Содержание
- 2. Contents 1. Understanding of Rate of Reaction 2. Factors Affecting Rate of Reaction 3. Collision Theory
- 3. Chemical reactions require varying lengths of time for completion, depending on the characteristics of the reactants
- 4. Chemical kinetics is the study of the speed with which a chemical reaction occurs and the
- 5. During chemical reaction, reactants are consumed, products are formed. Amount of reactants decreases↓, amount of product
- 6. mol/м3⋅s mol/м2⋅s mol/l⋅s THE RATE OF CHEMICAL REACTION HOMOGENEUOS REACTION RATE is the change moles of
- 7. FACTORS AFFECTING THE REACTION RATE: Nature of the reactant (gas, liquid or solid) Surface area Concentration
- 8. Nature of the reactant and products In organic reaction a large number of bonds are broken
- 9. HOW SURFACE AREA AFFECTS THE RATE OF REACTION? CaCO3(s)+2HCl(ℓ) → CaCl2(s)+H2O(ℓ)+CO2(g) marble piece marble powder HCl
- 10. LAW OF MASS ACTION At constant temperature, the rate of a chemical reaction is directly proportional
- 11. Homogeneous reactions which occur in one phase only. It may be a gaseous phase or a
- 12. THE EFFECT OF TEMPERATURE ON REACTION RATE described by principle of Vant Hoff “In the temperature
- 13. Why does increased temperature increase the rate of reaction? At a higher temperature, particles have more
- 14. The rate of a reaction depends on the temperature at which it is run. As the
- 15. DETERMINE THE ACTIVATION ENERGY Еа > 100 kJ – big value, the reaction proceeds very slowly
- 16. CHEMICAL EQUILIBRIUM The irreversible reaction moves in one direction only from reactant to product: The reversible
- 17. "At a given temperature, the product of concentrations of the reaction product each raised to the
- 18. 0 106 10-6 Reversible reaction Irreversible reaction No reaction occurs Value of chemical equilibrium constant
- 19. HOMOGENEOUS REACTION HETEROGENEOUS REACTION
- 20. The most important condition for the equilibrium is that ΔG of the reaction should be 0.
- 21. CONDITIONS THAT AFFECT THE EQUILIBRIUM The conditions that affect the equilibrium are described according to Le
- 22. FACTORS AFFECTING EQUILIBRIUM Some of the factors which affect the equilibrium constant value are: 1. Concentration.
- 23. 3. Pressure. The pressure change affects only on those systems where in at least one of
- 24. CATALYST A catalyst will change the rate of reaction. A catalyst only changes the rate of
- 25. Catalysts are substances that change the rate of a reaction without itself being consumed. Catalysts never
- 26. QUIZ ME NEXT 1 What is the discipline that studies chemical reactions with respect to reaction
- 27. QUIZ ME NEXT 2 What drives chemical reactions? Electrons Physical conditions Energy Activation Energy
- 28. QUIZ ME NEXT 3 Which one of the following reactions reacts the most rapidly at room
- 30. Скачать презентацию



























 Химическая природа и состав нефти и газа. Физико-химические свойства нефтей и нефтепродуктов
Химическая природа и состав нефти и газа. Физико-химические свойства нефтей и нефтепродуктов Молекулы и атомы. Простые и сложные вещества
Молекулы и атомы. Простые и сложные вещества Lead
Lead Презентация по Химии "Высшие природные полимеры - Белки и Нуклеиновые кислоты" - скачать смотреть
Презентация по Химии "Высшие природные полимеры - Белки и Нуклеиновые кислоты" - скачать смотреть  Алмаз, графит и фуллерен Строковой Натальи 9б
Алмаз, графит и фуллерен Строковой Натальи 9б  Взаимодействие кислоты с основанием
Взаимодействие кислоты с основанием Минералогия и кристаллография
Минералогия и кристаллография Оксид кремния
Оксид кремния Неметаллы. Аллотропия. Куцапкина Людмила Васильевна учитель химии ГБОУ гимназии № 343 Невского района Санкт- Петербурга
Неметаллы. Аллотропия. Куцапкина Людмила Васильевна учитель химии ГБОУ гимназии № 343 Невского района Санкт- Петербурга Рідкісні кристали і полімери. Їх властивості та застосування
Рідкісні кристали і полімери. Їх властивості та застосування Тема урока : Скорость химической реакции
Тема урока : Скорость химической реакции  Презентация Правила безопасности в кабинете химии
Презентация Правила безопасности в кабинете химии Глицерин
Глицерин Контейнерная химия: комплексоны, комплексообразование, применение
Контейнерная химия: комплексоны, комплексообразование, применение Тотығып фосфорлану
Тотығып фосфорлану Оксид серы(VI). Серная кислота
Оксид серы(VI). Серная кислота Явище ізомерії. Структурна ізомерія. Близнюки органічного світу
Явище ізомерії. Структурна ізомерія. Близнюки органічного світу Презентация по Химии "Водні Ресурси" - скачать смотреть
Презентация по Химии "Водні Ресурси" - скачать смотреть  Алюминий, его физические и химические свойства
Алюминий, его физические и химические свойства неметаллы Общая характеристика
неметаллы Общая характеристика Изучение содержания углекислого газа в классном помещении и определение оптимальных условий для проветривания. Муниципальное об
Изучение содержания углекислого газа в классном помещении и определение оптимальных условий для проветривания. Муниципальное об Вода. Свойства воды
Вода. Свойства воды Аттестационная работа. Рабочая программа курса внеурочной деятельности «Химия. Мои первые опыты»
Аттестационная работа. Рабочая программа курса внеурочной деятельности «Химия. Мои первые опыты» Протолитическое равновесие в водных и неводных растворах
Протолитическое равновесие в водных и неводных растворах Методы трансформации карбоновых кислот и их производных
Методы трансформации карбоновых кислот и их производных Простые вещества, металлы
Простые вещества, металлы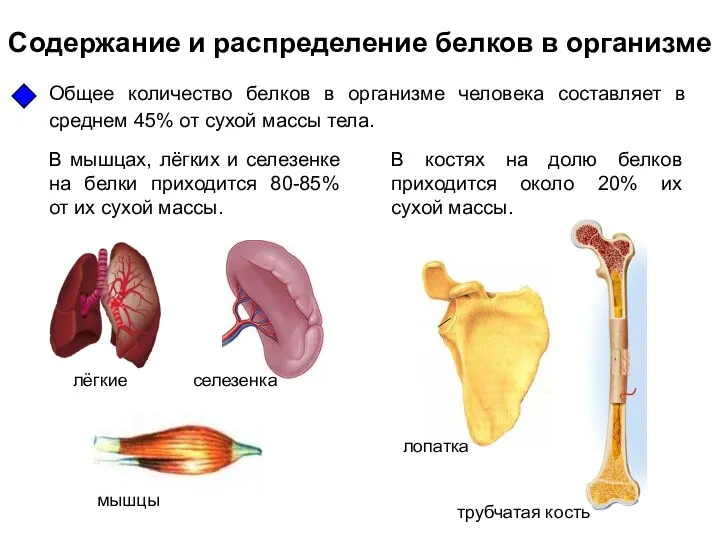 Содержание и распределение белков в организме
Содержание и распределение белков в организме Структурный тип
Структурный тип