Содержание
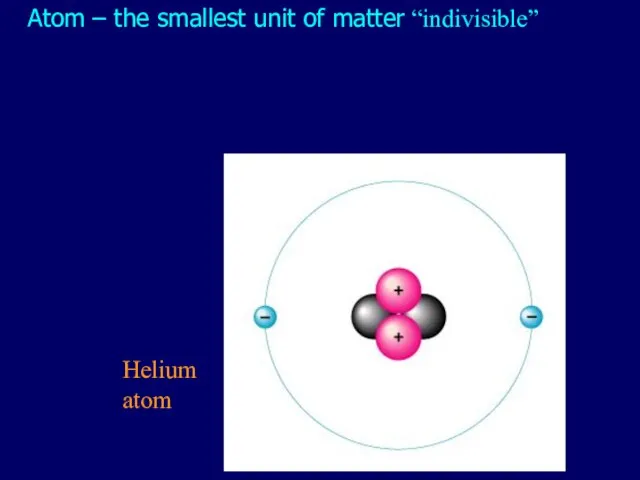
- 2. Atom – the smallest unit of matter “indivisible” Helium atom
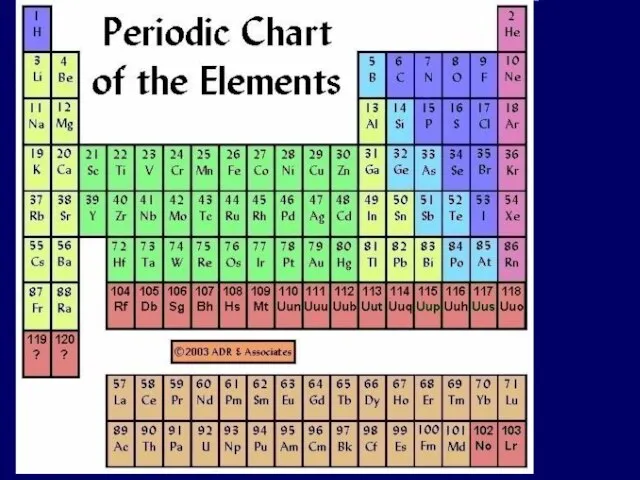
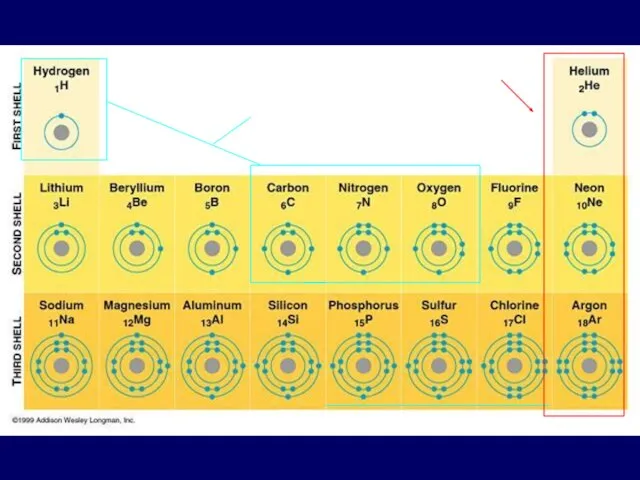
- 3. electron shells Atomic number = number of Electrons Electrons vary in the amount of energy they
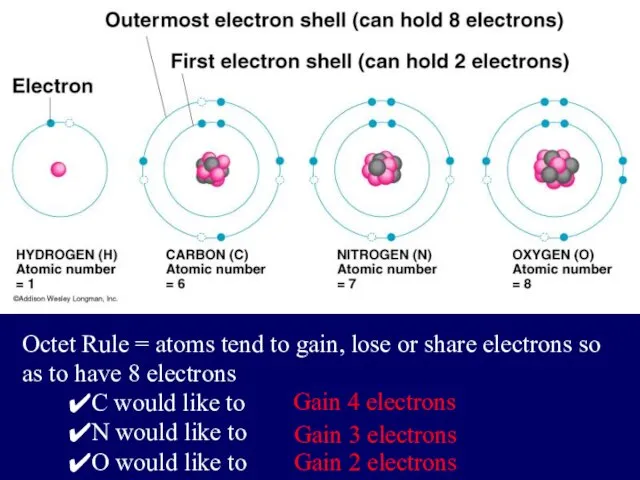
- 4. Electrons are placed in shells according to rules: The 1st shell can hold up to two
- 5. Octet Rule = atoms tend to gain, lose or share electrons so as to have 8
- 6. Why are electrons important? Elements have different electron configurations different electron configurations mean different levels of
- 9. Electron Dot Structures Symbols of atoms with dots to represent the valence-shell electrons 1 2 13
- 10. Chemical bonds: an attempt to fill electron shells Ionic bonds – Covalent bonds – Metallic bonds
- 11. Learning Check ● A. X would be the electron dot formula for 1) Na 2) K
- 12. IONIC BOND bond formed between two ions by the transfer of electrons
- 13. Formation of Ions from Metals Ionic compounds result when metals react with nonmetals Metals lose electrons
- 14. Formation of Sodium Ion Sodium atom Sodium ion Na ∙ – e− ⎯⎯→ Na + 2-8-1
- 15. Formation of Magnesium Ion Magnesium atom Magnesium ion ∙ Mg ∙ – 2e− ⎯→ Mg2+ 2-8-2
- 16. Some Typical Ions with Positive Charges (Cations) Group 1 Group 2 Group 13 H+ Mg2+ Al3+
- 17. Learning Check A. Number of valence electrons in aluminum 1) 1 e- 2) 2 e- 3)
- 18. Solution A. Number of valence electrons in aluminum 3) 3 e- B. Change in electrons for
- 19. Learning Check Give the ionic charge for each of the following: A. 12 p+ and 10
- 20. Ions from Nonmetal Ions In ionic compounds, nonmetals in 15, 16, and 17 gain electrons from
- 21. Fluoride Ion unpaired electron octet ∙ ∙ ∙ ∙ 1 - : F ∙ + e−
- 22. Ionic Bond Between atoms of metals and nonmetals with very different electronegativity Bond formed by transfer
- 24. Ionic Bonds: One Big Greedy Thief Dog!
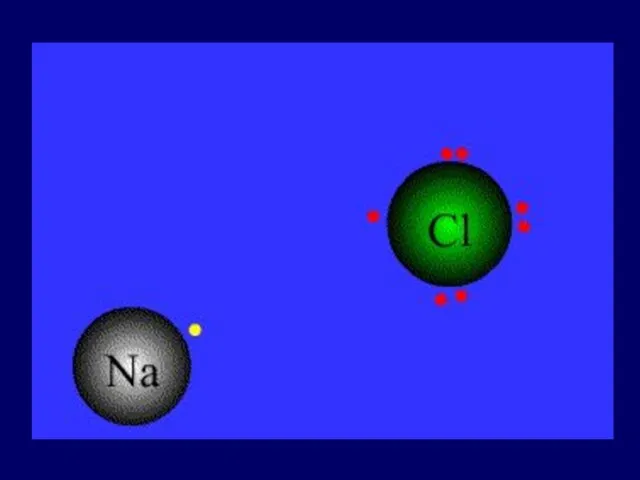
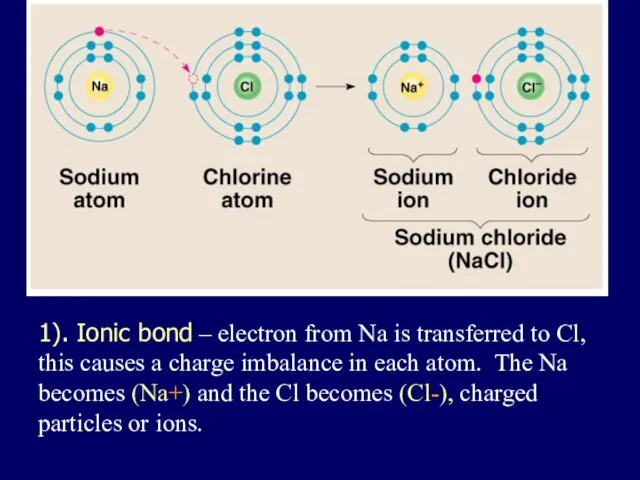
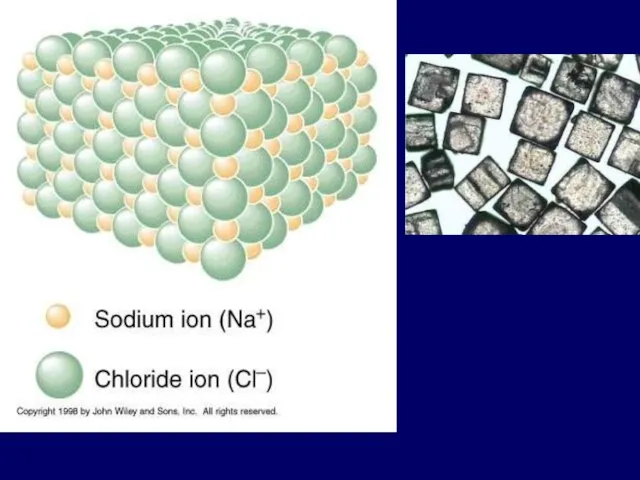
- 25. 1). Ionic bond – electron from Na is transferred to Cl, this causes a charge imbalance
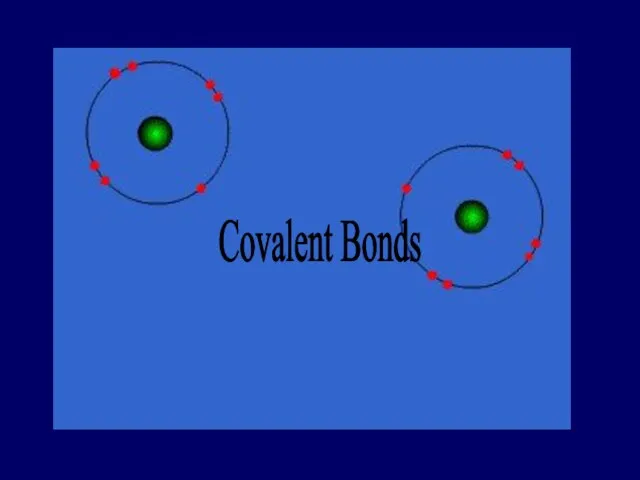
- 27. COVALENT BOND bond formed by the sharing of electrons
- 28. Covalent Bond Between nonmetallic elements of similar electronegativity. Formed by sharing electron pairs Stable non-ionizing particles,
- 29. Covalent Bonds
- 30. Bonds in all the polyatomic ions and diatomics are all covalent bonds
- 31. when electrons are shared equally NONPOLAR COVALENT BONDS H2 or Cl2
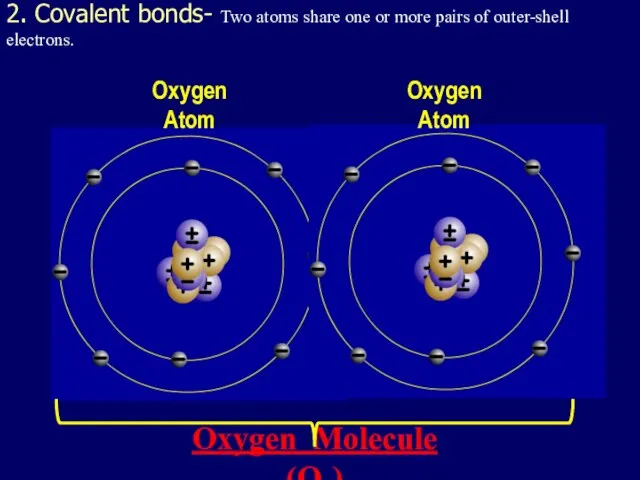
- 32. 2. Covalent bonds- Two atoms share one or more pairs of outer-shell electrons. Oxygen Atom Oxygen
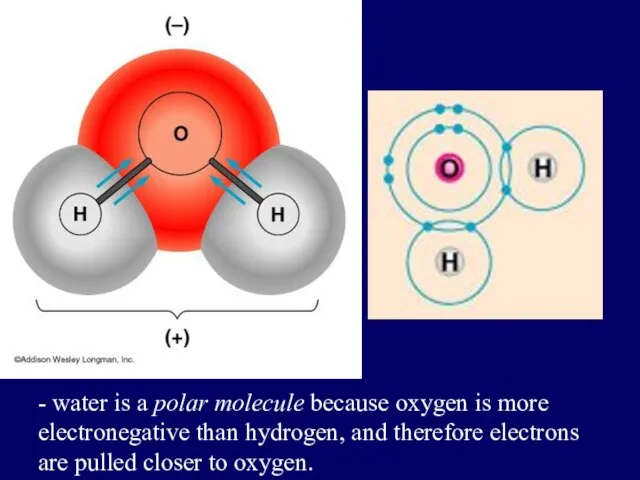
- 33. when electrons are shared but shared unequally POLAR COVALENT BONDS H2O
- 34. Polar Covalent Bonds: Unevenly matched, but willing to share.
- 35. - water is a polar molecule because oxygen is more electronegative than hydrogen, and therefore electrons
- 36. METALLIC BOND bond found in metals; holds metal atoms together very strongly
- 37. Metallic Bond Formed between atoms of metallic elements Electron cloud around atoms Good conductors at all
- 38. Metallic Bonds: Mellow dogs with plenty of bones to go around.
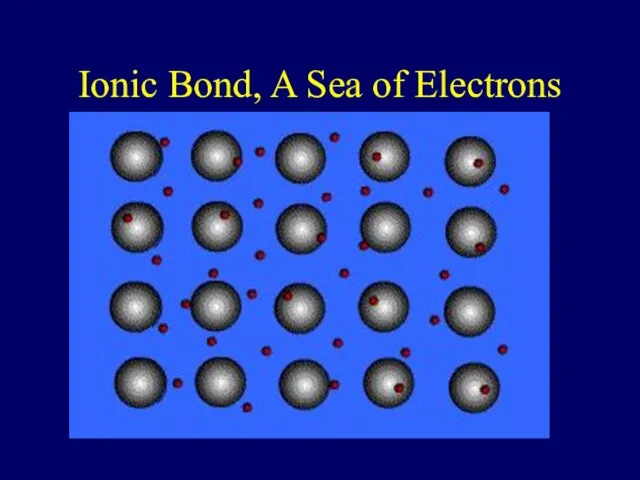
- 39. Ionic Bond, A Sea of Electrons
- 40. Metals Form Alloys Metals do not combine with metals. They form Alloys which is a solution
- 41. Formula Weights Formula weight is the sum of the atomic masses. Example- CO2 Mass, C +
- 42. Practice Compute the mass of the following compounds round to nearest tenth & state type of
- 45. Скачать презентацию































 Распределение Бозе-Эйнштейна. Конденсация Бозе-газа. Температура конденсации. Число частиц в конденсате
Распределение Бозе-Эйнштейна. Конденсация Бозе-газа. Температура конденсации. Число частиц в конденсате Презентация по химии Углеводороды
Презентация по химии Углеводороды  Сера и её соединения
Сера и её соединения Коррозия металлов
Коррозия металлов Жидкостная хроматография
Жидкостная хроматография Оксиди. Кислоти
Оксиди. Кислоти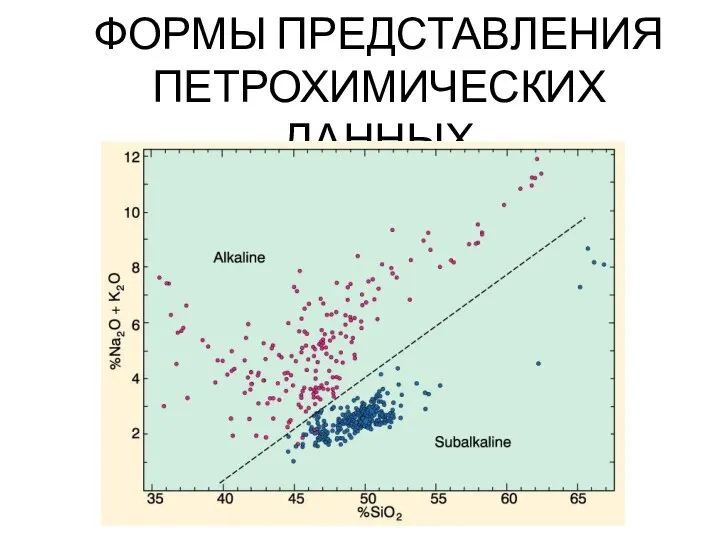 Формы представления петрохимических данных
Формы представления петрохимических данных Строение атома (8 класс)
Строение атома (8 класс) Презентация по Химии "СИММЕТРИЯ В ХИМИИ" - скачать смотреть
Презентация по Химии "СИММЕТРИЯ В ХИМИИ" - скачать смотреть  Презентация по Химии "Увлекательные факты из жизни великого химика." - скачать смотреть бесплатно
Презентация по Химии "Увлекательные факты из жизни великого химика." - скачать смотреть бесплатно Решение задач Скорость химических реакций 11 класс
Решение задач Скорость химических реакций 11 класс  ХИМИЧЕСКИЕ СВОЙСТВА АЛКАНОВ Презентацию составила учитель МОУ «Ягринская гимназия» г.Северодвинска Шапошникова Т.С.
ХИМИЧЕСКИЕ СВОЙСТВА АЛКАНОВ Презентацию составила учитель МОУ «Ягринская гимназия» г.Северодвинска Шапошникова Т.С.  Солеобразующие оксиды
Солеобразующие оксиды Геологические условия формирования месторождений полезных ископаемых. (Лекция 3)
Геологические условия формирования месторождений полезных ископаемых. (Лекция 3) Формы парфюмерно-косметической продукции
Формы парфюмерно-косметической продукции Гетерофункциональные производные бензольного ряда как лекарственные средства. Гетероциклические соединения. Алкалоиды
Гетерофункциональные производные бензольного ряда как лекарственные средства. Гетероциклические соединения. Алкалоиды Алкани. Ізомерія та номенклатура алканів
Алкани. Ізомерія та номенклатура алканів Презентация по Химии "Подгруппа углерода." - скачать смотреть
Презентация по Химии "Подгруппа углерода." - скачать смотреть  Химическая взрывчатка
Химическая взрывчатка Основы кристаллического строения металлов
Основы кристаллического строения металлов Виртуозы химического эксперимента
Виртуозы химического эксперимента Физические свойства спиртов
Физические свойства спиртов Валетные состояния атома углерода
Валетные состояния атома углерода Презентация на тему: Одноатомные спирты
Презентация на тему: Одноатомные спирты Основания, их состав, номенклатура и основные представители
Основания, их состав, номенклатура и основные представители Элементердің құрамына қарай бөлінуі
Элементердің құрамына қарай бөлінуі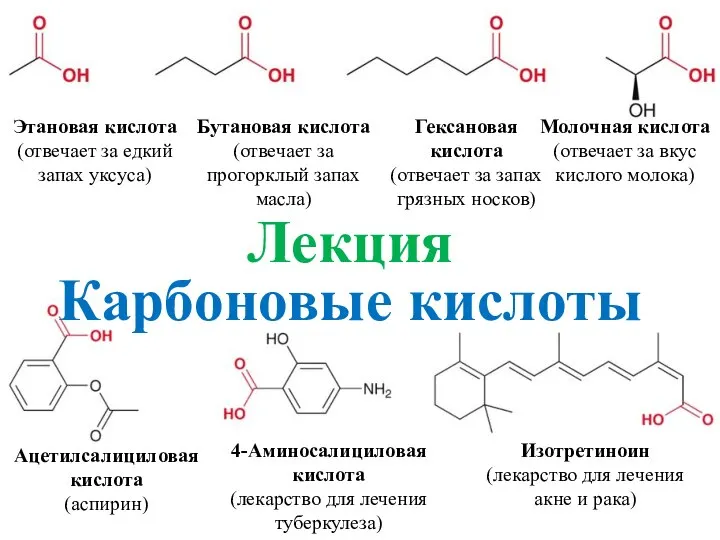 Этановая кислота Бутановая кислота
Этановая кислота Бутановая кислота Углеводы - 2
Углеводы - 2