Содержание
- 2. Introduction to Metabolism
- 3. Metabolism is the sum of an organism’s chemical reactions Metabolism is an emergent property of life
- 4. A metabolic pathway begins with a specific molecule and ends with a product The product of
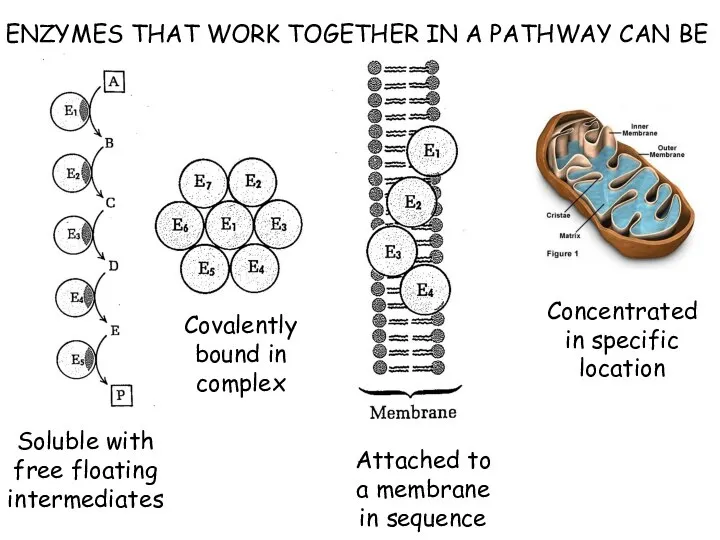
- 5. ENZYMES THAT WORK TOGETHER IN A PATHWAY CAN BE Soluble with free floating intermediates Covalently bound
- 6. CATABOLIC PATHWAY (CATABOLISM) Release of energy by the breakdown of complex molecules to simpler compounds EX:
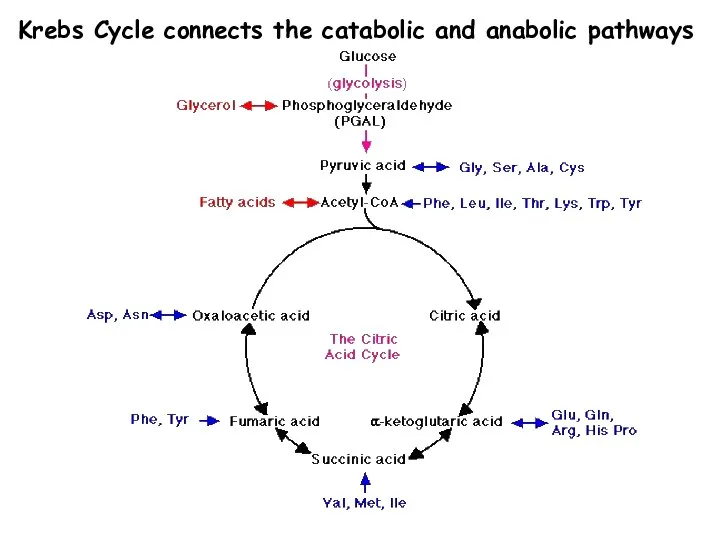
- 7. Krebs Cycle connects the catabolic and anabolic pathways
- 8. Forms of Energy ENERGY = capacity to cause change Energy exists in various forms (some of
- 9. KINETIC ENERGY – energy associated with motion HEAT (thermal energy) is kinetic energy associated with random
- 10. On the platform, the diver has more potential energy. Diving converts potential energy to kinetic energy.
- 11. THERMODYNAMICS = the study of energy transformations CLOSED system (EX: liquid in a thermos) = isolated
- 12. The First Law of Thermodynamics = energy of the universe is constant Energy can be transferred
- 13. The Second Law of Thermodynamics During every energy transfer or transformation entropy (disorder) of the universe
- 14. Chemical energy Heat CO2 First law of thermodynamics Second law of thermodynamics H2O ORGANISMS are energy
- 15. Free-Energy Change (ΔG) can help tell which reactions will happen ∆G = change in free energy
- 16. Exergonic and Endergonic Reactions in Metabolism EXERGONIC reactions (- ∆G) Release energy are spontaneous ENDERGONIC reactions
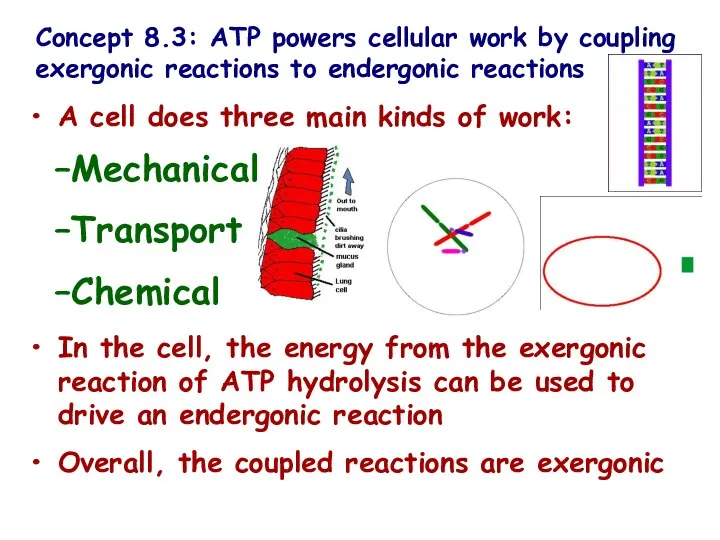
- 17. Concept 8.3: ATP powers cellular work by coupling exergonic reactions to endergonic reactions A cell does
- 18. Phosphate groups Ribose Adenine ATP (adenosine triphosphate) is the cell’s renewable and reusable energy shuttle ATP
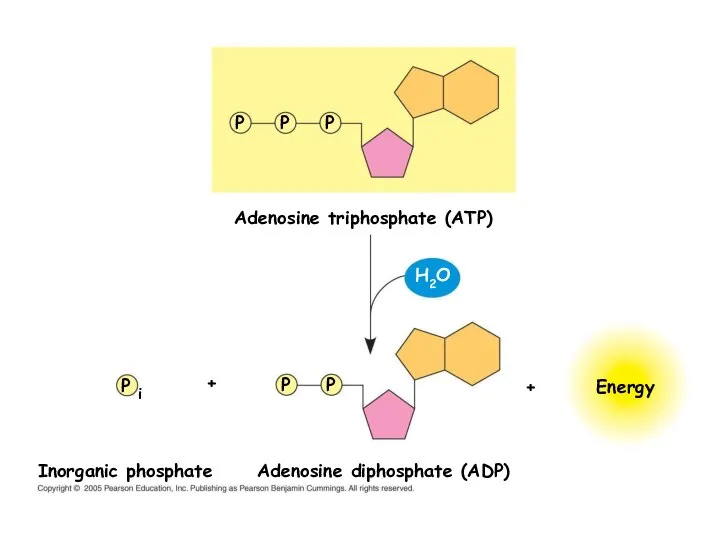
- 19. Adenosine triphosphate (ATP) Energy P P P P P P i Adenosine diphosphate (ADP) Inorganic phosphate
- 20. P i ADP Energy for cellular work provided by the loss of phosphate from ATP Energy
- 21. Endergonic reaction: DG is positive, reaction is not spontaneous Exergonic reaction: DG is negative, reaction is
- 23. Скачать презентацию
















 Кинетика химических реакций. (Лекция 12)
Кинетика химических реакций. (Лекция 12) Углеводороды. Природный газ. Алканы
Углеводороды. Природный газ. Алканы Простые и сложные вещества. Основные классы неорганических веществ. Номенклатура соединений
Простые и сложные вещества. Основные классы неорганических веществ. Номенклатура соединений Исследование химического состава шоколада и способов его приготовления
Исследование химического состава шоколада и способов его приготовления Природные источники углеводородов
Природные источники углеводородов Практическое занятие № 1. Техника безопасности при работе в химической лаборатории. Описание веществ
Практическое занятие № 1. Техника безопасности при работе в химической лаборатории. Описание веществ Химические свойства карбокатионов
Химические свойства карбокатионов Аттестационная работа. Многогранники: от молекулы до кристалла
Аттестационная работа. Многогранники: от молекулы до кристалла Химия переходных элементов
Химия переходных элементов ОКСИДИ – це бінарні сполуки , що складаються з двох елементів , одним з яких є Оксиген . Загальна формула оксидів : E2On n – вале
ОКСИДИ – це бінарні сполуки , що складаються з двох елементів , одним з яких є Оксиген . Загальна формула оксидів : E2On n – вале Анализ смеси катионов первойтретьей аналитических групп по систематическому анализу. Химическое равновесие
Анализ смеси катионов первойтретьей аналитических групп по систематическому анализу. Химическое равновесие ВКР: Организация проведения товароведной экспертизы пищевкусовых товаров
ВКР: Организация проведения товароведной экспертизы пищевкусовых товаров Презентация по Химии "Азотная кислота" - скачать смотреть
Презентация по Химии "Азотная кислота" - скачать смотреть  Історія виникнення мила Розмаїтого Дмитра, 9-Б клас
Історія виникнення мила Розмаїтого Дмитра, 9-Б клас  Электрохимические методы анализа. Лекция 3
Электрохимические методы анализа. Лекция 3 Тривиальные названия неорганических соединений
Тривиальные названия неорганических соединений Структура аминокислот
Структура аминокислот Дифференцированный подход в обучении химии
Дифференцированный подход в обучении химии Физико-химические методы анализа
Физико-химические методы анализа Кинетические методы анализа
Кинетические методы анализа Полипропилен. Молекулалық құрылымы
Полипропилен. Молекулалық құрылымы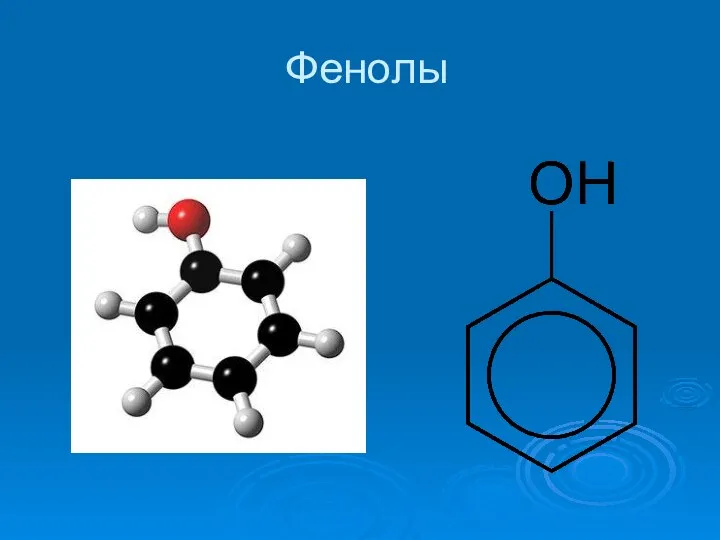 Фенолы. Физические свойства. Получение
Фенолы. Физические свойства. Получение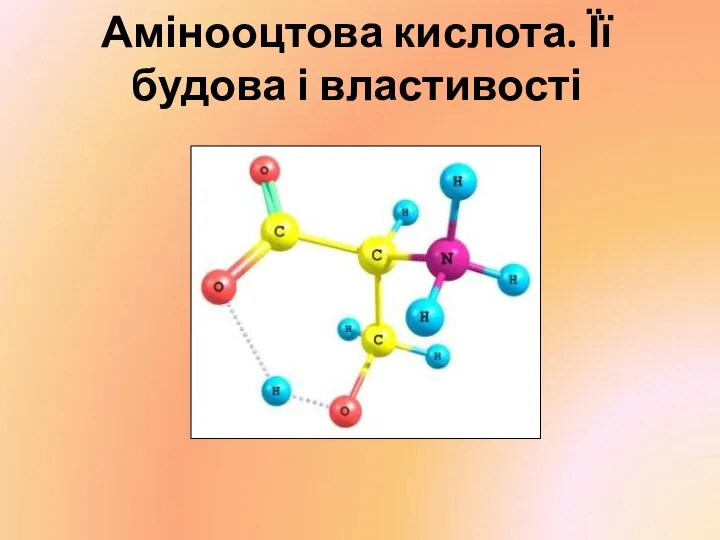 Амінооцтова кислота. Її будова і властивості
Амінооцтова кислота. Її будова і властивості Скорость химических реакций. Факторы, влияющие на скорость химической реакции
Скорость химических реакций. Факторы, влияющие на скорость химической реакции Пестицидтердің қоршаған ортаға зиян-ын тигізбеу жүйесі. Пестицидтерді қолданудың физикалық-химиялық негіздері
Пестицидтердің қоршаған ортаға зиян-ын тигізбеу жүйесі. Пестицидтерді қолданудың физикалық-химиялық негіздері Пища, с точки зрения химика
Пища, с точки зрения химика Изомерия алкенов. Цель урока: закрепить понятие изомеры, познакомить с геометрической изомерией
Изомерия алкенов. Цель урока: закрепить понятие изомеры, познакомить с геометрической изомерией  Классификация минералов по химическому принципу
Классификация минералов по химическому принципу