Содержание
- 2. Formation of the Earth Apollo Space Program (1960’s) Otto Schmidt Cosmic Dust Planet (100 million years)
- 3. Thermal Consequences Earth’s Core Molten Fe ( Density 7.86 g/cc) Ni ( Density 8.9 g/cc) Outer
- 4. Formation of the Mantle The less dense material will go toward the surface (Polar Oxides of
- 5. Isotope Distribution of the Earth Investigation of the History of the Earth primarily relied on isotope
- 6. Appearance of the Atmosphere Did the atmosphere suddenly appear ? Isotope Analysis gives a clue Claude
- 7. Isotopes of Xe Xenon has 9 isotopes With the following distribution 124Xe 0.1% , 126Xe 0.09%,
- 8. Distribution of Xe isotopes Nucleosynthesis gives rise to 129Xe β- Decay of 129I 129Xe (t1/2 =
- 9. Differentiation The Atmosphere was formed due to OUT GASSING of the mantle (Heat) & Volcanic Activity
- 10. Age of differentiation From the ratio of 129 Xe in the Mantle to that of 129
- 11. Ratios of Isotopes The Argon trapped in Mantle evolved from the radioactive decay of 40K 40K
- 12. Conclusions from Isotope Analysis ∴ If outgassing occurred at the beginning the atmosphere would not contain
- 13. Collecting the evidence The other 15% has arisen due to slow release over 4.4 billion years
- 14. Early Atmosphere Majors: CO2, N2, H2O (Water Vapour) Traces: CH4, NH3, SO2, HCl Water Vapour Oceans
- 15. Origin of Life Stanley Miller (1950) “ Early Earth ” Experimental Setup CH4, NH3, H2, H2O(g)
- 16. Formation of Simple Amino Acids Glycine was found How Glycine (NH2CH2COOH) Formed HCOH + NH3 +
- 17. Murchison Meteor A number of the compounds discovered in the discharge fractions are precursors to life.
- 18. Early Energy System The first living organisms gained their energy by a fermentation of the organic
- 19. Role of Blue Green Algae Blue Green Algae & Photosynthetic Bacteria developed to use water as
- 20. Decline of Anaerobic Bacteria Problem for Anaerobic Organisms Evidence of the appearance of Oxygen is indicated
- 21. Oxygen Rich Planet Oxygen Rich Planet The build up of Oxygen in the atmosphere led to
- 22. Oxygen Rich Planet Respiration utilized the photosynthetic Compounds (Sugar ) to produce Energy (CH2O)n + nO2
- 23. The trouble with oxygen The ultilization of oxygen in producing energy resulted in emergence Eukarotic cells
- 24. The present atmosphere The present atmosphere has arisen from (1) The distance of the earth from
- 25. Distance from the Sun The distance from the Sun determines the kinetic energy (KE) of the
- 26. Influence of Earth’s Mass The ability of molecules to remain in the atmosphere is also related
- 27. Escape Velocity Escape Velocity (Ve) Ve = (2Gm/R)1/2 m = Mass of the Planet G= Universal
- 28. Escape Velocity The ability of molecules to remain in an atmosphere is related to the mass.
- 29. No H or He in Earth’s Atmosphere At 600 K (Upper Atmosphere ) For H atoms
- 30. Little CO2 in atmosphere For Oxygen only 1 in 1084 atoms exceeds the escape velocity .This
- 31. Earth ,Venus & Mars Surface Characteristics of Planets Temperature Pressure (bar)* Venus 732 K (459oC) 90
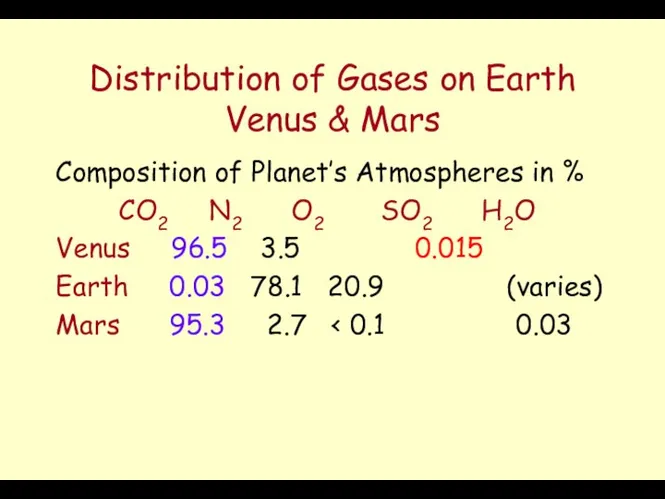
- 32. Distribution of Gases on Earth Venus & Mars Composition of Planet’s Atmospheres in % CO2 N2
- 33. Role of Shellfish Presence of Life on Earth has removed Carbon dioxide from the Atmosphere and
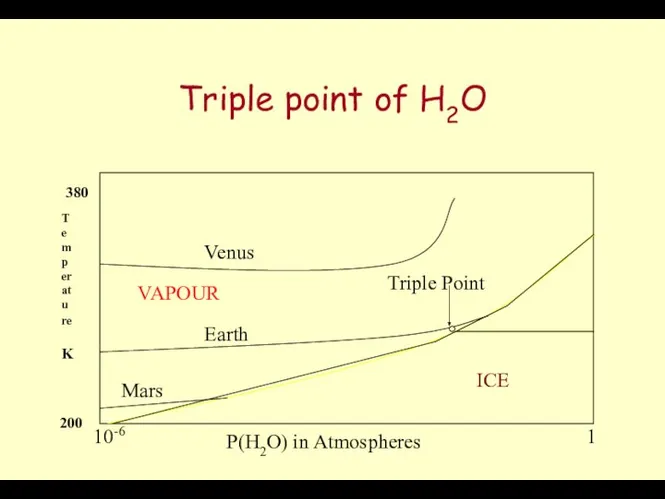
- 34. Triple point of H2O P(H2O) in Atmospheres Temperature K Venus Earth Mars ICE WATER VAPOUR Triple
- 35. Water ( Solid,Liquid, Gas) The Surface temperature of the Earth at 1 atmosphere Pressure is close
- 36. Super Greenhouse & Acid Rain On Venus ,the high level of CO2 and its distance from
- 37. Current Atmosphere Composition of Current Atmosphere %Vol N2, O2, Ar, CO2, H2O 78.08 20.95 0.93 0.03
- 38. Present Level of Oxygen The present level of Oxygen in the atmosphere is balanced at a
- 39. Structure of Atmosphere Earth’s Atmosphere Earth’s Surface Troposphere Stratosphere Mesosphere Thermosphere REGION 10-16 km (-56oC) 50
- 40. Ozone Layer Ozone in the Stratosphere ≈ 16 - 50km above the Earth’s Surface acts as
- 41. Ozone and Radiation Oxygen that lies above the stratosphere filters out UV light 120nm - 220nm
- 42. Effects of Reduction in Ozone (Effects of Reduction) 1% Reduction In O3 2% increase in UV-B
- 43. Chlorofluorocarbons & Ozone Destruction of the Ozone Layer discovered in 1970’s by CFC’s ( Chlorofluorocarbons) First
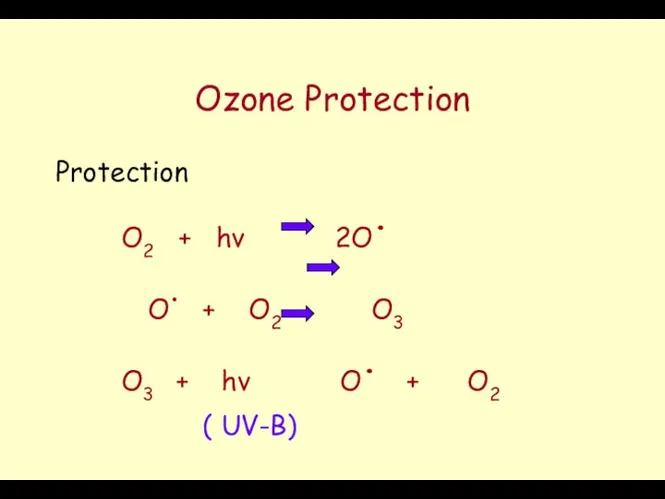
- 44. Ozone Protection Protection O2 + hν 2O. O. + O2 O3 O3 + hν O. +
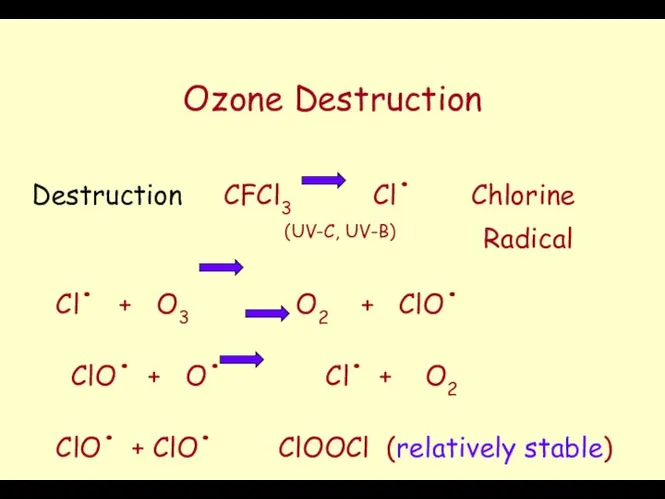
- 45. Ozone Destruction Destruction CFCl3 Cl. Chlorine (UV-C, UV-B) Radical Cl. + O3 O2 + ClO. ClO.
- 46. Control of CFC’s CFC’s are now under strict control and their use has been curtailed. Australia
- 47. Uses of CFC’s Compound Use CFC- 11 CFCl3 Refrigeration, aerosol, foam CFC-12 CF2Cl2 sterilization, cosmetics food
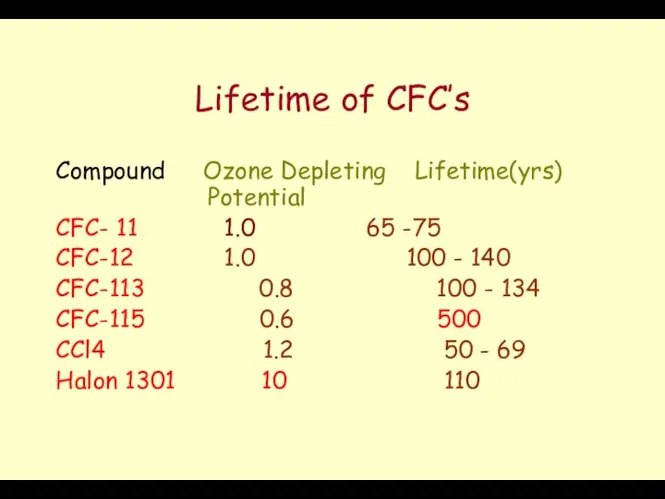
- 48. Lifetime of CFC’s Compound Ozone Depleting Lifetime(yrs) Potential CFC- 11 1.0 65 -75 CFC-12 1.0 100
- 49. Naming of CFC’s ( 90 Rule) CFC’s name is related to its Formula. CFC 123 123
- 50. Chloromonoxide Evidence for the destruction has been linked to the catalytically active Chloro monoxide ClO. &
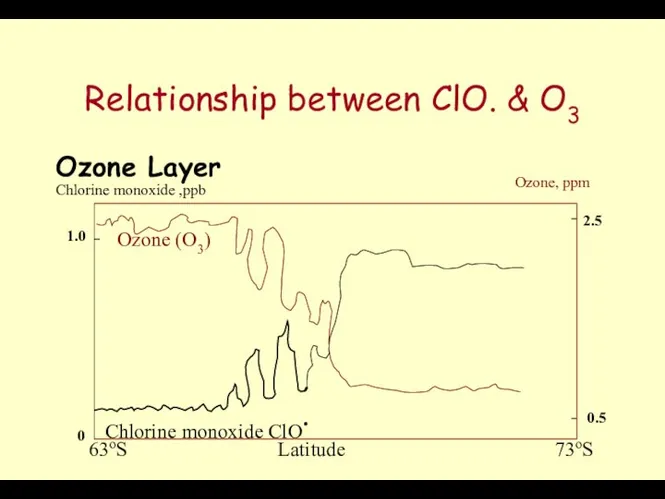
- 51. Relationship between ClO. & O3 Ozone Layer Ozone (O3) Chlorine monoxide ClO. Chlorine monoxide ,ppb Ozone,
- 52. Thickness of Ozone Layer The thickness of the Ozone Layer is expressed in Dobson units (DU)
- 53. Other Ozone Depleters But has the reduction and removal of CFC’s solved the problem of the
- 54. Interactive Catalytic Forms Destruction: Halide Radicals destroy Ozone. The majority of Chlorine does not exit as
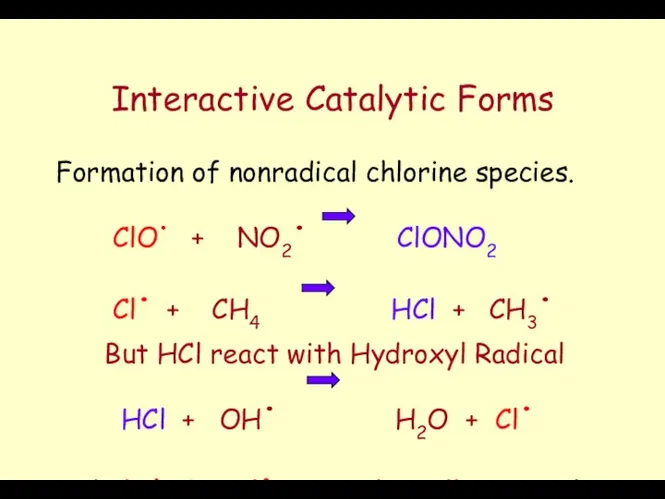
- 55. Interactive Catalytic Forms Formation of nonradical chlorine species. ClO. + NO2. ClONO2 Cl. + CH4 HCl
- 56. Origin of Ozone Hole The major destruction of the hole in the lower atmosphere occurs as
- 57. Ice crystal formation Nitric acid in the atmosphere forms from the reaction between OH.& NO2. Catalytically
- 58. Possible Role of CO2 “ CO2 acts as a blanket in the lower atmosphere,” says Salawitch.
- 59. Impenetrable Vortex formation The usual warming mechanism from of O + O2 O3 + Heat is
- 60. PSC’s Matter cannot readily enter this vortex and the air inside is isolated and remains cold
- 61. HCL attachment Gas phase HCl attaches to the ice particle Crystal HNO3.3H2O of HCl HCl HCl
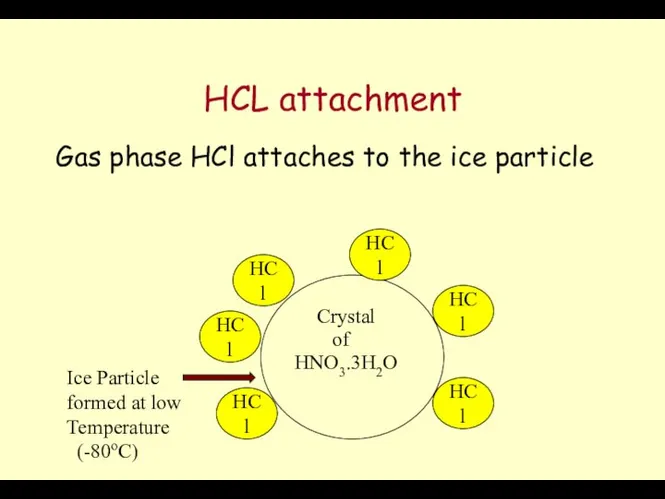
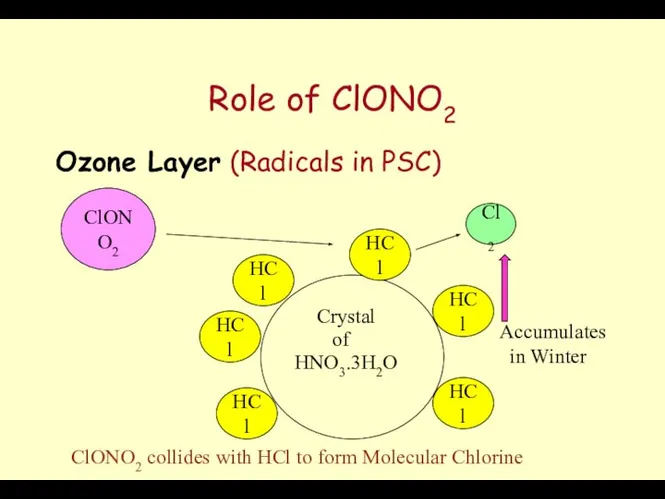
- 62. Role of ClONO2 Ozone Layer (Radicals in PSC) Crystal HNO3.3H2O of HCl HCl HCl HCl HCl
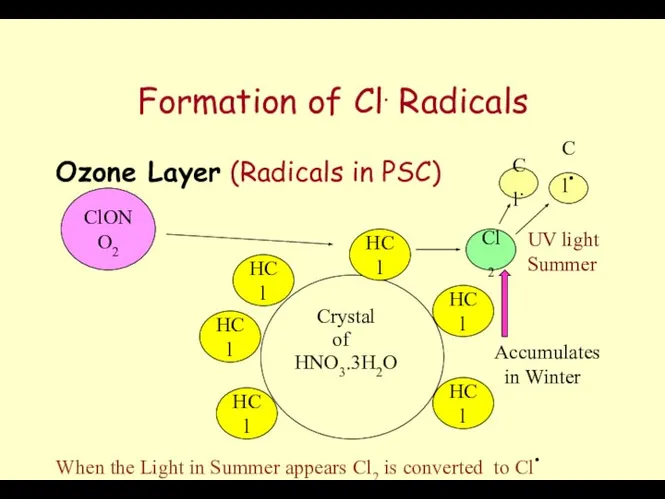
- 63. Formation of Cl. Radicals Ozone Layer (Radicals in PSC) Crystal HNO3.3H2O of HCl HCl HCl HCl
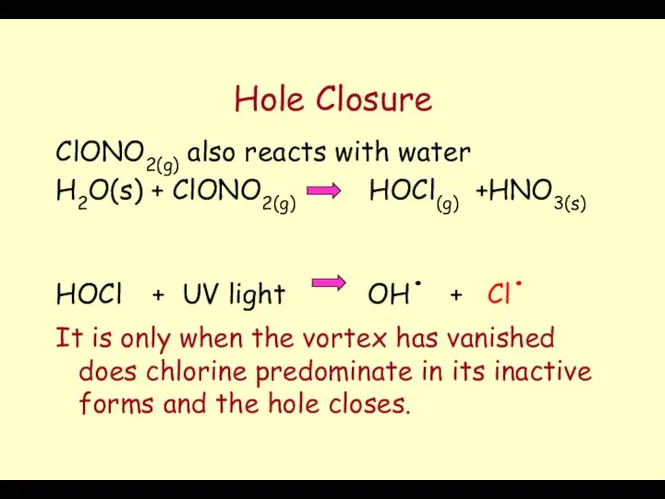
- 64. Hole Closure ClONO2(g) also reacts with water H2O(s) + ClONO2(g) HOCl(g) +HNO3(s) HOCl + UV light
- 65. Dimer ClOOCl ClO. also builds up in the dark and this dimerizes to for a relatively
- 66. Antarctic and Arctic Vortexes Ozone Layer (PSC’s) The Antarctic vortex is more intense than the Arctic
- 67. Possible Link Ozone Layer “But PSC’s were here long before any one had the bright Idea
- 68. Further Reading Ozone Layer “The Hole Story” by G.Walker New Scientist, p24 , March 2000 Websites
- 70. Скачать презентацию
























































 Внесение удобрений разбрасыванием
Внесение удобрений разбрасыванием Номенклатура органических соединений
Номенклатура органических соединений Презентація уроку «Різноманітність речовин. Поняття про прості та складні речовини, неорганічні та органічні речовини» Підготув
Презентація уроку «Різноманітність речовин. Поняття про прості та складні речовини, неорганічні та органічні речовини» Підготув Химические реакции
Химические реакции  Комплексные соединения
Комплексные соединения Химические свойства алкенов
Химические свойства алкенов Структурно-механiчнi (реологiчнi) властивостi харчових мас
Структурно-механiчнi (реологiчнi) властивостi харчових мас Химическая кинетика. (Лекция 4)
Химическая кинетика. (Лекция 4) Ископаемое сырье органической химии
Ископаемое сырье органической химии L-2-Chemical bonds
L-2-Chemical bonds Возникновение и развитие понятия «валентность» в период с 1850 по 1865 гг
Возникновение и развитие понятия «валентность» в период с 1850 по 1865 гг Нанотехнології в нашому житті. Загроза цивілізації, або стрибок у майбутнє
Нанотехнології в нашому житті. Загроза цивілізації, або стрибок у майбутнє Фізичні та хімічні явища. Хімічні реакції та явища, що їх супроводжують
Фізичні та хімічні явища. Хімічні реакції та явища, що їх супроводжують  Тест 1.Тип кристаллической решетки у белого фосфора…. а) молекулярный б) атомный в) ионный а) молекулярный Р4 2. У фосфора…..
Тест 1.Тип кристаллической решетки у белого фосфора…. а) молекулярный б) атомный в) ионный а) молекулярный Р4 2. У фосфора…..  Презентация по химии Галогены в организме человека
Презентация по химии Галогены в организме человека Презентация по Химии "Многоатомные спирты" - скачать смотреть бесплатно_
Презентация по Химии "Многоатомные спирты" - скачать смотреть бесплатно_ Решение экспериментальных задач по теме "Подгруппы азота и углерода"
Решение экспериментальных задач по теме "Подгруппы азота и углерода" Определение ректификации
Определение ректификации Степень окисления
Степень окисления Люминесцентный анализ
Люминесцентный анализ Растворы и процессы, проходящие в растворах. Протолитическая теория кислот и оснований
Растворы и процессы, проходящие в растворах. Протолитическая теория кислот и оснований Презентация по Химии "Витаминный алфавит" - скачать смотреть
Презентация по Химии "Витаминный алфавит" - скачать смотреть  Исследование свойств и применение в медицине алкалоидов. Извлечение алкалоидов из травы белены черной.
Исследование свойств и применение в медицине алкалоидов. Извлечение алкалоидов из травы белены черной. Периодическая система химических элементов Д.И. Менделеева и строение атома
Периодическая система химических элементов Д.И. Менделеева и строение атома Комплексні сполуки
Комплексні сполуки Нуклеиновые кислоты
Нуклеиновые кислоты Презентация по Химии "В даний час з нафти отримують тисячі продуктів." - скачать смотреть бесплатно
Презентация по Химии "В даний час з нафти отримують тисячі продуктів." - скачать смотреть бесплатно Белки простые и сложные
Белки простые и сложные