric Process An isochoric process is a constant volume process. When the volume of a
- 13. 1.2 Heat Capacity The amount of heat required to raise a certain mass of a material
- 14. 1.2.1 Heat Capacity of Ideal Gas CV = heat capacity at constant volume CV = 3/2
- 15. 2.0 You can’t break even (2nd Law) Think about what it means to not “break even”.
- 16. Slide courtesy of NASA
- 17. 2.1 Concerning the 2nd Law The second law of thermodynamics introduces the notion of entropy (S),
- 18. 2.2 Implications of the 2nd Law Time marches on If you watch a movie, how do
- 19. 2.3 Direction of a Process The 2nd Law helps determine the preferred direction of a process
- 20. 2.4 Heat Engine A device which transforms heat into work is called a heat engine This
- 21. 2.4.1 Cycles It is beyond the scope of this presentation, but here would be a good
- 22. 2.4.2 The Carnot Cycle Image from Keta - Wikipedia
- 23. 2.4.2.1 Carnot explained Curve A (1 → 2): Isothermal expansion at TH Work done by the
- 24. 2.4.2.2 Area under PV curve The area under the PV curve represents the quantity of work
- 25. 2.5 Engine Efficiency The thermal efficiency of a heat engine is e = 1 + QC/QH
- 26. 2.6 Practical Uses Automobile engines, refrigerators, and air conditioners all work on the principles laid out
- 27. 3.0 You can’t get out (3rd Law) No system can reach absolute zero This is one
- 28. 3.1 Implications of 3rd Law MIT researchers achieved 450 picokelvin in 2003 (less than ½ of
- 29. 4.0 The Zeroth Law The First and Second Laws were well entrenched when an additional Law
- 30. Slide courtesy of NASA
- 32. Скачать презентацию





























 Электрические трёхфазные цепи
Электрические трёхфазные цепи Реактивное движение Урок в 9 классе Сидорина Ольга Павловна учитель физики ГБОУ СОШ 371 г.Москва 2013год
Реактивное движение Урок в 9 классе Сидорина Ольга Павловна учитель физики ГБОУ СОШ 371 г.Москва 2013год Оптическая система. Лучевая и волновая оптика
Оптическая система. Лучевая и волновая оптика Пленочные наноматериалы и технологии их получения
Пленочные наноматериалы и технологии их получения Грузовые электромобили
Грузовые электромобили Законы Ома и Кирхгофа
Законы Ома и Кирхгофа Подшипники. Конструкция подшипника скольжения
Подшипники. Конструкция подшипника скольжения Сейсморазведка. Разложение сейсмических колебаний
Сейсморазведка. Разложение сейсмических колебаний Телеграфная связь
Телеграфная связь Будова атома. Закріпити знання про будову атома. Узагальнити знання про стан електронів у атомі
Будова атома. Закріпити знання про будову атома. Узагальнити знання про стан електронів у атомі Бернулли теңдеуі
Бернулли теңдеуі Принцип Гюйгенса в виде формулы Кирхгофа
Принцип Гюйгенса в виде формулы Кирхгофа Электрическое поле в проводниках и дилектриках
Электрическое поле в проводниках и дилектриках Магнетизм. Магнитное поле
Магнетизм. Магнитное поле Коливальний рух у природі і техніці
Коливальний рух у природі і техніці Сложный четырехполюсник (задача)
Сложный четырехполюсник (задача) Лекция 12
Лекция 12 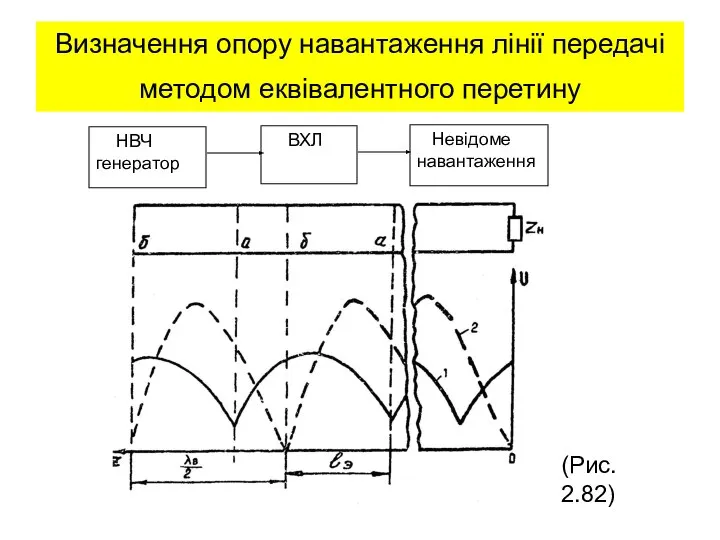 Визначення опору навантаження лінії передачі методом еквівалентного перетину. (Лекция 15)
Визначення опору навантаження лінії передачі методом еквівалентного перетину. (Лекция 15) 25.8. Элементы зонной теории твердого тела
25.8. Элементы зонной теории твердого тела Гипотеза де Бройля. Комптоновская длина волны и длина волны де Бройля
Гипотеза де Бройля. Комптоновская длина волны и длина волны де Бройля Законы термодинамики
Законы термодинамики Ядерный магнитный резонанс
Ядерный магнитный резонанс Силы в природе
Силы в природе Презентация Виды сил 7 класс
Презентация Виды сил 7 класс Расчеты на прочность механических конструкций
Расчеты на прочность механических конструкций Подпрыгивающая вода
Подпрыгивающая вода Примеры решения задач по теме Движение с постоянным ускорением свободного падения
Примеры решения задач по теме Движение с постоянным ускорением свободного падения Датчики давления
Датчики давления