Содержание
- 2. Lecture 6 Heat flow and the first law of thermodynamics. Kind of thermodynamic process. Adiabatic processes.
- 3. Heat When the temperature of a thermal system in contact with a neighboring system changes, we
- 4. Mechanical equivalent of heat Mechanical energy is not conserserved in the presence of nonconservative forces. It
- 5. Specific heat capacity The heat capacity C of a particular sample of a substance is defined
- 6. Energy transfer and specific heat capacity From this definition, we can relate the energy Q transferred
- 8. Dependence of specific heat capacity on temperature Specific heat varies with temperature. For example, the specific
- 9. Dependence of specific heat capacity on volume and pressure Measured values of specific heats are found
- 10. Phase transition It can be that transfer of energy does not result in a change in
- 11. Latent heat Quantitative measure of phase transition is latent heat L: Q=±mL Latent heat of fusion
- 13. State variables - Thermodynamic process - Thermal equilibrium We describe the state of a system using
- 14. Work and heat in thermodynamic process The total work done by the gas as its volume
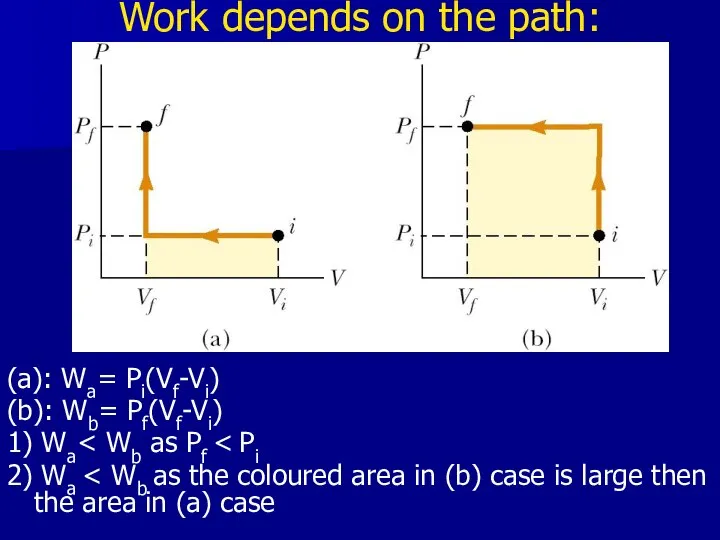
- 15. Work depends on the path: (a): Wa= Pi(Vf-Vi) (b): Wb= Pf(Vf-Vi) 1) Wa 2) Wa
- 16. Two ways of energy transfer There exist two ways in which energy can be transferred between
- 17. The First Law of Thermodynamics The change in internal energy ΔU of the system is equal
- 18. The first law of thermodynamics is a special case of the law of conservation of energy
- 19. Ideal Gas Processes Here W is work done by the system, ΔQ - heat flow into
- 20. Adiabatic (no heat flow, Q=0): ΔW = -ΔU The curve of adiabatic process is described by
- 21. Polytropic processes PVγ = const, γ=const. Isobaric γ=0 Isotermic γ=1 Adiabatic γ= CP/CV Isochoric γ=∞
- 22. Cyclic Processes If a nonisolated system is performing a cyclic process, the change in the internal
- 24. Скачать презентацию




















 Устройство и техническое обслуживание ходовой системы сортиментовоза Камаз
Устройство и техническое обслуживание ходовой системы сортиментовоза Камаз Своя игра 1
Своя игра 1 Тормозной путь
Тормозной путь След Архимеда Работу выполнили Ученики 6 «А», «Б» класса М О У школы №93 Варлашин Сергей Александрович ; Лисецкий Илья Анатольевич
След Архимеда Работу выполнили Ученики 6 «А», «Б» класса М О У школы №93 Варлашин Сергей Александрович ; Лисецкий Илья Анатольевич Кригинг в петрофизическом моделировании. Алгоритм кригинга
Кригинг в петрофизическом моделировании. Алгоритм кригинга Викторина, посвящённая юбилею М. В. Ломоносова
Викторина, посвящённая юбилею М. В. Ломоносова Решение задач. Импульс, энергия, законы сохранения
Решение задач. Импульс, энергия, законы сохранения Конструкция тележек
Конструкция тележек Термодинамические потоки. Явление переноса в газах: диффузия, теплопроводность и вязкость. Эффузия в разреженном газе
Термодинамические потоки. Явление переноса в газах: диффузия, теплопроводность и вязкость. Эффузия в разреженном газе Индуктивные элементы
Индуктивные элементы Виды диэлектриков и их поляризация
Виды диэлектриков и их поляризация Припої. Загальні властивості
Припої. Загальні властивості Презентация по физике "Колебательный контур. Свободные и вынужденные колебания. Резонанс" - скачать
Презентация по физике "Колебательный контур. Свободные и вынужденные колебания. Резонанс" - скачать  Учитель: Попова И.А. МОУ СОШ № 30 Белово 2010
Учитель: Попова И.А. МОУ СОШ № 30 Белово 2010  Развитие взглядов на природу света. Принцип Гюйгенса. Закон отражения света. Закон преломления света
Развитие взглядов на природу света. Принцип Гюйгенса. Закон отражения света. Закон преломления света Электропроводность диэлектриков
Электропроводность диэлектриков Формулы и величины
Формулы и величины Аттестация 1. Электродинамика
Аттестация 1. Электродинамика Люминесцентные лампы
Люминесцентные лампы Червячная передача
Червячная передача Дифракция света. Тема 8
Дифракция света. Тема 8 ЕСДП
ЕСДП Простейшие задачи в координатах
Простейшие задачи в координатах Обнаружение магнитного поля по его действию на электрический ток. Правило левой руки
Обнаружение магнитного поля по его действию на электрический ток. Правило левой руки Законы Ньютона
Законы Ньютона  Аттестационная работа. Физический практикум в 11 классе с использованием лабораторного оборудования
Аттестационная работа. Физический практикум в 11 классе с использованием лабораторного оборудования Тормоза грузовых и пассажирских вагонов
Тормоза грузовых и пассажирских вагонов Электрический ток и его использование. 8 класс
Электрический ток и его использование. 8 класс