Содержание
- 3. Silicon is a chemical element with symbol Si and atomic number 14. A hard and brittle
- 4. Production Ferrosilicon, an iron-silicon alloy that contains varying ratios of elemental silicon and iron, accounts for
- 5. Health effects of silicon Silicon concentrates in no particular organ of the body but is found
- 6. Silicate minerals are rock-forming minerals made up of silicate groups. They are the largest and most
- 7. Silicate minerals Quartz SiO2 Quartz is one of the most common mineral in Earth’s crust! Silica
- 8. Weathering Weathering is the breaking down of rocks, soil and minerals as well as wood and
- 9. Rocks gradually wear away. This is called weathering. There are three types of weathering: 1.physical weathering
- 13. Скачать презентацию
Silicon is a chemical element with symbol Si and atomic number 14. A hard and brittle crystalline solid
Silicon is a chemical element with symbol Si and atomic number 14. A hard and brittle crystalline solid
Production
Ferrosilicon, an iron-silicon alloy that contains varying ratios of elemental silicon
Production
Ferrosilicon, an iron-silicon alloy that contains varying ratios of elemental silicon
Health effects of silicon
Silicon concentrates in no particular organ of
Health effects of silicon
Silicon concentrates in no particular organ of
Silicate minerals are rock-forming minerals made up of silicate groups. They are the largest and most
Silicate minerals are rock-forming minerals made up of silicate groups. They are the largest and most
Silicate minerals
Quartz SiO2
Quartz is one of the most common mineral in
Silicate minerals
Quartz SiO2
Quartz is one of the most common mineral in
Silica (Si) and Oxygen (O) are the only elements within pure quartz. If a cooling magma has silica leftover after feldsparsform, quartz is likely to form.
Quartz can be found in all sorts of rocks. Igneous rocks sometimes contain large quartz crystals. Metamorphic rocks such as gneiss also have large quartz crystals. Sedimentary rocks such as sandstone are often made of tons of little pieces of quartz crystals. In fact, most sand is made of quartz because it is hard and does not weather away easily. Some pieces of quartz are white like milk but most are clear like glass, sometimes with a little pink or gray tinge of color.
Olivine (Mg, Fe)2SiO4
Olivine looks like little green crystals. It is typically found in some igneous and metamorphic rocks. Often the crystals are so small that you need to use your hand lens or magnifying glass to see them clearly.
Shape: Orthorhombic (usually a many-sided prism that has an overall sphere shape)
Luster: Greasy
Color: Green (but sometimes yellow or brown)
Streak: White
Hardness: 6.5-7 on Mohs Hardness Scale
Fracture: Conchoidal, brittle
Weathering
Weathering is the breaking down of rocks, soil and minerals as well as wood and artificial materials
Weathering
Weathering is the breaking down of rocks, soil and minerals as well as wood and artificial materials
Rocks gradually wear away. This is called weathering. There are three
Rocks gradually wear away. This is called weathering. There are three
1)Physical weathering
Physical weathering is caused by physical changes such as changes in temperature, freezing and thawing, and the effects of wind, rain and waves.
Wind, rain and waves
Wind, rain and waves can all cause weathering. The wind can blow tiny grains of sand against a rock. These wear the rock away and weather it. Rain and waves can also wear away rock over long periods of time.
3)Biological weathering
Animals and plants can wear away rocks. This is called biological weathering. For example, burrowing animals such as rabbits can burrow into a crack in a rock, making it bigger and splitting the rock.
2)Chemical weathering
The weathering of rocks by chemicals is called chemical weathering. Rainwater is naturally slightly acidic because carbon dioxide from the air dissolves in it. Minerals in rocks may react with the rainwater, causing the rock to be weathered.
Acid rain
When fossil fuels such as coal, oil and natural gas are burned, carbon dioxideand sulphur dioxide escape into the air. These dissolve in the water in the clouds and make the rainwater more acidic than normal. When this happens, we call the rain 'acid rain‘.










 Единый государственный экзамен по ХИМИИ (консультация 2) О.П. Костенко, проректор ГОУ РК «ИПК РО» по НМР
Единый государственный экзамен по ХИМИИ (консультация 2) О.П. Костенко, проректор ГОУ РК «ИПК РО» по НМР История развития биохимического знания и биохимии как науки
История развития биохимического знания и биохимии как науки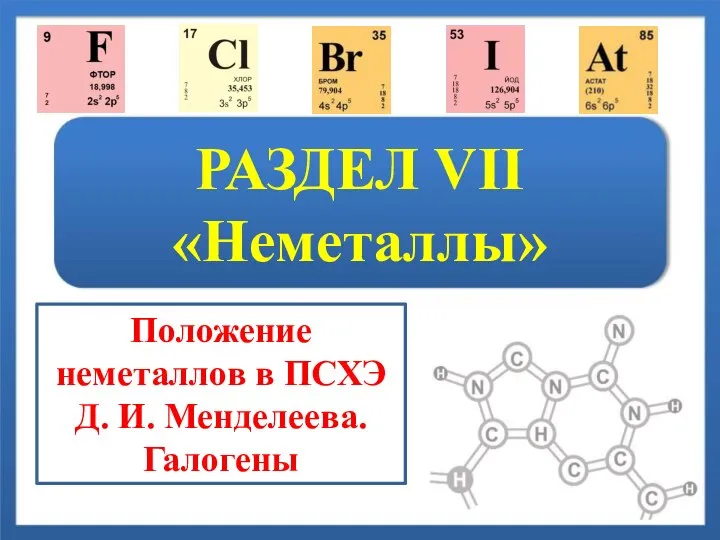 Неметаллы. Положение неметаллов в ПСХЭ Д.И. Менделеева. Галогены
Неметаллы. Положение неметаллов в ПСХЭ Д.И. Менделеева. Галогены Простые и сложные вещества. Основные классы неорганических веществ. Номенклатура соединений
Простые и сложные вещества. Основные классы неорганических веществ. Номенклатура соединений Биоорганическая химия
Биоорганическая химия Как «архангельский мужик … стал разумен и велик». О Михаиле Васильевиче Ломоносове
Как «архангельский мужик … стал разумен и велик». О Михаиле Васильевиче Ломоносове  Строение гетероциклических соединений
Строение гетероциклических соединений МЕТАЛЛЫ диктант 9 класс
МЕТАЛЛЫ диктант 9 класс  Основные породы. Классификация основных пород
Основные породы. Классификация основных пород Общая фармакология
Общая фармакология Хром
Хром Презентация по Химии "Жёсткость воды" - скачать смотреть
Презентация по Химии "Жёсткость воды" - скачать смотреть  Аэробное окисление углеводов
Аэробное окисление углеводов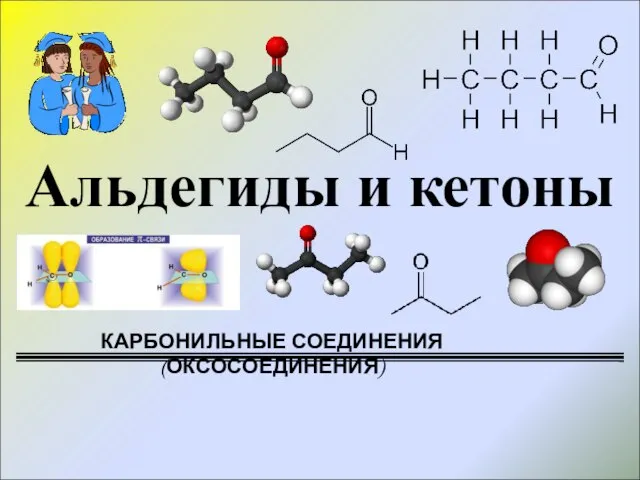 Карбонильные соединения (оксосоединения)
Карбонильные соединения (оксосоединения) История каучука
История каучука Химическая музыка
Химическая музыка Строение атома
Строение атома Скорость химических реакций. Факторы, влияющие на скорость химической реакции
Скорость химических реакций. Факторы, влияющие на скорость химической реакции Синтез нанослоев неорганических веществ методами ионно-коллоидного и коллоидного наслаивания. (Лекция 10)
Синтез нанослоев неорганических веществ методами ионно-коллоидного и коллоидного наслаивания. (Лекция 10) Масс-спектрометрия (МС)
Масс-спектрометрия (МС) Презентация по Химии "Скло Будівельний матеріал" - скачать смотреть бесплатно
Презентация по Химии "Скло Будівельний матеріал" - скачать смотреть бесплатно Распределительная хроматография
Распределительная хроматография Поверхностные явления. Адсорбция. (Лекция 1)
Поверхностные явления. Адсорбция. (Лекция 1) Свойства солёной воды. Море у меня в стакане
Свойства солёной воды. Море у меня в стакане Ауа. Ауаның құрамы №1 көрсетілім «Ауа мен оттегіде жану реакциясы»
Ауа. Ауаның құрамы №1 көрсетілім «Ауа мен оттегіде жану реакциясы» Круговорот химических веществ в природе Выполнила: Подсыпанина Наташа Ученица: 10 «А» класса Учитель: Макарова Е. И.
Круговорот химических веществ в природе Выполнила: Подсыпанина Наташа Ученица: 10 «А» класса Учитель: Макарова Е. И. А. Общая характеристика подгруппы углерода: Подгруппа углерода - главная подгруппа IV группы, которую составляют углерод-С. к
А. Общая характеристика подгруппы углерода: Подгруппа углерода - главная подгруппа IV группы, которую составляют углерод-С. к Introduction to metabolism
Introduction to metabolism