Содержание
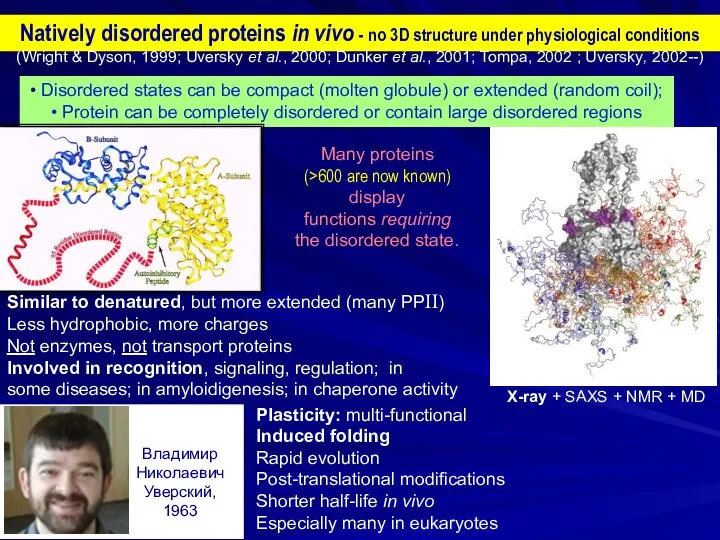
- 2. Natively disordered proteins in vivo - no 3D structure under physiological conditions • Disordered states can
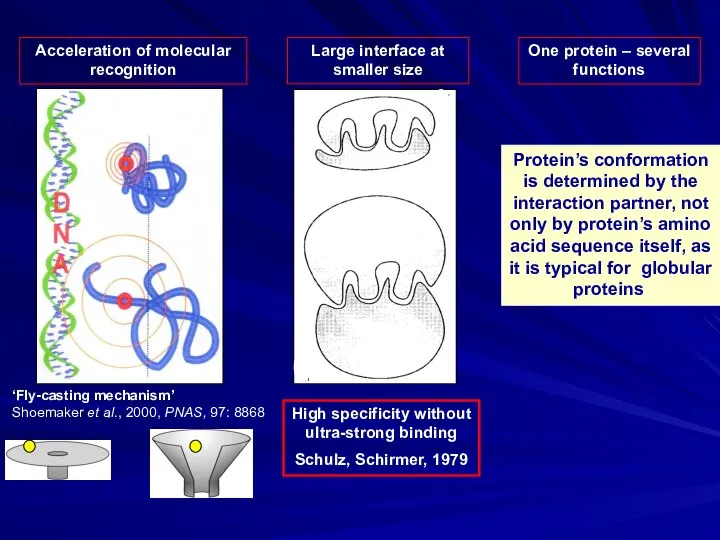
- 3. Acceleration of molecular recognition One protein – several functions Protein’s conformation is determined by the interaction
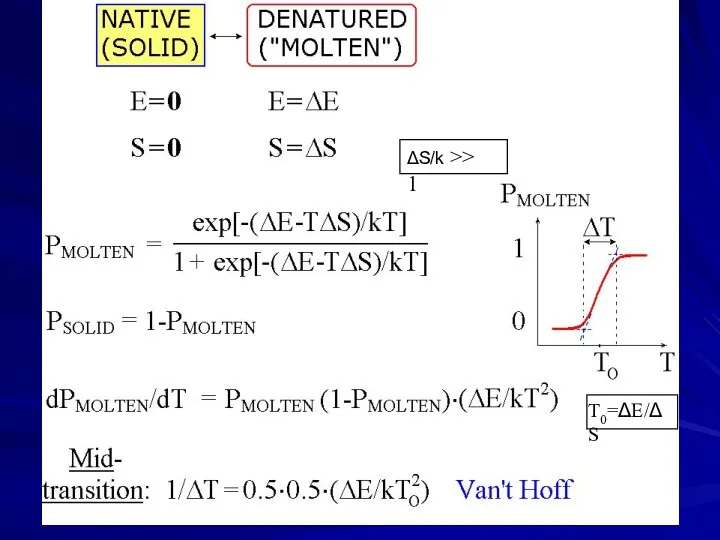
- 4. Protein denaturation in vitro: cooperative transition Solid protein structures can denaturate (decay), and then re-nature (fold)
- 5. transition
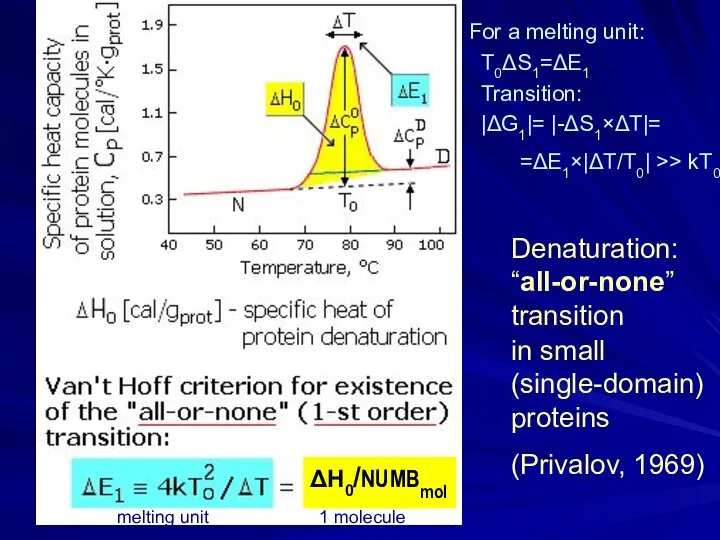
- 6. Denaturation: “all-or-none” transition in small (single-domain) proteins (Privalov, 1969) For a melting unit: T0ΔS1=ΔE1 Transition: |ΔG1|=
- 7. ΔS/k >> 1 T0=ΔE/ΔS
- 8. Jacobus Henricus van 't Hoff, Jr. (1852 –1911) The first Nobel prize in Chemistry, 1901 ПРИВАЛОВ
- 10. “All-or-none” decay of native protein structure: Ensures reliability and robustness of protein functioning Solid native state,
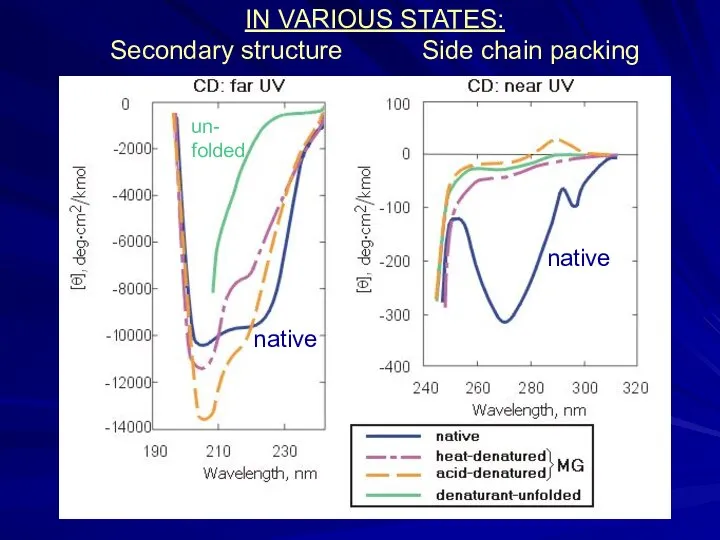
- 11. IN VARIOUS STATES: Secondary structure Side chain packing native un- folded native
- 12. “all-or-none” “all-or -none” “all-or-one”? sharp but gradual?
- 13. Евгений Исаакович Шахнович, 1957 Дмитрий Александрович Долгих, 1954 Геннадий Васильевич Семисотнов, 1947 Олег Борисович Птицын (1929-99)
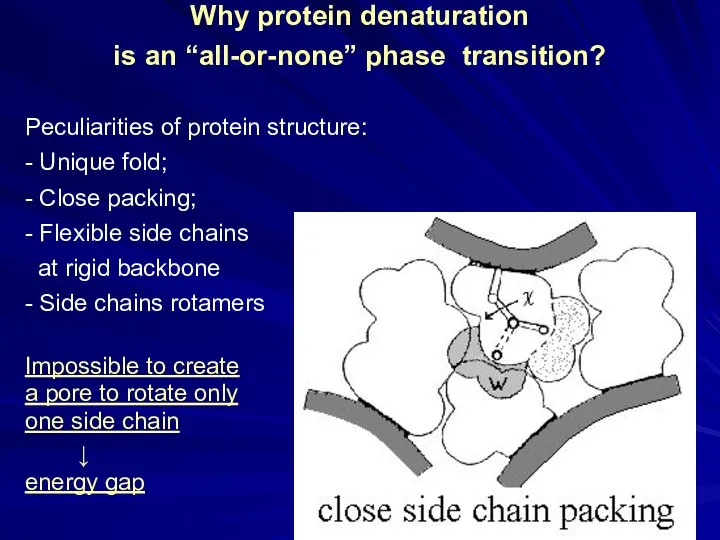
- 14. Why protein denaturation is an “all-or-none” phase transition? Peculiarities of protein structure: - Unique fold; -
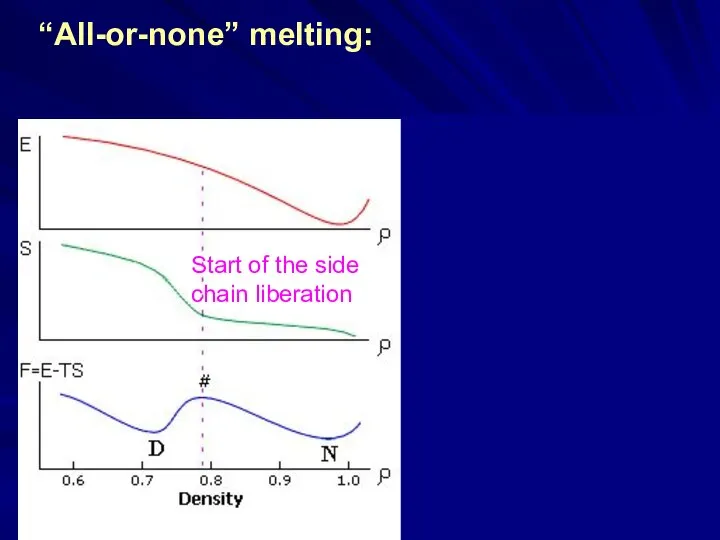
- 15. Start of the side chain liberation “All-or-none” melting:
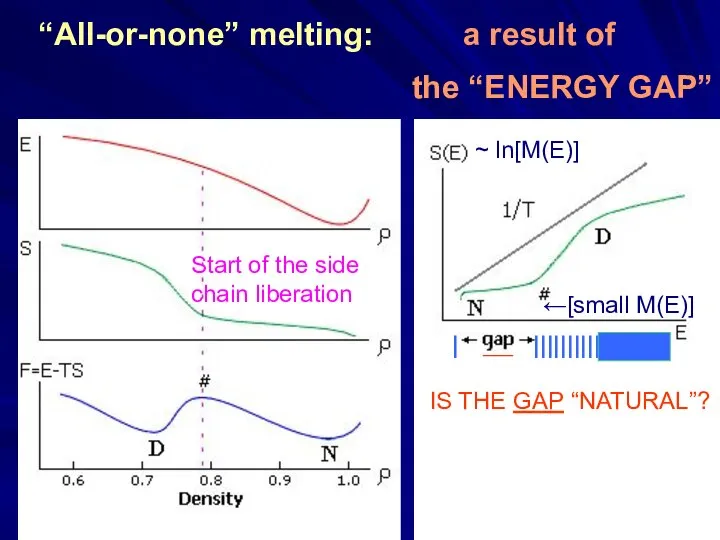
- 16. “All-or-none” melting: a result of the “ENERGY GAP” Start of the side chain liberation ~ ln[M(E)]
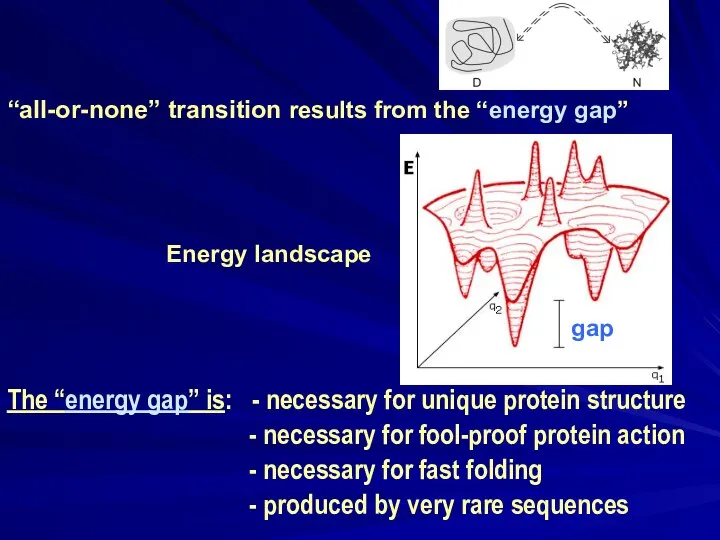
- 17. “all-or-none” transition results from the “energy gap” Energy landscape The “energy gap” is: - necessary for
- 18. GAP WIDTH: MAIN PROBLEM OF EXPERIMENTAL PROTEIN PHYSICS PHYSICAL ESTIMATE: =??? BIOLOGICAL ESTIMATE: 1 0F ~1010
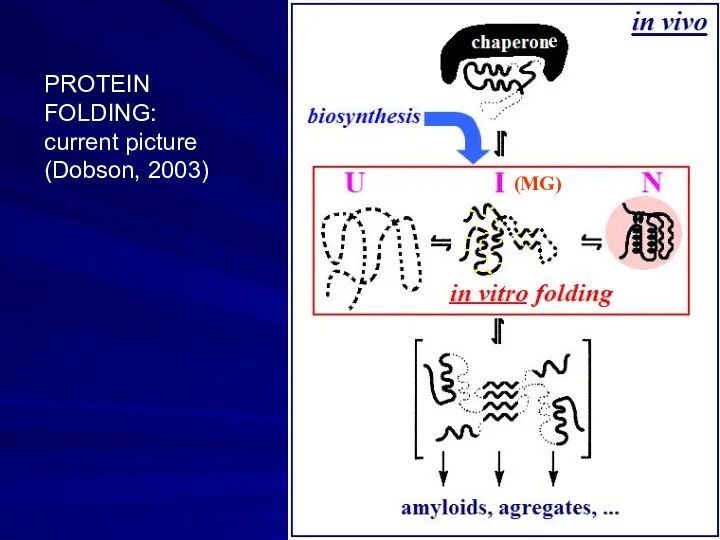
- 19. e PROTEIN FOLDING: current picture (Dobson, 2003) (MG)
- 21. Скачать презентацию








 Инструмент для фиксации элементов конструкций
Инструмент для фиксации элементов конструкций Презентация Робототехника и искусственный интеллект
Презентация Робототехника и искусственный интеллект  Силы в природе
Силы в природе Элемент Вольта
Элемент Вольта Ультрафиолетовое излучение
Ультрафиолетовое излучение Расчет собственной емкости космического аппарата с учетом параметров плазмы
Расчет собственной емкости космического аппарата с учетом параметров плазмы Ядерная энергия: её сущность и использование в технике и технологиях
Ядерная энергия: её сущность и использование в технике и технологиях Магниты на основе современных массивных высокотемпературных сверхпроводников
Магниты на основе современных массивных высокотемпературных сверхпроводников Основные положения МКТ
Основные положения МКТ Електричний струм. Початок. Луїджи Гальвані (1737-1798) Досліди з препарованою жабою Поява терміну “гальванічний елемент”
Електричний струм. Початок. Луїджи Гальвані (1737-1798) Досліди з препарованою жабою Поява терміну “гальванічний елемент”  Безопасность движения
Безопасность движения Постоянный ток. (Тема 5)
Постоянный ток. (Тема 5) Физика. Введение в предмет
Физика. Введение в предмет Кристалічні та аморфні тіла
Кристалічні та аморфні тіла Решение задач. Импульс, энергия, законы сохранения
Решение задач. Импульс, энергия, законы сохранения Физика. Молекулярно-кинетическая теория
Физика. Молекулярно-кинетическая теория Мембраны с нанопорами. Микро и нанокапсулы
Мембраны с нанопорами. Микро и нанокапсулы Диффузионное приближение Z
Диффузионное приближение Z Планетарная (ядерная) модель атома Атом – микрочастица, состоящая из положительно заряженного ядра и окружающих его электронов
Планетарная (ядерная) модель атома Атом – микрочастица, состоящая из положительно заряженного ядра и окружающих его электронов Измерение размеров малых тел. Лабораторная работа № 2
Измерение размеров малых тел. Лабораторная работа № 2 Волновые процессы
Волновые процессы Муфты приводов
Муфты приводов Электрооборудование автомобилей. Комплексные системы управления автомобильным двигателем. (Урок 13)
Электрооборудование автомобилей. Комплексные системы управления автомобильным двигателем. (Урок 13) Метод подобия явлений. Числа подобия. Критерии подобия
Метод подобия явлений. Числа подобия. Критерии подобия Магнитное поле
Магнитное поле Философские проблемы науки и техники. Типы рациональности. Неклассическая рациональность. (Лекция 9.2)
Философские проблемы науки и техники. Типы рациональности. Неклассическая рациональность. (Лекция 9.2) Действия электрического тока
Действия электрического тока Способы представления и параметры несинусоидальных величин
Способы представления и параметры несинусоидальных величин