Содержание
- 2. Олег Борисович Птицын (1929-1999)
- 3. PROTEIN PHYSICS LECTURE 1 Introduction & overview
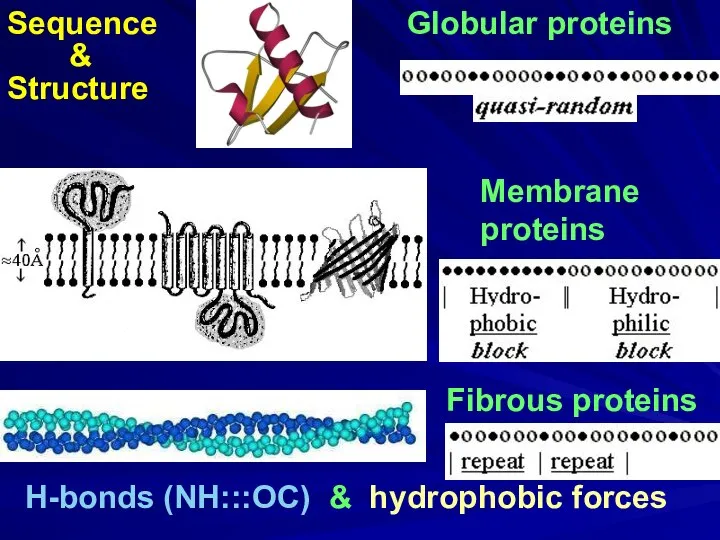
- 4. Globular proteins Fibrous proteins H-bonds (NH:::OC) & hydrophobic forces Membrane proteins
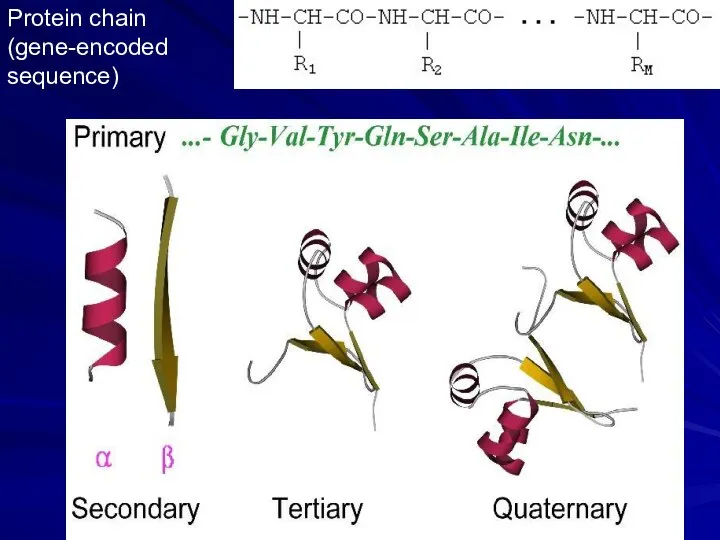
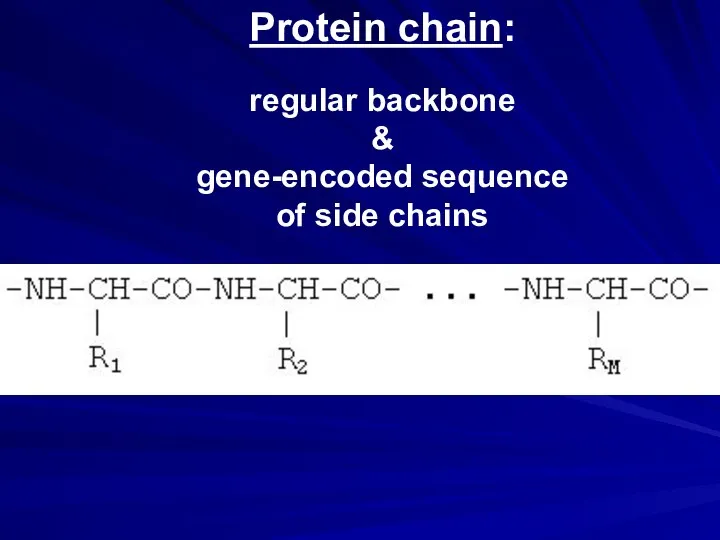
- 5. Protein chain (gene-encoded sequence)
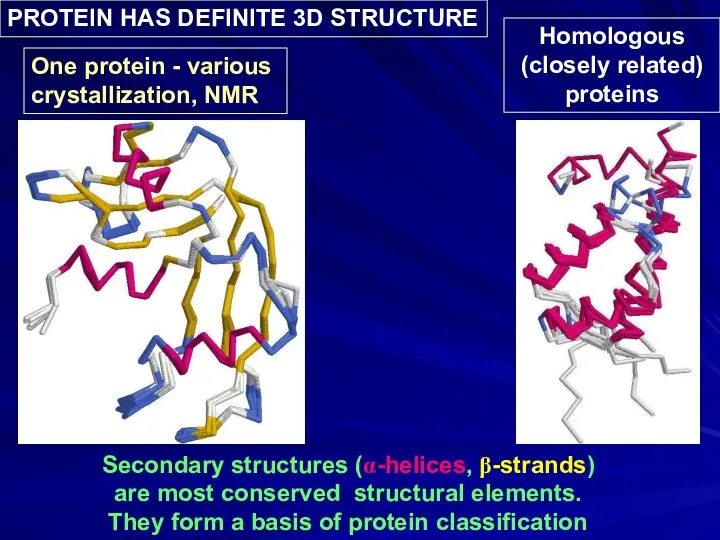
- 6. Secondary structures (α-helices, β-strands) are most conserved structural elements. They form a basis of protein classification
- 9. Globular proteins Fibrous proteins H-bonds (NH:::OC) & hydrophobic forces Membrane proteins Sequence & Structure
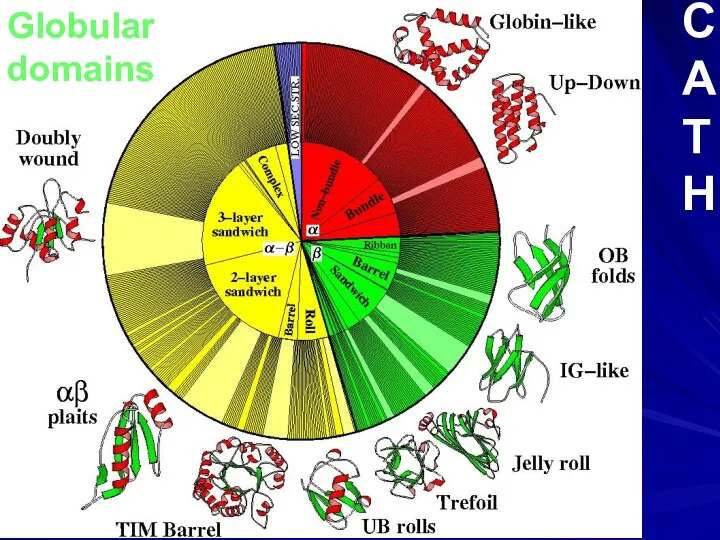
- 10. Globular domains C A T H
- 11. PROTEIN CHAIN CAN FORM ITS UNIQUE 3D STRUCTURE SPONTANEOUSLY IN VITRO
- 12. phase separation
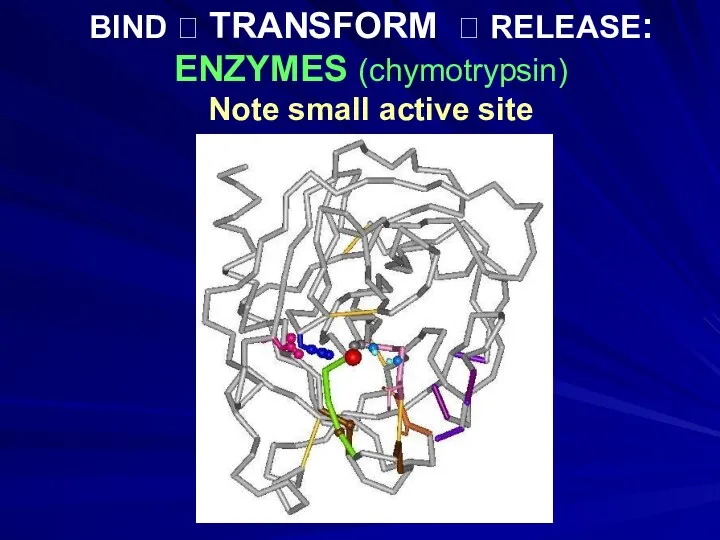
- 13. BIND ? TRANSFORM ? RELEASE: ENZYMES (chymotrypsin) Note small active site
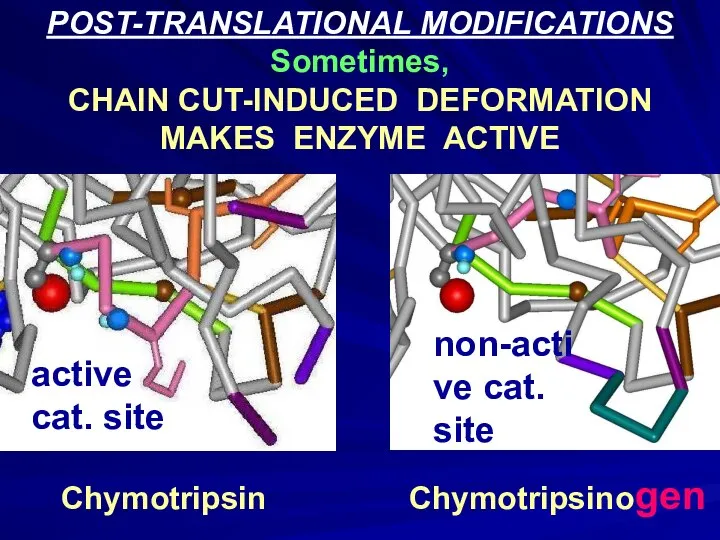
- 14. POST-TRANSLATIONAL MODIFICATIONS Sometimes, CHAIN CUT-INDUCED DEFORMATION MAKES ENZYME ACTIVE Chymotripsin Chymotripsinogen active cat. site non-active cat.
- 15. POST-TRANSLATIONAL MODIFICATIONS: (especially in eukaryotes): PROTEIN CHAIN CUTS (proteolysis), - SPLICING (inteins) - CYCLIZATION - INTERNAL
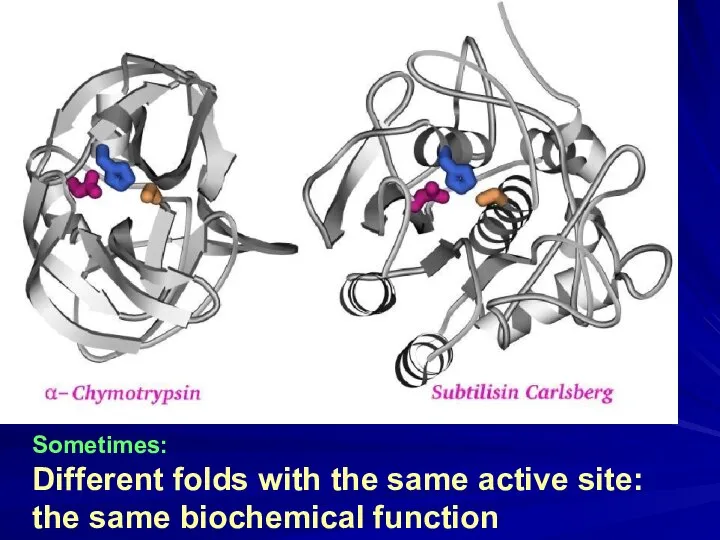
- 16. Sometimes: Different folds with the same active site: the same biochemical function
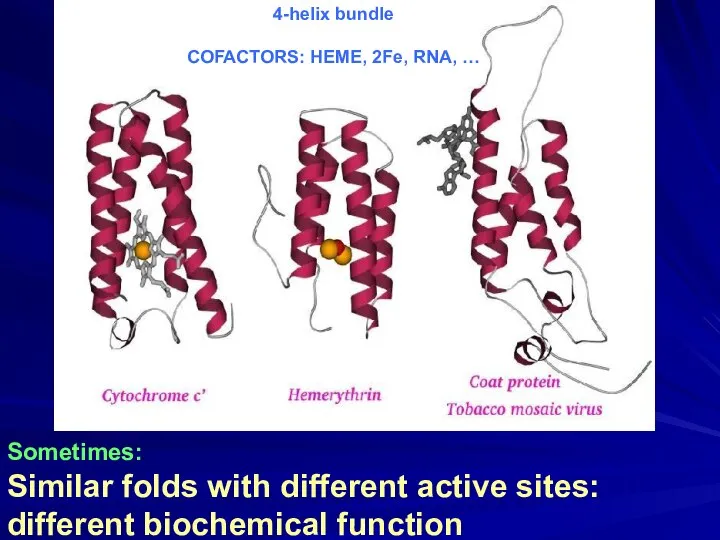
- 17. Sometimes: Similar folds with different active sites: different biochemical function 4-helix bundle COFACTORS: HEME, 2Fe, RNA,
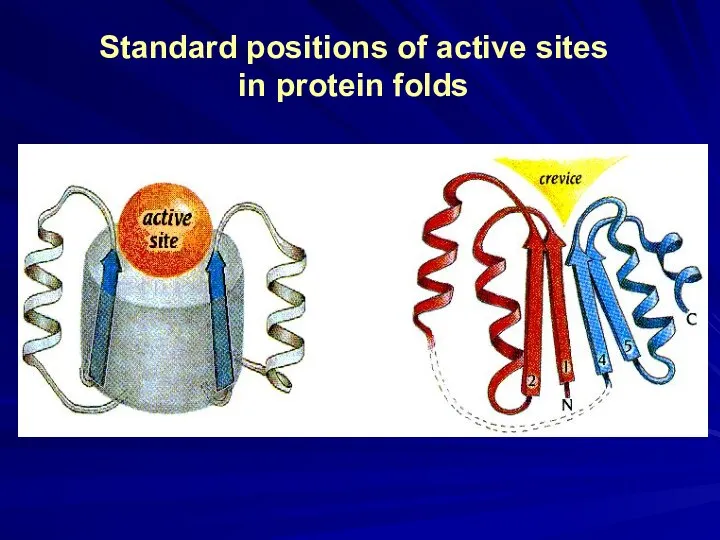
- 18. Standard positions of active sites in protein folds
- 19. Natively disordered protein: X-ray + SAXS + NMR + MD simulations
- 20. Chaperone GroEL
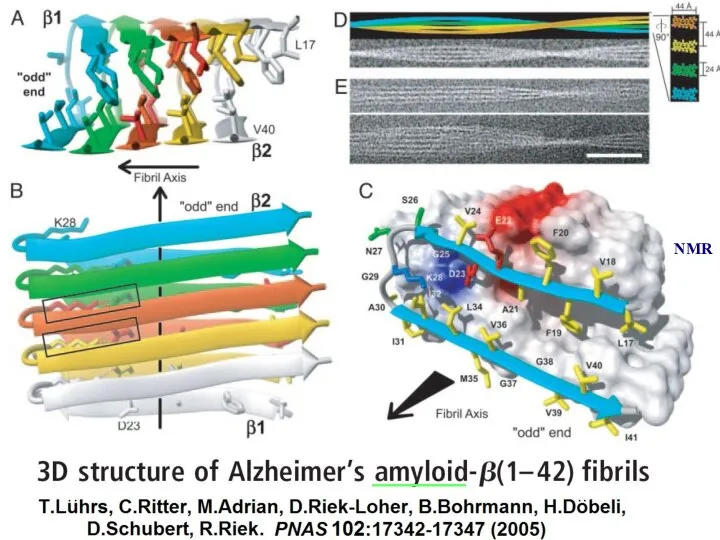
- 21. ______ NMR
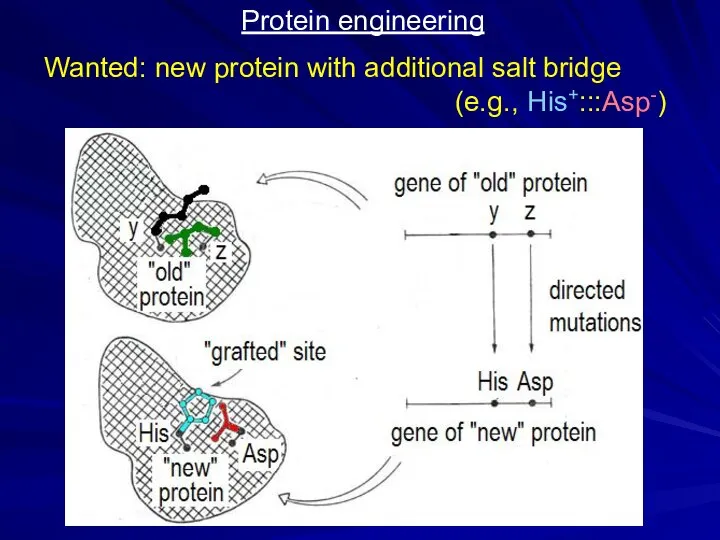
- 22. Protein engineering Wanted: new protein with additional salt bridge (e.g., His+:::Asp-)
- 23. PROTEIN PHYSICS LECTURE 2 Elementary interactions: covalent
- 24. Protein chain: regular backbone & gene-encoded sequence of side chains
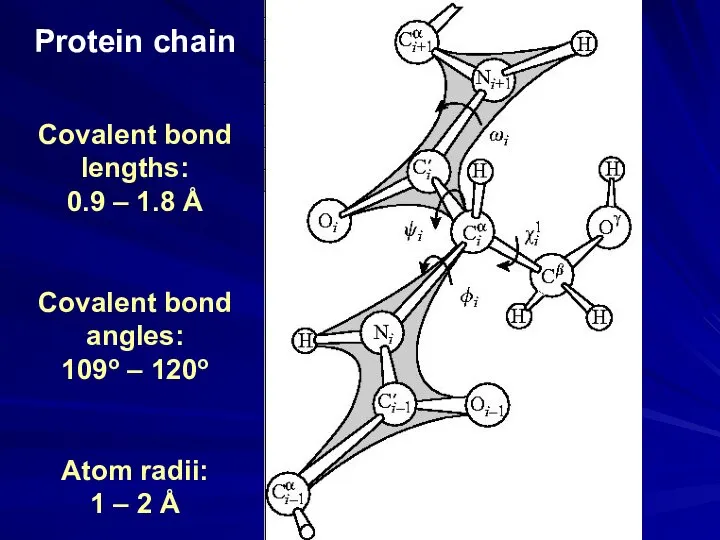
- 25. Protein chain Covalent bond lengths: 0.9 – 1.8 Å Covalent bond angles: 109o – 120o Atom
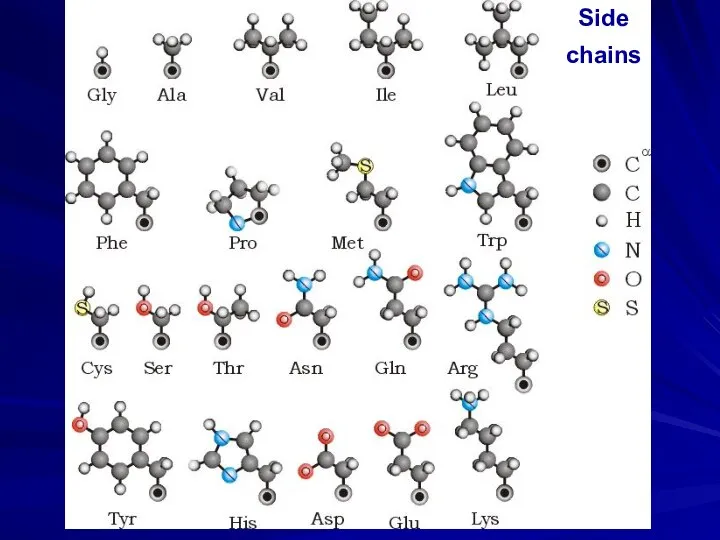
- 26. Side chains
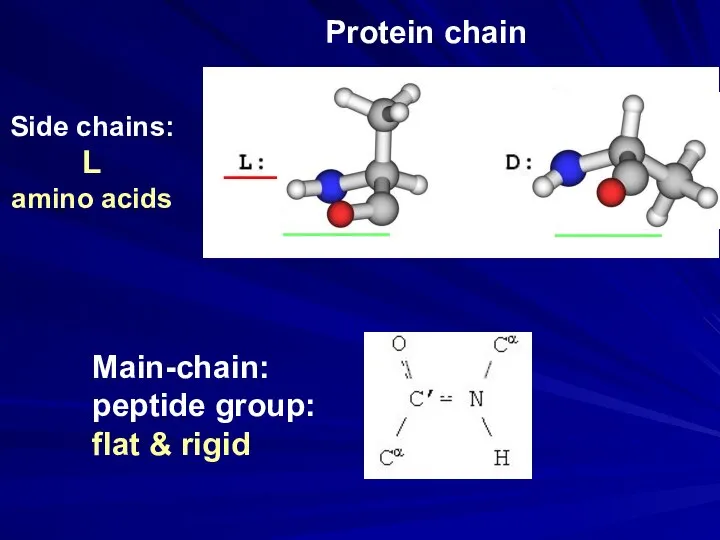
- 27. Main-chain: peptide group: flat & rigid Side chains: L amino acids ___ ______ ______ Protein chain
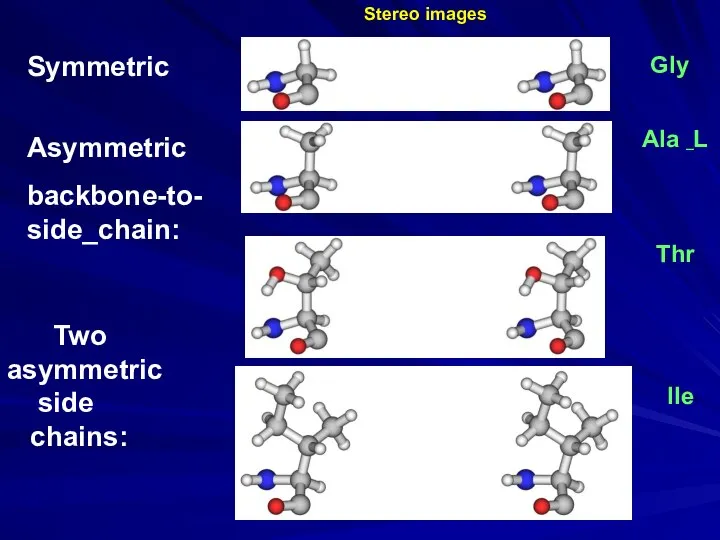
- 28. Ala _L Gly Thr Ile Two asymmetric side chains: Symmetric Asymmetric backbone-to- side_chain: Stereo images
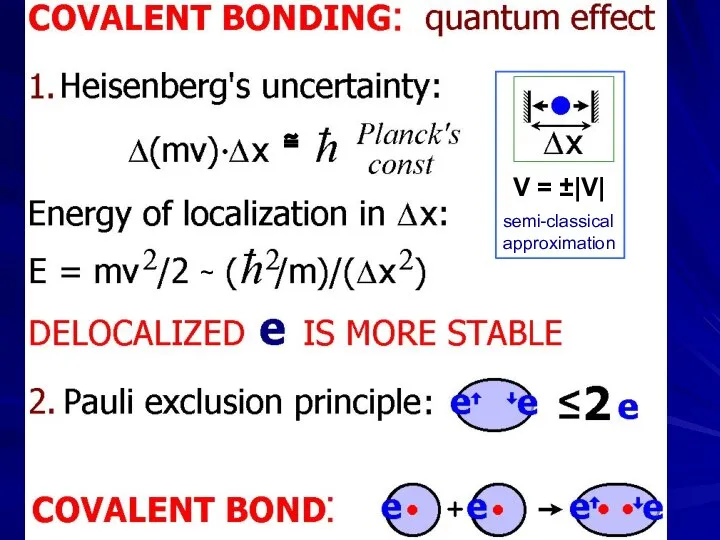
- 29. ~ V = ±|V| ≅ semi-classical approximation
- 30. Werner Karl Heisenberg (1901-76) — Nobel Prize 1932 Wolfgang Ernst Pauli ) (1900-58) — Nobel Prize
- 31. Peptide group: flat & rigid sp2 + p sp2 + p Covalent bonding in peptide group:
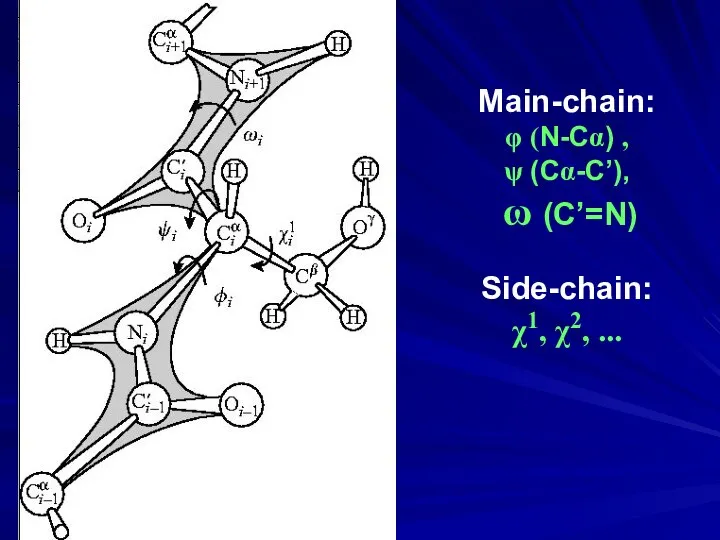
- 32. Main-chain: φ (N-Cα) , ψ (Cα-C’), ω (C’=N) Side-chain: χ1, χ2, ...
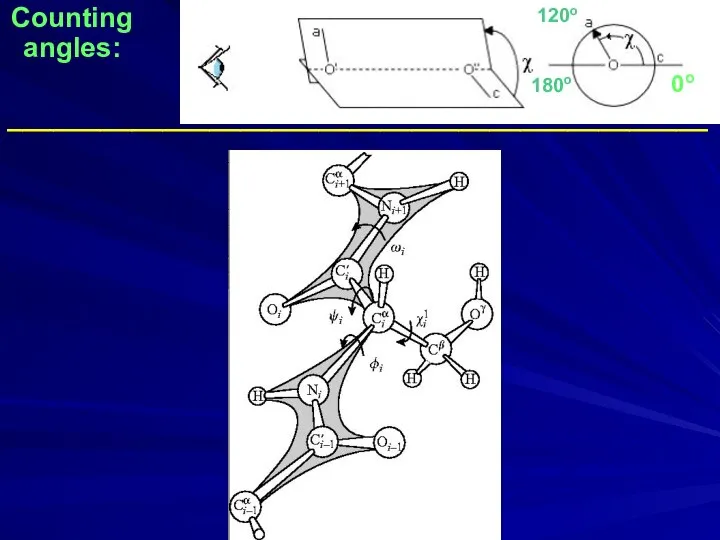
- 33. Counting angles: _____________________________________________ 0o 180o 120o
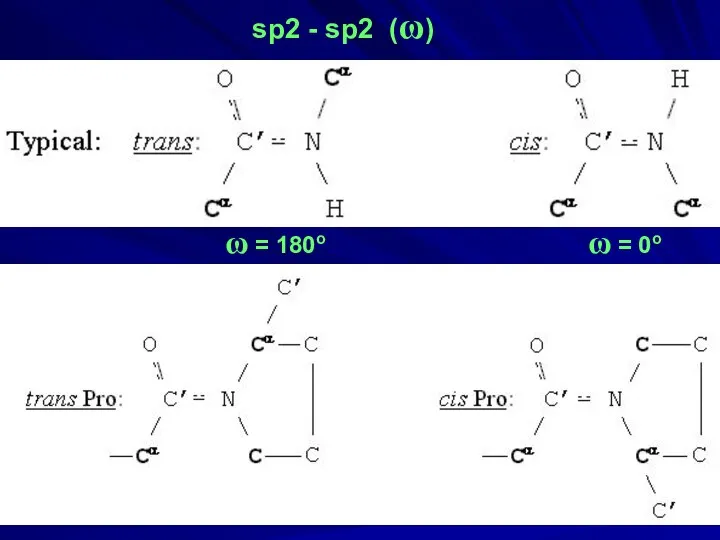
- 34. sp2 - sp2 (ω) ω = 180o ω = 0o
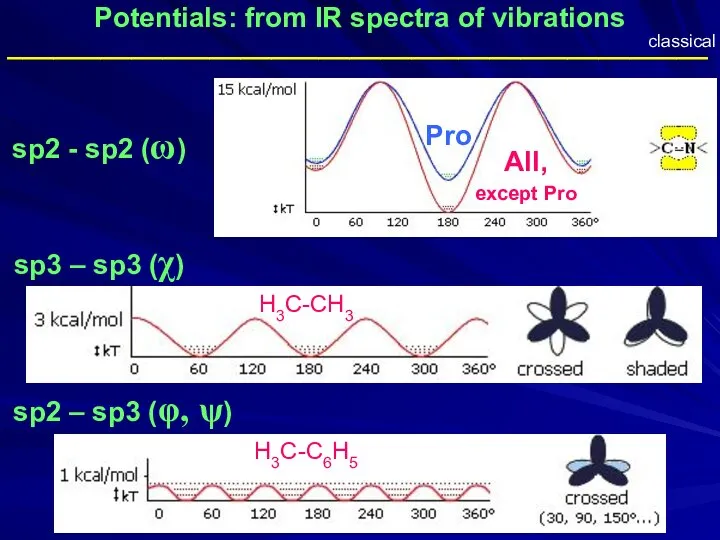
- 35. Potentials: from IR spectra of vibrations sp2 - sp2 (ω) sp3 – sp3 (χ) sp2 –
- 36. Harold Abraham Scheraga (1921) Paul John Flory (1910-85) — Nobel Prize 1974 Александр Исаакович Китайгородский (1914–1985)
- 38. Скачать презентацию














 Конвективный теплообмен
Конвективный теплообмен Презентация по физике "Производство, передача и потребление электроэнергии" - скачать
Презентация по физике "Производство, передача и потребление электроэнергии" - скачать  Воздух, какой он
Воздух, какой он Посвящение в юные физики
Посвящение в юные физики Воздействие лазерного излучения на мишени
Воздействие лазерного излучения на мишени Составитель: Гринякин Станислав Александрович Руководитель: Талалай Ольга Георгиевна, учитель физики
Составитель: Гринякин Станислав Александрович Руководитель: Талалай Ольга Георгиевна, учитель физики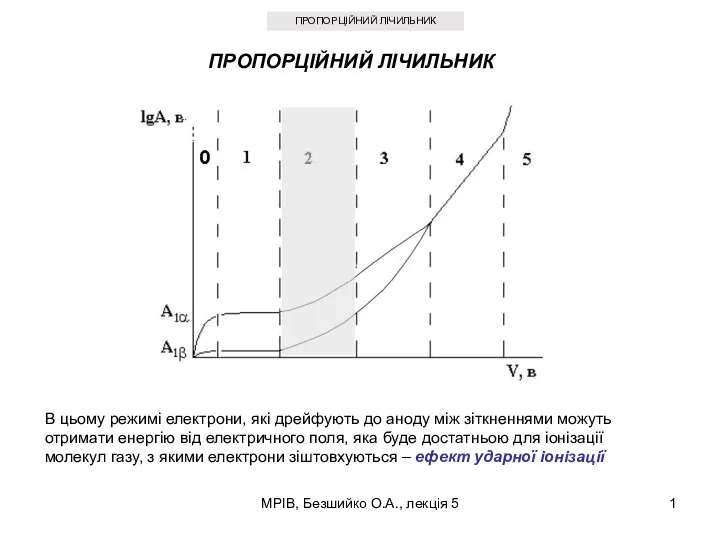 Пропорційний лічильник
Пропорційний лічильник Медициналық интроскопияның негізгі техникалық құралдары. Фотоэлектрлік түрлендіргіштер
Медициналық интроскопияның негізгі техникалық құралдары. Фотоэлектрлік түрлендіргіштер Обзор доказательств ложности утверждений о возможности самосовершенствования
Обзор доказательств ложности утверждений о возможности самосовершенствования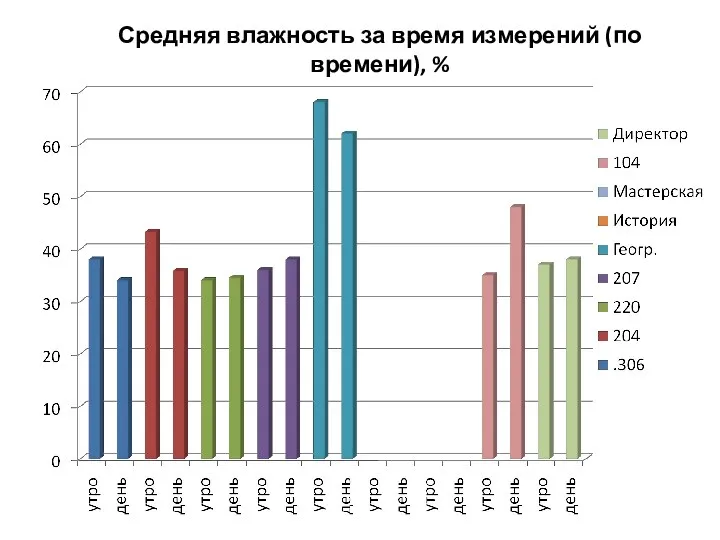 Таблицы. Средняя влажность за время измерений (по времени), %
Таблицы. Средняя влажность за время измерений (по времени), % Плоское движение капсулы в атмосфере
Плоское движение капсулы в атмосфере Гипотеза де Бройля. Дифракция электронов. Микрочастица в двухщелевом интерферометре. Соотношение неопределенностей Гейзенберга
Гипотеза де Бройля. Дифракция электронов. Микрочастица в двухщелевом интерферометре. Соотношение неопределенностей Гейзенберга Электрический ток в газах
Электрический ток в газах Сложение сил. Равнодействующая сил. Физика 7 класс
Сложение сил. Равнодействующая сил. Физика 7 класс Приведение системы сил к данному центру
Приведение системы сил к данному центру История русской философии Западники
История русской философии Западники Проект: Вычисление реагентов расходуемых за год. год.
Проект: Вычисление реагентов расходуемых за год. год.  Прямолинейные колебания точки
Прямолинейные колебания точки Общие указания к построению эпюр поперечных сил и изгибающих моментов
Общие указания к построению эпюр поперечных сил и изгибающих моментов Как можно избежать действия электрического тока при случайном прикосновении к электроприбору, которое оказалось под напряжением?
Как можно избежать действия электрического тока при случайном прикосновении к электроприбору, которое оказалось под напряжением? Исследование гиратора на основе слоистой структуры феррит никеля–метглас–цирконат-титанат свинца
Исследование гиратора на основе слоистой структуры феррит никеля–метглас–цирконат-титанат свинца Сила упругости. Закон Гука
Сила упругости. Закон Гука Тректі мембраналарды алу: иондардың зат арқылы жүруі
Тректі мембраналарды алу: иондардың зат арқылы жүруі Модели атомов. Опыт Резерфорда. Презентация по физике
Модели атомов. Опыт Резерфорда. Презентация по физике История развития телевидения
История развития телевидения Презентация Тепловые электростанции
Презентация Тепловые электростанции  Ортогональное частотное разделение со многими поднесущими (OFDM)
Ортогональное частотное разделение со многими поднесущими (OFDM) Структурный анализ и синтез механизмов
Структурный анализ и синтез механизмов