Содержание
- 3. What does rate of reaction mean? The speed of different chemical reactions varies hugely. Some reactions
- 4. Rates of reaction Why are some reactions faster than others?
- 5. Reactions, particles and collisions Reactions take place when particles collide with a certain amount of energy.
- 6. Changing the rate of reactions increased temperature increased concentration of dissolved reactants, and increased pressure of
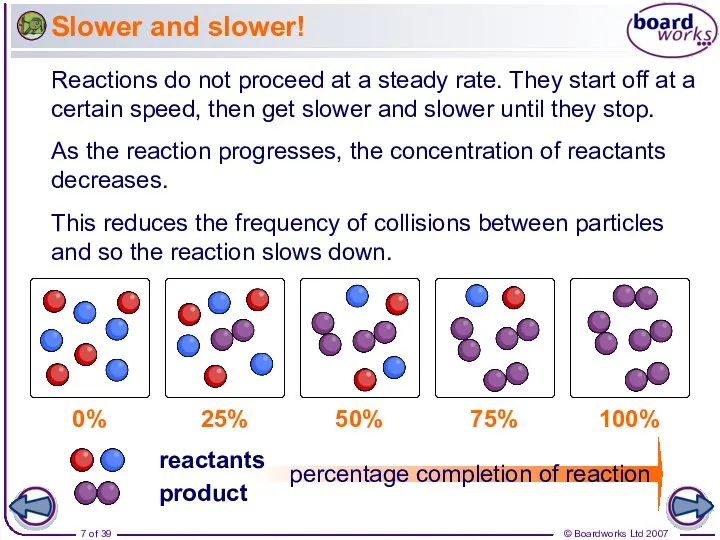
- 7. Slower and slower! Reactions do not proceed at a steady rate. They start off at a
- 8. Graphing rates of reaction
- 9. Reactant–product mix
- 10. How can rate of reaction be measured? Measuring the rate of a reaction means measuring the
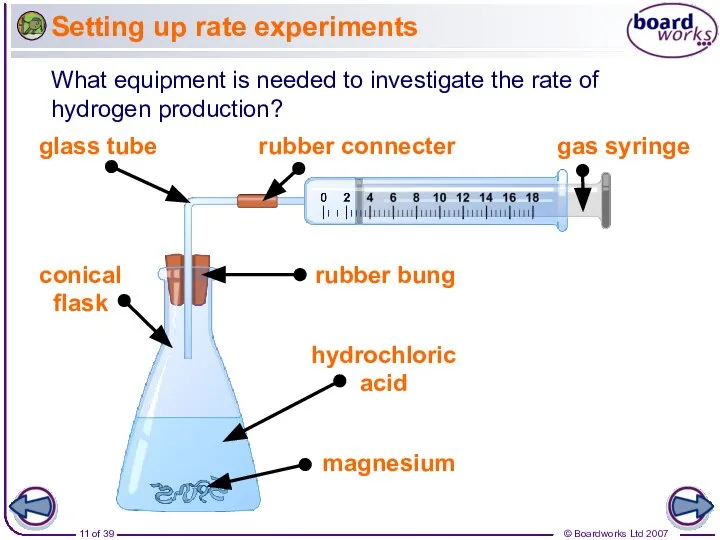
- 11. Setting up rate experiments What equipment is needed to investigate the rate of hydrogen production?
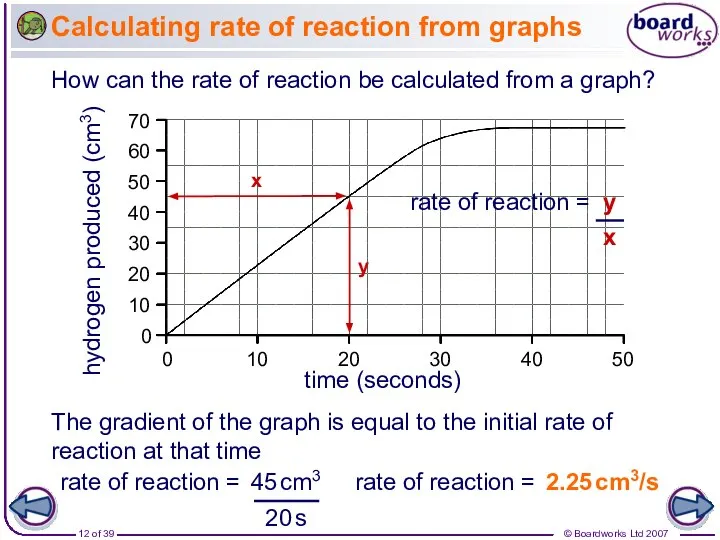
- 12. hydrogen produced (cm3) time (seconds) 10 20 30 40 50 10 20 30 40 50 60
- 13. The reactant/product mix
- 14. Collisions and reactions: summary
- 16. Temperature and collisions How does temperature affect the rate of particle collision?
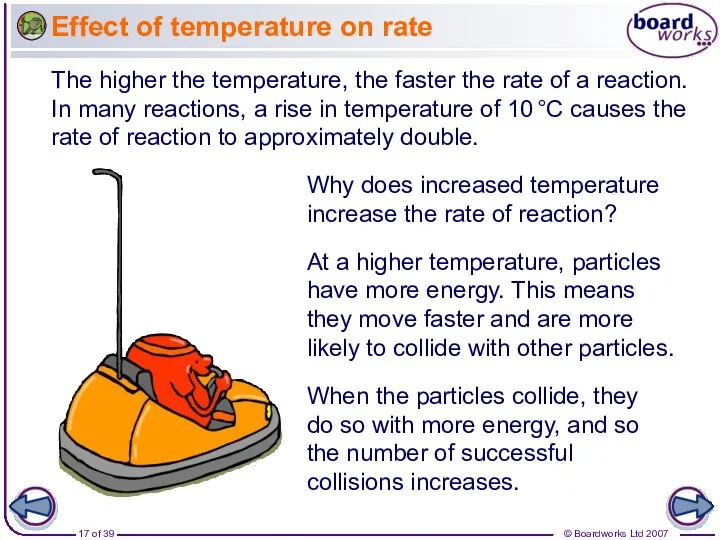
- 17. Effect of temperature on rate The higher the temperature, the faster the rate of a reaction.
- 18. Temperature and particle collisions
- 19. Temperature and batteries Why are batteries more likely to rundown more quickly in cold weather? At
- 20. How does temperature affect rate? The reaction between sodium thiosulfate and hydrochloric acid produces sulfur. Sulfur
- 21. The effect of temperature on rate
- 23. Effect of concentration on rate of reaction The higher the concentration of a dissolved reactant, the
- 24. Concentration and particle collisions
- 25. The effect of concentration on rate
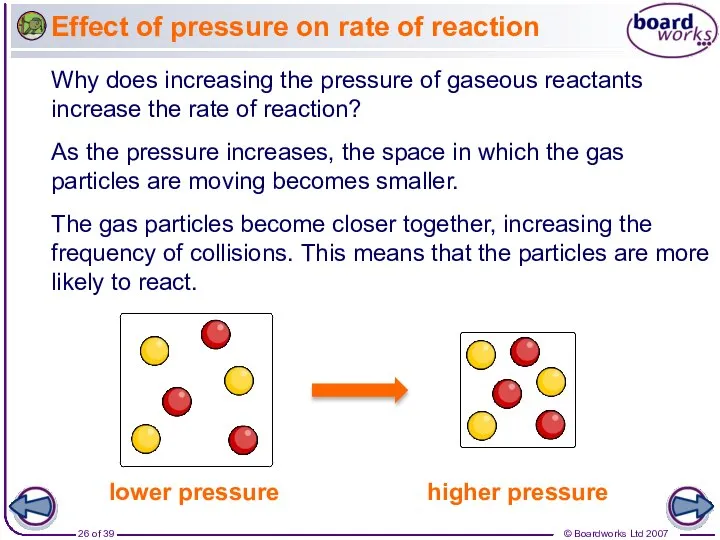
- 26. Effect of pressure on rate of reaction The gas particles become closer together, increasing the frequency
- 28. Effect of surface area on rate of reaction Any reaction involving a solid can only take
- 29. Surface area and particle collisions
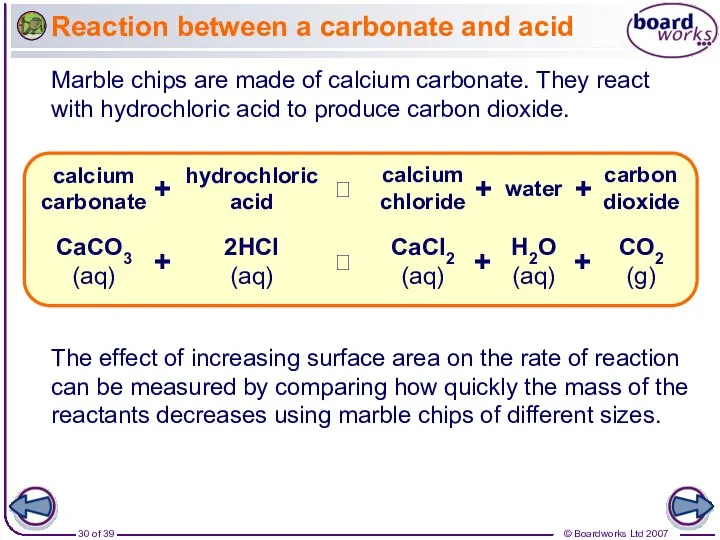
- 30. Reaction between a carbonate and acid Marble chips are made of calcium carbonate. They react with
- 31. The effect of surface area on rate
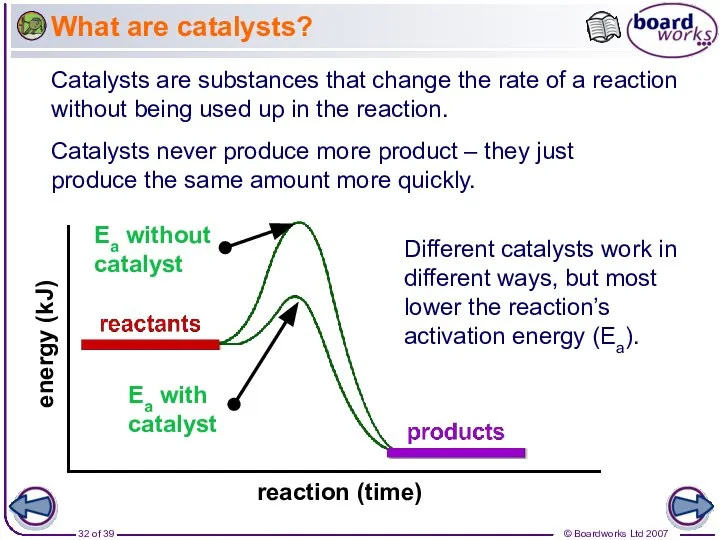
- 32. What are catalysts? Catalysts are substances that change the rate of a reaction without being used
- 33. Everyday catalysts Nickel is a catalyst in the production of margarine (hydrogenation of vegetable oils). Many
- 34. Catalysts in industry Catalysts are also essential for living cells. Biological catalysts are special types of
- 36. Glossary activation energy – The amount of energy needed to start a reaction. catalyst – A
- 37. Anagrams
- 38. Rates of reaction: summary
- 40. Скачать презентацию



























 Химия и здоровье
Химия и здоровье Термохимия. Термодинамические параметры
Термохимия. Термодинамические параметры Презентация по Химии "общая характеристика металлов" - скачать смотреть
Презентация по Химии "общая характеристика металлов" - скачать смотреть  Алкены. Состав, строение, свойства и химические превращения
Алкены. Состав, строение, свойства и химические превращения Оксиды, их свойства, получение
Оксиды, их свойства, получение Природные источники углеводородов
Природные источники углеводородов Химическая связь
Химическая связь Токсикологически важные вещества, подвергающиеся обязательному судебно-химическому исследованию
Токсикологически важные вещества, подвергающиеся обязательному судебно-химическому исследованию Перекисное окисление липидов. Характеристика, продукты, биологическая и патофизиологическая роль
Перекисное окисление липидов. Характеристика, продукты, биологическая и патофизиологическая роль Неметаллы, особенности строения
Неметаллы, особенности строения Кристаллическая решетка
Кристаллическая решетка Химические реакторы
Химические реакторы Теория строения
Теория строения Презентация Алкены
Презентация Алкены Исследование минералов в параллельном свете с одним поляризатором
Исследование минералов в параллельном свете с одним поляризатором Водородная связь
Водородная связь Кислородсодержащие органические соединения
Кислородсодержащие органические соединения Соединения химических элементов
Соединения химических элементов Нефть и газ. Химический состав и свойства
Нефть и газ. Химический состав и свойства Теория создание стекла, оптические кристаллы
Теория создание стекла, оптические кристаллы Непредельные углеводороды
Непредельные углеводороды Степень окисления
Степень окисления Термохимия. Закон Гесса и его следствия
Термохимия. Закон Гесса и его следствия Використання неметалів та їх сполук , кругообіг неметалів у природі
Використання неметалів та їх сполук , кругообіг неметалів у природі  Синтетикалық пиретроидтар. Инсектицидтердің тиомочевиндер, нейтротоксиндер, гормоналды, авермектиндер, карбаматтар
Синтетикалық пиретроидтар. Инсектицидтердің тиомочевиндер, нейтротоксиндер, гормоналды, авермектиндер, карбаматтар Шампуни: классификация по составу и назначению
Шампуни: классификация по составу и назначению Основы кристаллографии
Основы кристаллографии Химия элементов IVA группы
Химия элементов IVA группы