Содержание
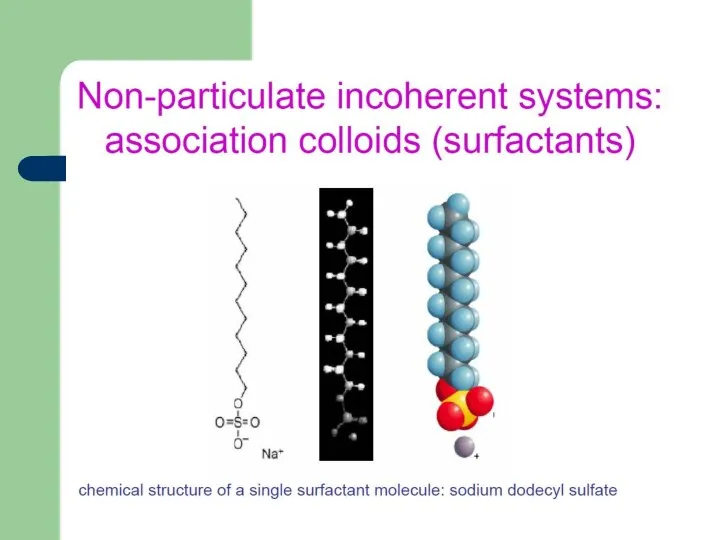
- 2. Colloidal Chemistry A colloid is a substance microscopically dispersed evenly throughout another substance. The designation ‘‘colloid’’
- 4. Colloidal Science an interdisciplinary intersection dealing with colloids, heterogeneous systems consisting of a mechanical mixture of
- 6. Surface tension
- 7. Dispersed liquids in nature
- 9. Minimal surfaces Costa’s minimal surface (1982) Catenoid Helicoid
- 10. There is an extra energy associated with a surface Molecules on the surface are less bounded
- 11. Surface phenomena The increased surface area of the phase separation is associated with the transition of
- 12. Surface tension σ is the work required for the creation of 1 m2 of surface [σ]=
- 13. Surface tension Surface tension depends on: the nature of fluid σ(Н2О)=72,8 J/m2; σ(serum)=45,4 J/m2). temperature (↑t
- 14. The surface tension can be thought of as a force F A The energy per unit
- 15. A didactic setup (Cf. Wilhelmy plate)
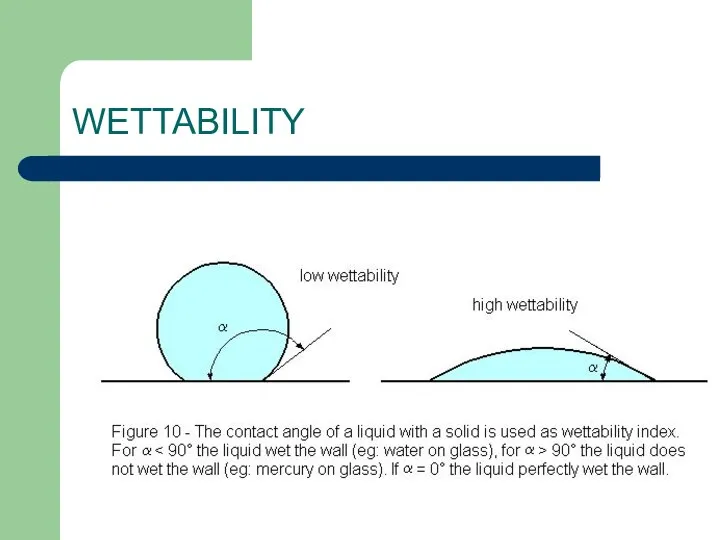
- 16. WETTABILITY Why does one fabric absorb water well while another seems to refuse it? Why does
- 17. WETTABILITY
- 18. Cohesion (n. lat. cohaerere "stick or stay together") or cohesive attraction or cohesive force is the
- 19. CAPILLARITY
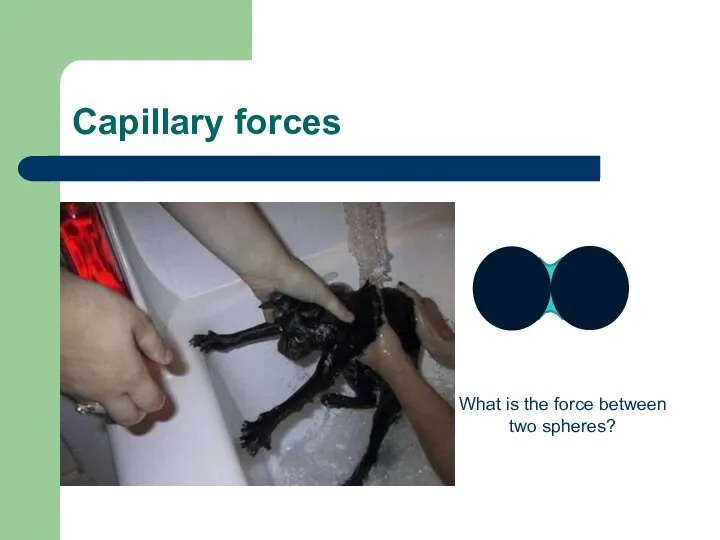
- 20. Capillary forces What is the force between two spheres?
- 21. SORPTION
- 22. Medical & biological importance: Assimilation of nutrients and drugs Transfer of O2 and CO2 from the
- 23. Sorption -change in the concentration of one or more components in heterogeneous systems Sorption - the
- 24. Adsorption Adsorption is spontaneous change of component concentration in the surface layer compared to the volume
- 25. Gibbs Equation G - the amount of adsorbed substance [mole/m2] а – equilibrium activity of the
- 26. Surface activity The ability of the solute to change surface tension is called surface activity (γ)
- 27. Traube-Duclos rule: When extending the chain-CH2 - in homological series of surface activity increases in 3-3,5
- 28. SAS, SIS, SNS Surface-active substances (SAS): reduce σ solvent. σ solution O. SAS: alcohols, organic acids,
- 29. The isotherm of surface tension The dependence of σ on the concentration the dissolved substance at
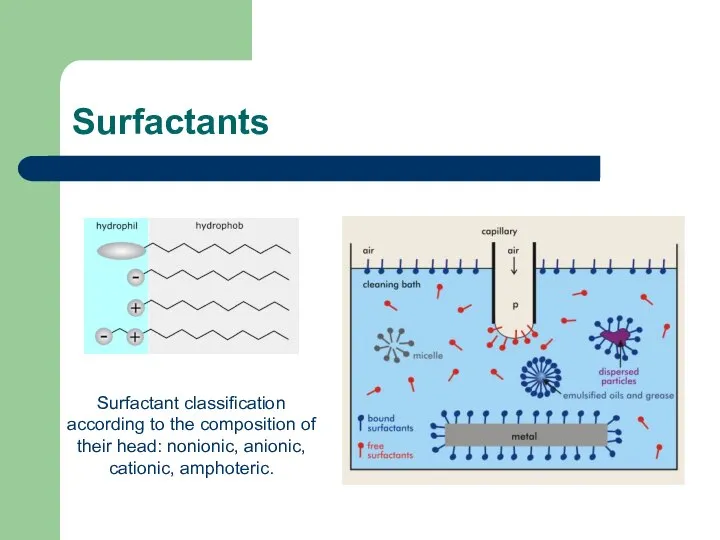
- 31. Surfactants Surfactant classification according to the composition of their head: nonionic, anionic, cationic, amphoteric.
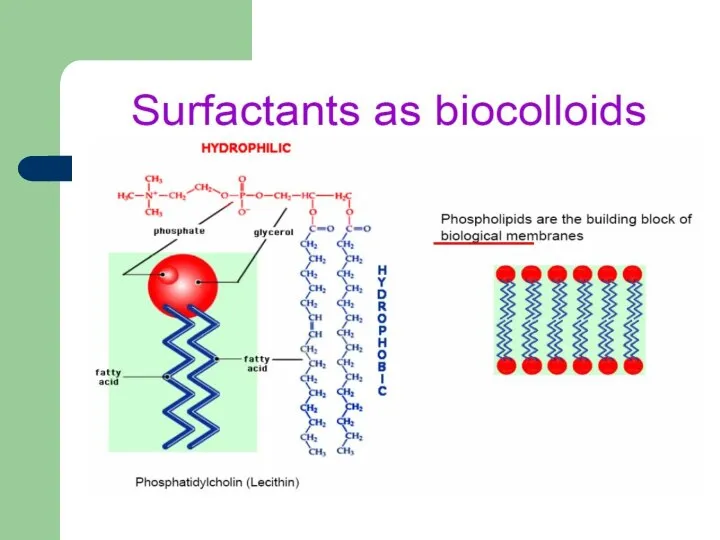
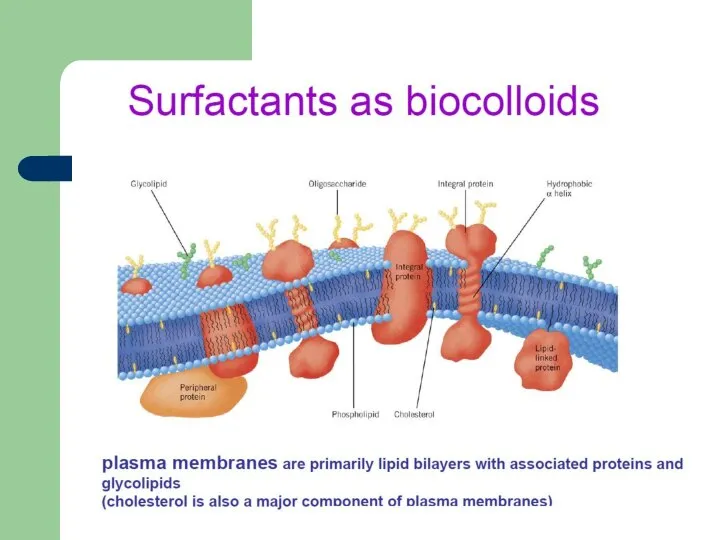
- 35. Surfactants in nature Pulmonary surfactants
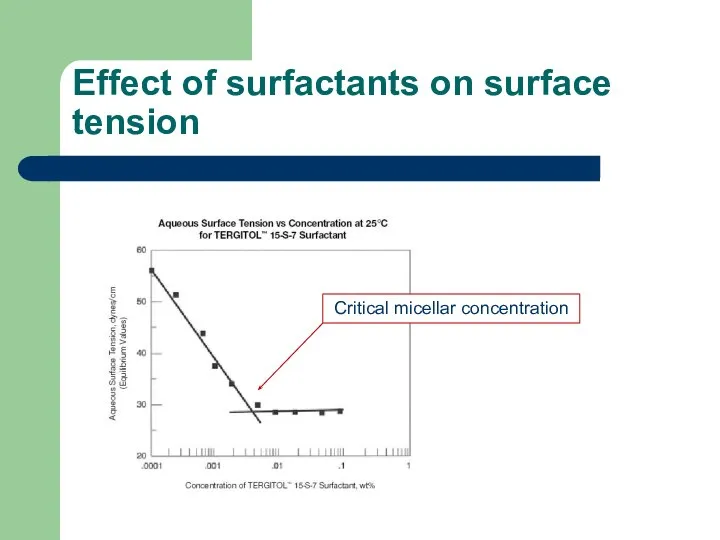
- 36. Effect of surfactants on surface tension Critical micellar concentration
- 37. What is the pressure in a soap bubble ? « Amusons nous sur la terre comme
- 38. What can you tell about the pressure in a liquid from the shape of a rising
- 39. ADSORPTION ON THE LIQUID-GAS BORDER
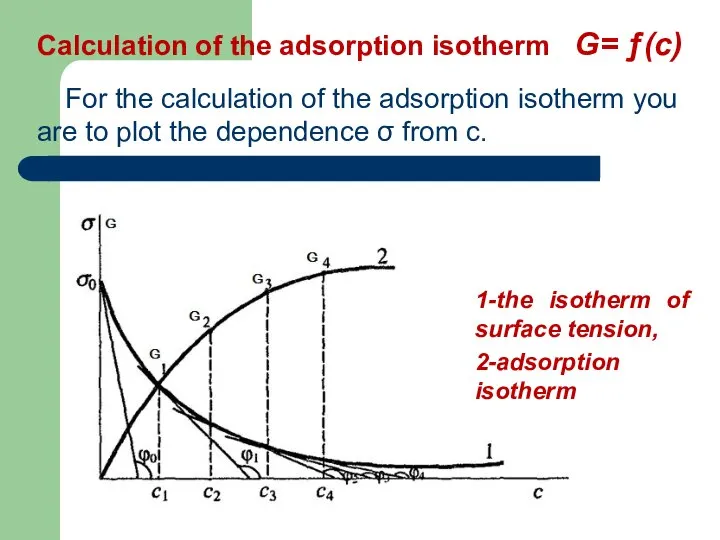
- 40. Calculation of the adsorption isotherm G= ƒ(с) For the calculation of the adsorption isotherm you are
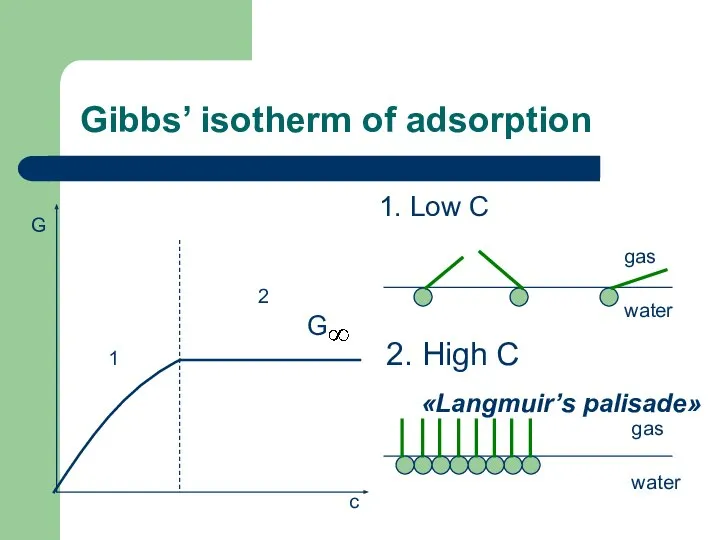
- 41. Gibbs’ isotherm of adsorption 1. Low С 1 2 G c gas water 2. High С
- 42. ADSORPTION ON THE SOLID-GAS BORDER
- 43. Adsorption by solids The adsorption value depends on: 1. The size of the adsorbent surface if
- 44. Freundlich equation А = x/m = k · p1/n G = x/m = КF · Cn
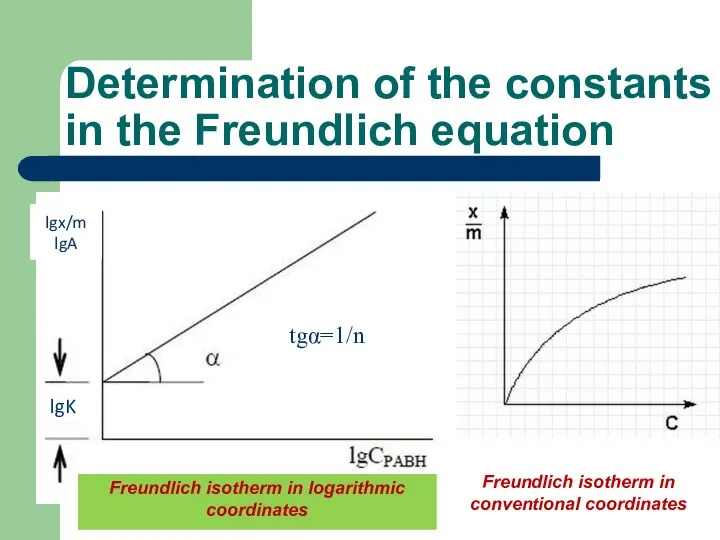
- 45. Determination of the constants in the Freundlich equation lg A = lg k + 1/n lg
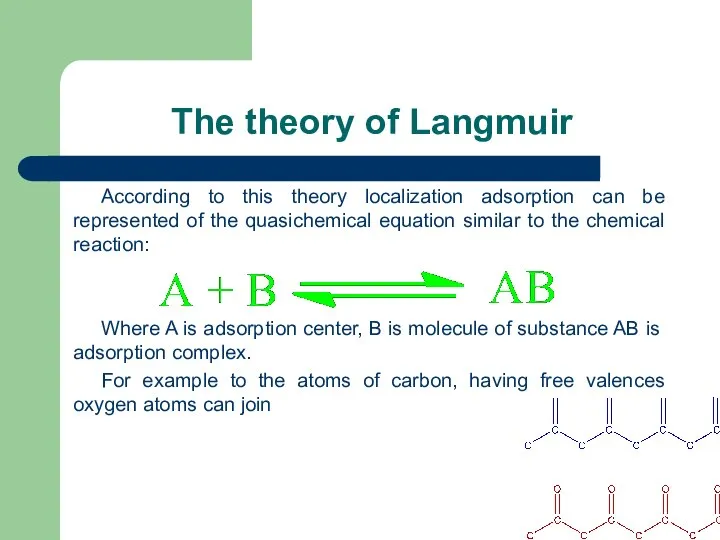
- 46. The theory of Langmuir 1) On each absorption place on the surface of the adsorbent can
- 47. The theory of Langmuir According to this theory localization adsorption can be represented of the quasichemical
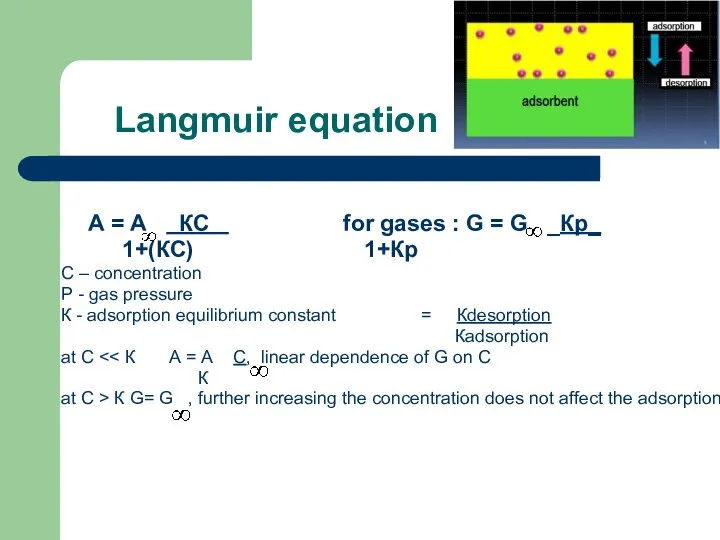
- 48. Langmuir equation А = А КС for gases : G = G _Кр_ 1+(КС) 1+Кр С
- 49. To find the constants A ∞ and K linear formula of Langmuir equation is used. Substituting
- 50. Langmuir’s isotherm of adsorption с G
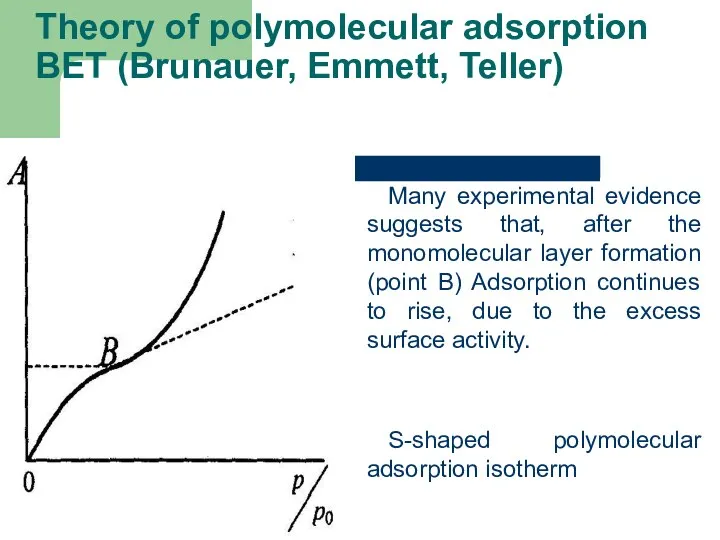
- 51. Theory of polymolecular adsorption BET (Brunauer, Emmett, Teller) Many experimental evidence suggests that, after the monomolecular
- 52. ADSORPTION ON THE BORDER OF SOLID – SOLUTION In the study of adsorption from solutions on
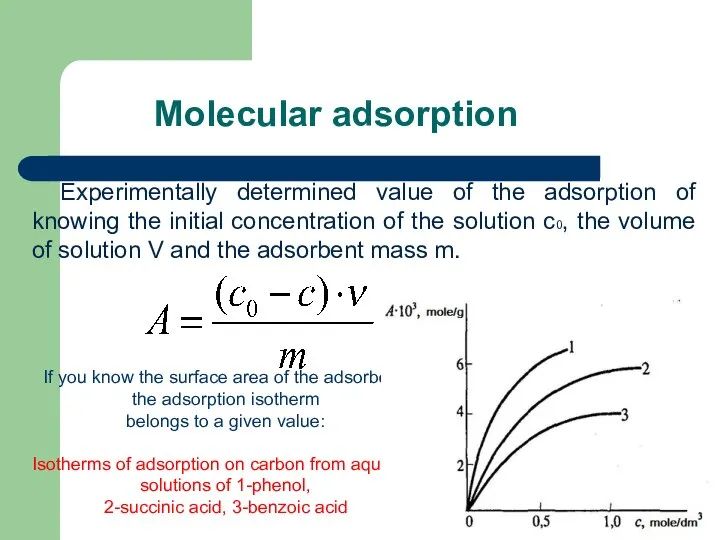
- 53. Molecular adsorption Experimentally determined value of the adsorption of knowing the initial concentration of the solution
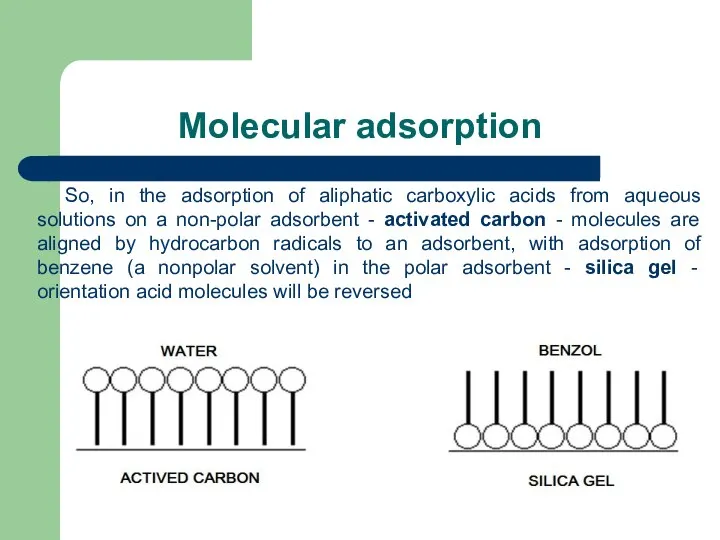
- 54. Molecular adsorption So, in the adsorption of aliphatic carboxylic acids from aqueous solutions on a non-polar
- 55. Conclusion From the above that is confirmed, that: For adsorption SAS from the nonpolar or low-polar
- 56. The ion exchange adsorption The ion exchange adsorption - a process in which the adsorbent is
- 57. Chromatography Chromatography is dynamic method of analysis based on multiply repeated processes of sorption and desorption.
- 58. Chromatography is physical chemical method used to separate substances analytical objectives formulations objectives Used for identification
- 59. From the history of chromatography Mikhail Semenovich Tsvet (1872—1919) Birthday of chromatography – 21.03.1903 The report
- 60. «No other discovery had such a huge long lasting effect in organic chemistry as the analysis
- 61. The principle of chromatographic separation of substances The stationary phase The mobile phase Molecules of substances
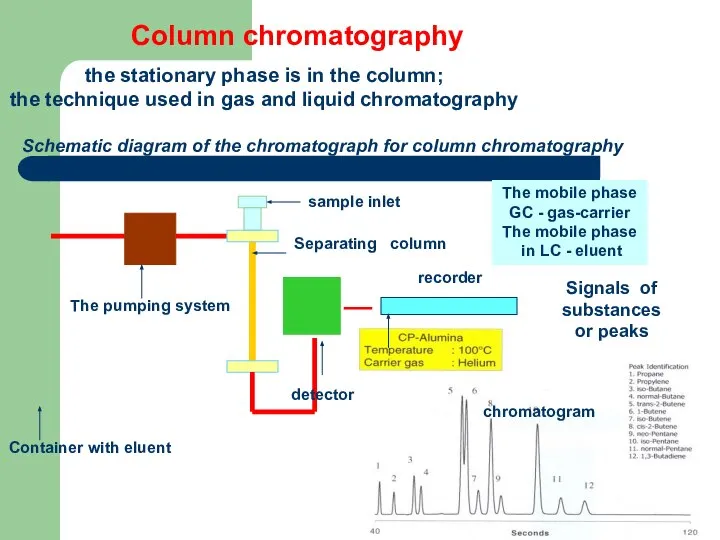
- 62. Column chromatography the stationary phase is in the column; the technique used in gas and liquid
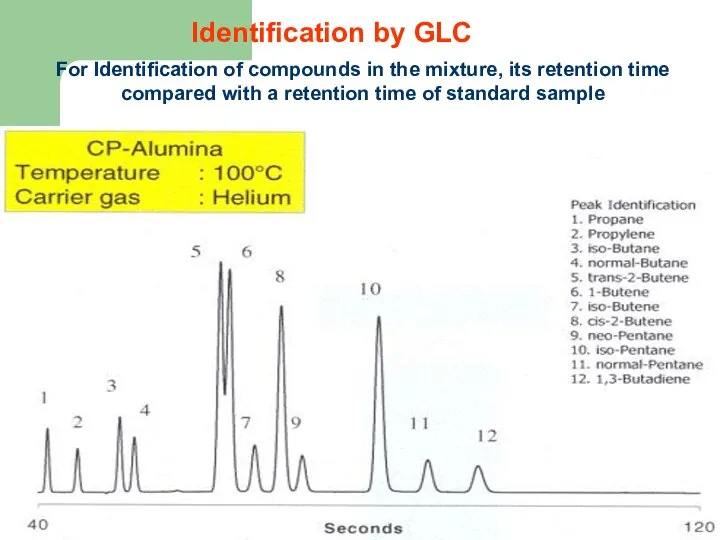
- 63. Identification by GLC For Identification of compounds in the mixture, its retention time compared with a
- 64. HPLC Agilent Technologies
- 65. HPLC Milichrom
- 66. HPLC HP
- 67. GLC “Agilent Technologies”
- 68. Enterosorption It is method of treatment of various diseases, based on the ability of enterosorbents bind
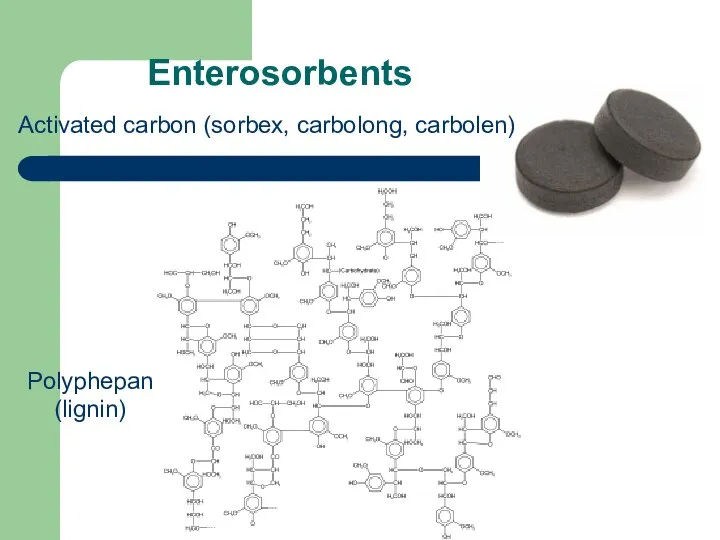
- 69. Enterosorbents Polyphepan (lignin) Activated carbon (sorbex, carbolong, carbolen)
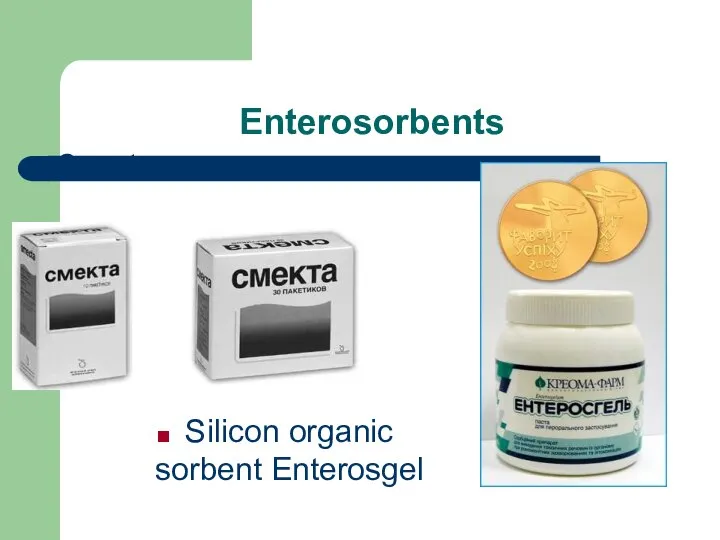
- 70. Enterosorbents Smecta Silicon organic sorbent Enterosgel
- 71. Enterosorption Enterosorption is part of efferent therapy (from the Latin word efferens means output). Also enterosorption,
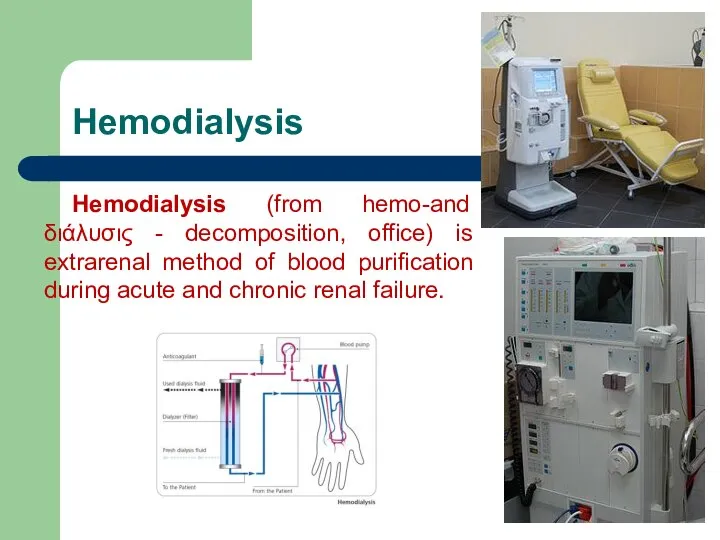
- 72. Hemodialysis Hemodialysis (from hemo-and διάλυσις - decomposition, office) is extrarenal method of blood purification during acute
- 73. Peritoneal dialysis Peritoneal dialysis (dialysis Greek expansion, separation) is method of cleansing the blood of endogenous
- 74. Plasmapheresis Membrane plasma filter contains a chamber for the flow of blood separated from the cameras
- 76. Скачать презентацию





















![Gibbs Equation G - the amount of adsorbed substance [mole/m2] а](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/1404189/slide-24.jpg)































 Диены (диолефины, алкадиены)
Диены (диолефины, алкадиены) Конденсированные гетероциклические соединения. Пурины
Конденсированные гетероциклические соединения. Пурины Атомно – молекулярное учение
Атомно – молекулярное учение Получение меламина из карбамида
Получение меламина из карбамида Chemical equilibrium. (Chapter 3)
Chemical equilibrium. (Chapter 3) Понятие об адсорбционной влаге
Понятие об адсорбционной влаге Вспененные полимерные материалы. Классификация и принцип действия вспенивателей
Вспененные полимерные материалы. Классификация и принцип действия вспенивателей Презентация по Химии "Отбеливатели" - скачать смотреть
Презентация по Химии "Отбеливатели" - скачать смотреть  Монокристаллы CdTe и твердые растворы на его основе. Свойства, получение, применение
Монокристаллы CdTe и твердые растворы на его основе. Свойства, получение, применение Кислые породы умеренно-щелочного ряда
Кислые породы умеренно-щелочного ряда Углерод. Кремний
Углерод. Кремний Формальдегід. Будова та шкідливість
Формальдегід. Будова та шкідливість Мұнай. Мұнайдың шығу тарихы
Мұнай. Мұнайдың шығу тарихы Выделение урана из растворов (пульп)
Выделение урана из растворов (пульп) Средства и методы стерилизации
Средства и методы стерилизации Физические явления в химии. Чистые вещества и смеси
Физические явления в химии. Чистые вещества и смеси Кислоты, их состав и название. Цель урока: 1. Сформировать понятия о кислотах. 2. Рассмотреть состав, название и классификацию ки
Кислоты, их состав и название. Цель урока: 1. Сформировать понятия о кислотах. 2. Рассмотреть состав, название и классификацию ки Кислотно-основное титрование
Кислотно-основное титрование ГБОУ СОШ № 1981
ГБОУ СОШ № 1981 Склеивание древесины. Клеи
Склеивание древесины. Клеи Методы термического обезвреживания промышленных газов
Методы термического обезвреживания промышленных газов Молярный объём
Молярный объём Применение метода кислотно-основного титрования в количественном анализе химических веществ и лекарственных средств. (Лекция 7)
Применение метода кислотно-основного титрования в количественном анализе химических веществ и лекарственных средств. (Лекция 7) Алканы. Гомологи, изомеры, номенклатура.
Алканы. Гомологи, изомеры, номенклатура.  Геохимические методы исследований
Геохимические методы исследований Сера и её соединения
Сера и её соединения Презентация по Химии "Хлор - физиологическая и патологическая роль в организме человека" - скачать смотреть бесплатно
Презентация по Химии "Хлор - физиологическая и патологическая роль в организме человека" - скачать смотреть бесплатно Кристаллооптический метод в петрографии
Кристаллооптический метод в петрографии