Содержание
- 2. Lecture 5 MOLECULAR-KINETIC THEORY OF IDEAL GASES THE MOLECULAR BASIS OF THERMAL PHYSICS EVAPORATION AND BOILING
- 3. Main assumptions for Ideal Gas Model The number of molecules in the gas is large, and
- 4. MOLECULAR-KINETIC THEORY OF IDEAL GASES An ideal gas is in a cubical container with sides of
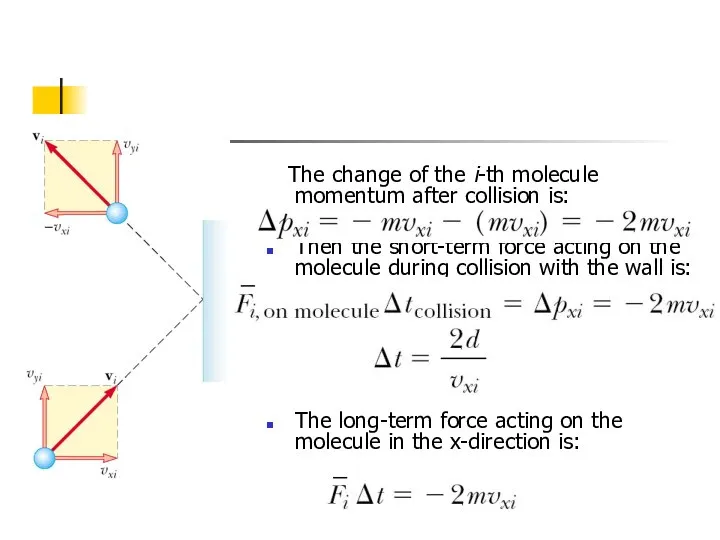
- 5. The change of the i-th molecule momentum after collision is: Then the short-term force acting on
- 6. Using previous expressions we can find x-component of the long-term average force exerted by the wall
- 8. N is the number of molecules This result indicates that the pressure of a gas is
- 9. Molecular interpretation of temperature So we have Experimentally found the equation of state for an ideal
- 10. Theorem of equipartition of energy We can transform the last expression into Generalization of this result
- 11. Root-mean square speed of molecules Using the equation of state for an Ideal gas here n
- 12. Internal Energy In the molecular-kinetic model internal energy of a gas equals the sum of kinetic
- 13. Equation of State for an Ideal Gas Found experimentally: n is the number of moles of
- 14. The Boltzmann Distribution Law We found average kinetic energy of a molecule. But all molecules move
- 15. The Boltzmann Distribution Law Where n0 is defined such that n0dE is the number of molecules
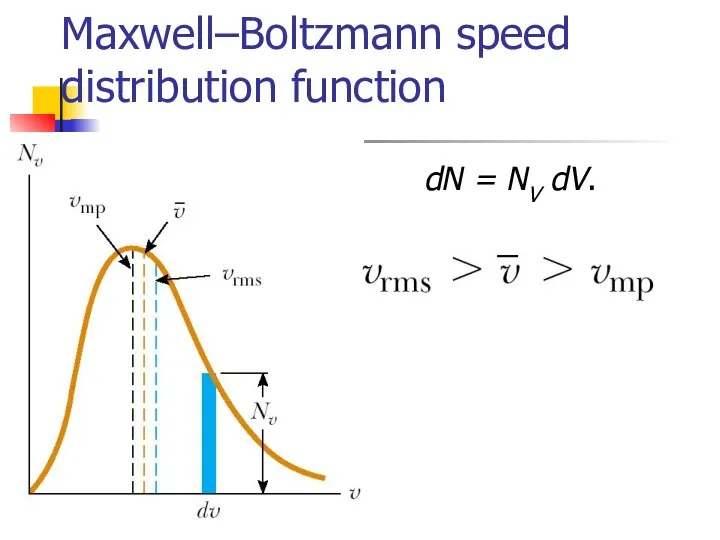
- 16. Maxwell–Boltzmann speed distribution function If N is the total number of molecules, then the number of
- 17. Maxwell–Boltzmann speed distribution function dN = NV dV.
- 18. Gas molecules velocities Root mean square Mean Most probable
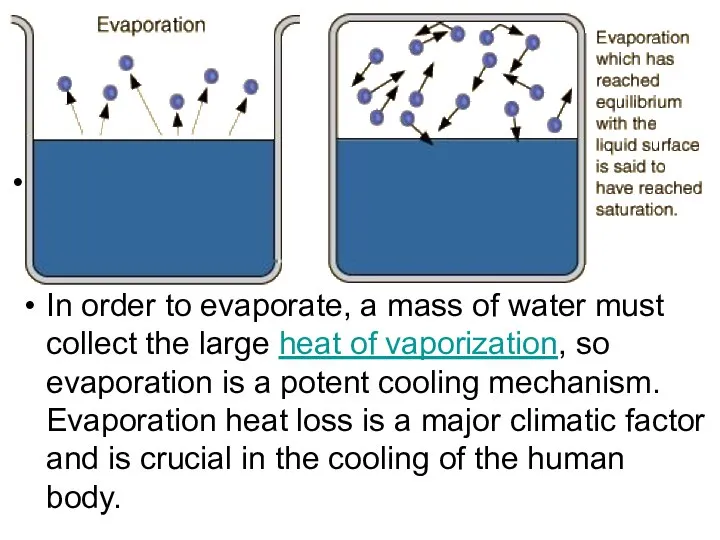
- 19. Evaporation We know that liquids evaporate when they’re below boiling temperature. The speed distribution curve for
- 20. . In order to evaporate, a mass of water must collect the large heat of vaporization,
- 21. Saturation Vapor Pressure Ordinary evaporation is a surface phenomenon - some molecules have enough kinetic energy
- 22. The process of evaporation in a closed container will proceed until there are as many molecules
- 23. Evaporation vs Boiling Ordinary evaporation is a surface phenomenon - since the vapor pressure is low
- 24. Boiling Point The boiling point is defined as the temperature at which the saturated pressure of
- 25. Collisions is the collision cross section. Then the volume it sweeps is . So this molecule
- 26. Then we can find mean collision time Considering movement of the target molecules: N is the
- 27. Mean free path is an average distance between collisions:
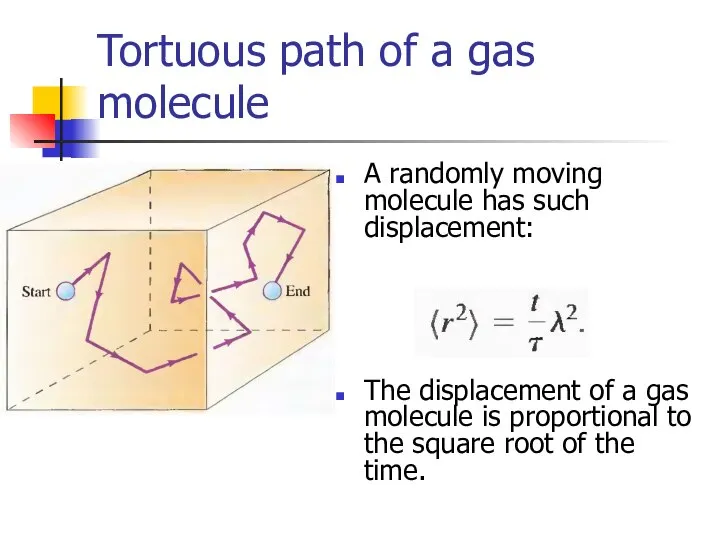
- 28. Tortuous path of a gas molecule A randomly moving molecule has such displacement: The displacement of
- 29. Transport Phenomena By means of collisions that molecules can carry physical properties (momentum, energy, concentration, etc.)
- 30. Some terms The critical temperature of a gas is that temperature above which the gas will
- 31. Van der Waals Gas n is the number of moles of the gas Term represents long-range
- 34. Скачать презентацию



























 Электростатическое поле в веществе
Электростатическое поле в веществе Презентация по физике Давление. Единицы давления
Презентация по физике Давление. Единицы давления Ожэ-электронная спектроскопия
Ожэ-электронная спектроскопия Титрование с применением флюоресцентных индикаторов
Титрование с применением флюоресцентных индикаторов Теоретичские основы теплотехники
Теоретичские основы теплотехники Динамика вращательного движения. (Лекция 5)
Динамика вращательного движения. (Лекция 5) Квантовая и ядерная физика. Раздел Молекулярные спектры
Квантовая и ядерная физика. Раздел Молекулярные спектры Термодинамика. Работа в термодинамике
Термодинамика. Работа в термодинамике Урок № 41. Задачи
Урок № 41. Задачи Презентация Изопроцессы в газах 10 класс
Презентация Изопроцессы в газах 10 класс  Опыты Герца Физика 11 класс
Опыты Герца Физика 11 класс  Равноускоренное прямолинейное движение
Равноускоренное прямолинейное движение Презентация на тему Трансформатор. Передача электрической энергии на расстояние.
Презентация на тему Трансформатор. Передача электрической энергии на расстояние. Поляризация света
Поляризация света Arduino. Электрическая схема
Arduino. Электрическая схема Микроосновы взаимозаменяемости деталей
Микроосновы взаимозаменяемости деталей Давление. Единицы давления. Способы уменьшения и увеличения давления Методическая разработка Васенина Н.Д. учителя физики
Давление. Единицы давления. Способы уменьшения и увеличения давления Методическая разработка Васенина Н.Д. учителя физики  Сравнение энергоисточников
Сравнение энергоисточников Температуралық тепе-теңдік күйіндегі денелердің сәуле шығаруы. Қара дененің сәуле шығару заңдары. (Лекция 14)
Температуралық тепе-теңдік күйіндегі денелердің сәуле шығаруы. Қара дененің сәуле шығару заңдары. (Лекция 14) Аттестационная работа. Физика. Домашняя лаборатория. 7 класс
Аттестационная работа. Физика. Домашняя лаборатория. 7 класс Явление электромагнитной индукции. Магнитный поток. Закон электромагнитной индукции
Явление электромагнитной индукции. Магнитный поток. Закон электромагнитной индукции Презентация ШКАЛА ЭЛЕКТРОМАГНИТНЫХ ИЗЛУЧЕНИЙ
Презентация ШКАЛА ЭЛЕКТРОМАГНИТНЫХ ИЗЛУЧЕНИЙ  Метод поиска решений творческих задач. Метод фокальных объектов
Метод поиска решений творческих задач. Метод фокальных объектов Терагерцевое излучение
Терагерцевое излучение Основные характеристики линий передачи
Основные характеристики линий передачи Механический инструмент для запиловки острых углов, сварных швов
Механический инструмент для запиловки острых углов, сварных швов Уравнение Бернулли Подъемная сила крыла
Уравнение Бернулли Подъемная сила крыла  Викторина по физике Выполнила : Морозова Л. В-учитель физики школы №18, г. Кемерово
Викторина по физике Выполнила : Морозова Л. В-учитель физики школы №18, г. Кемерово