Содержание
- 2. * Topic 1 – Atomic Structure and the Periodic Table
- 3. * * Periodic Table Introduction How would you arrange these elements into groups?
- 4. * Development of the Periodic Table
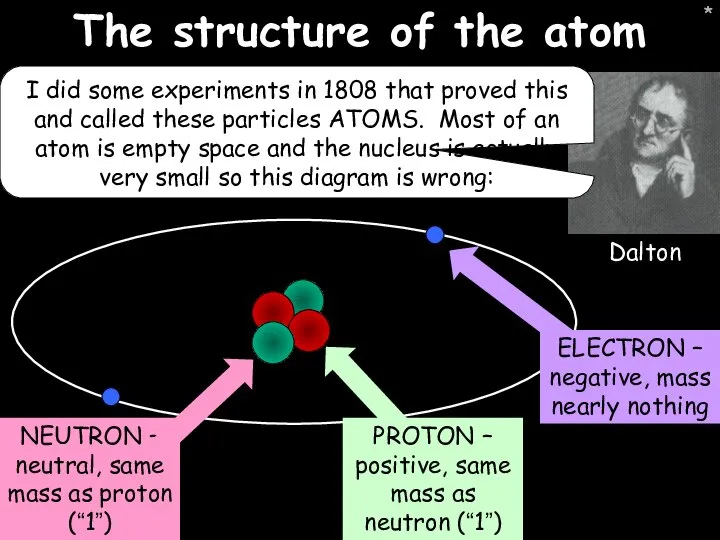
- 5. * * The structure of the atom I did some experiments in 1808 that proved this
- 6. * * Mass and atomic number
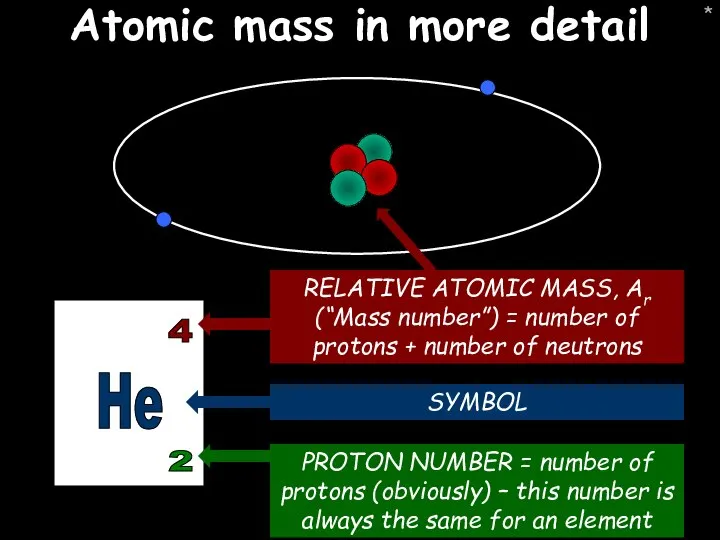
- 7. * Atomic mass in more detail
- 8. * * Mass and atomic number H 1 1 B 5 11 O 8 16 Na
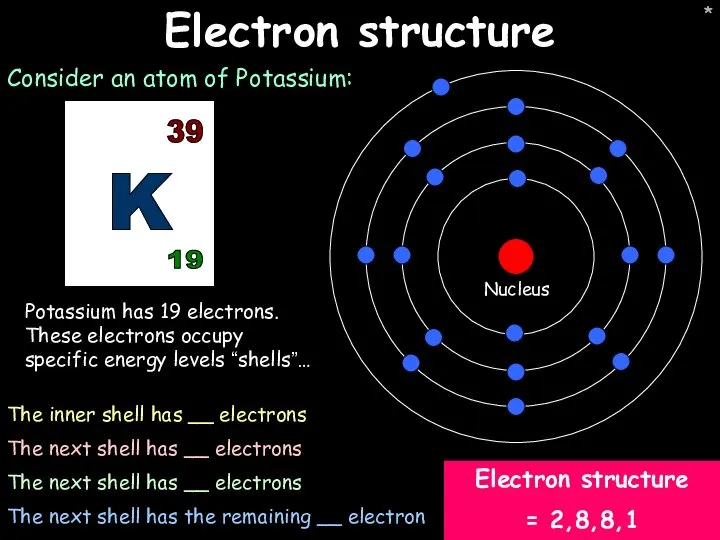
- 9. * * Electron structure Consider an atom of Potassium: Potassium has 19 electrons. These electrons occupy
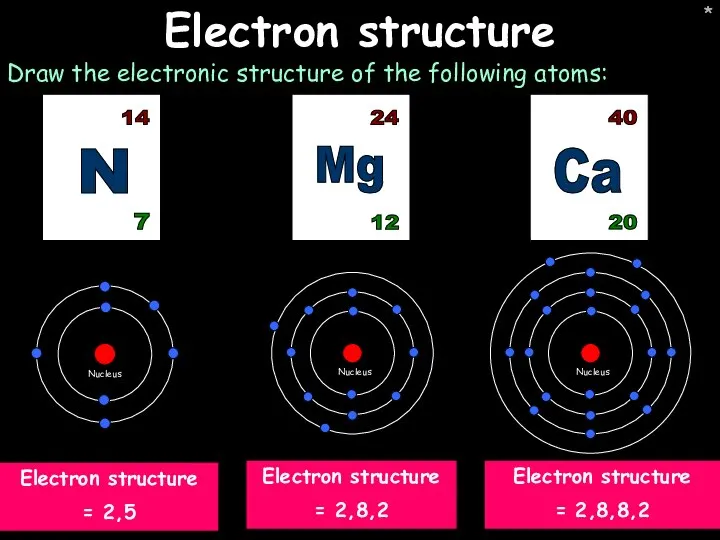
- 10. * * Electron structure Draw the electronic structure of the following atoms: Ca 20 40 Electron
- 11. * * Periodic table Horizontal rows are called PERIODS
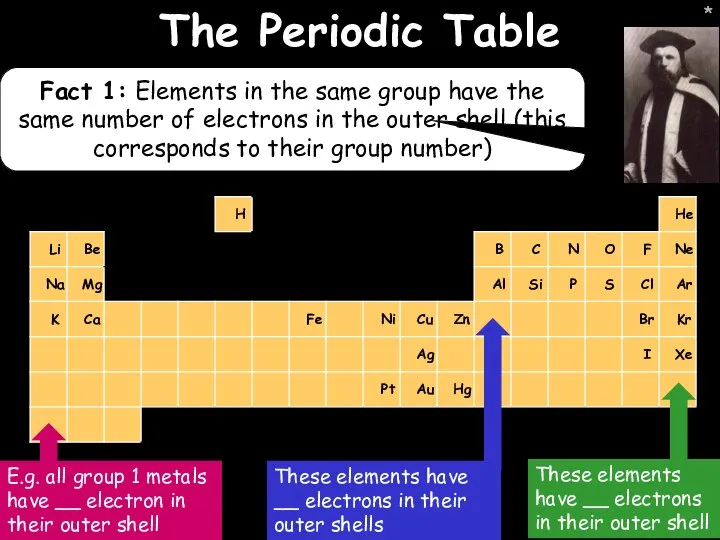
- 12. * * The Periodic Table Fact 1: Elements in the same group have the same number
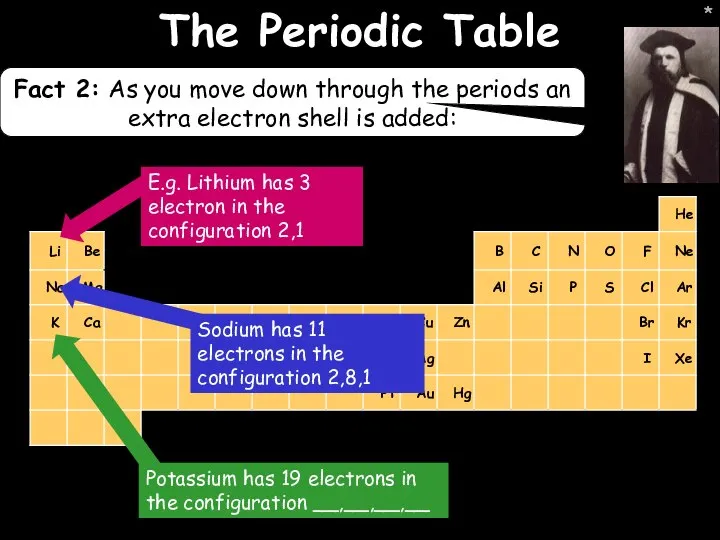
- 13. * * The Periodic Table Fact 2: As you move down through the periods an extra
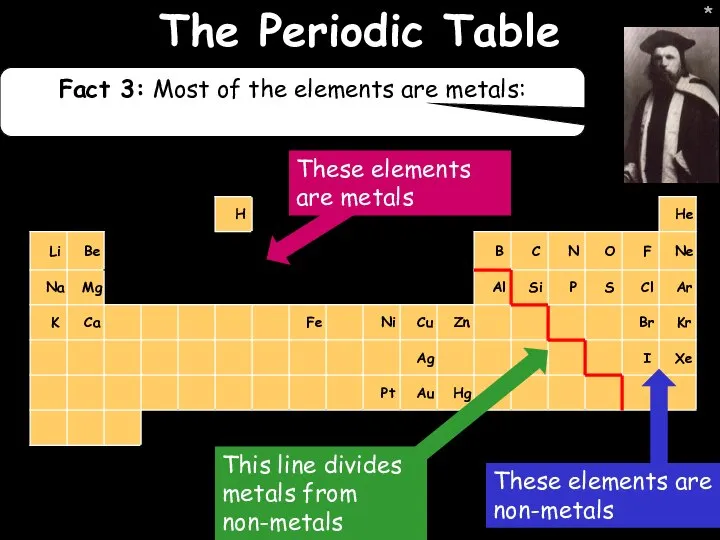
- 14. * * The Periodic Table Fact 3: Most of the elements are metals:
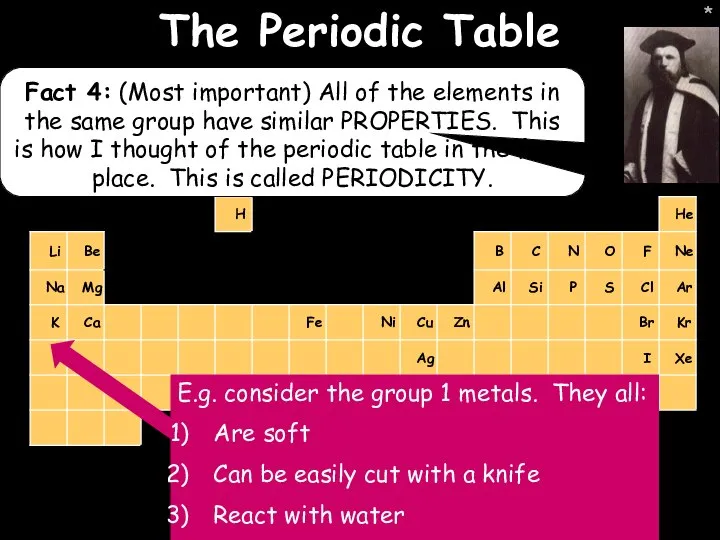
- 15. * * The Periodic Table Fact 4: (Most important) All of the elements in the same
- 16. * * Isotopes An isotope is an atom with a different number of neutrons:
- 17. * Strange atomic masses When you look at a periodic table sometimes the atomic mass is
- 18. * Topic 2 – Ionic Compounds and Analysis
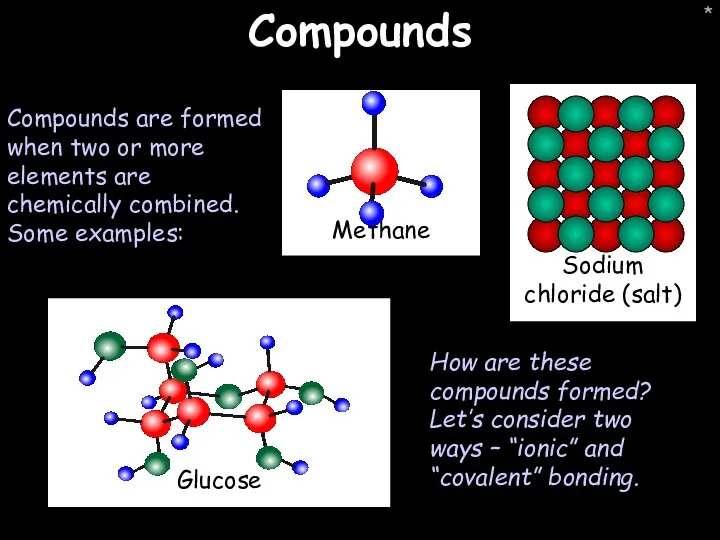
- 19. * Compounds Compounds are formed when two or more elements are chemically combined. Some examples: How
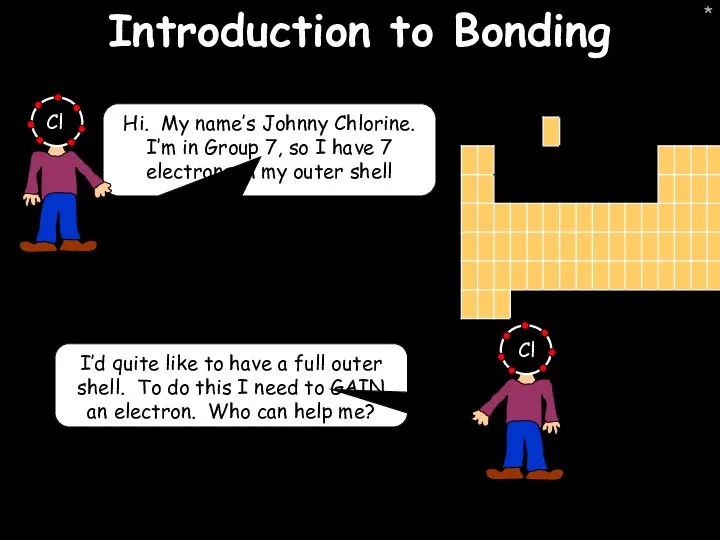
- 20. * Introduction to Bonding Hi. My name’s Johnny Chlorine. I’m in Group 7, so I have
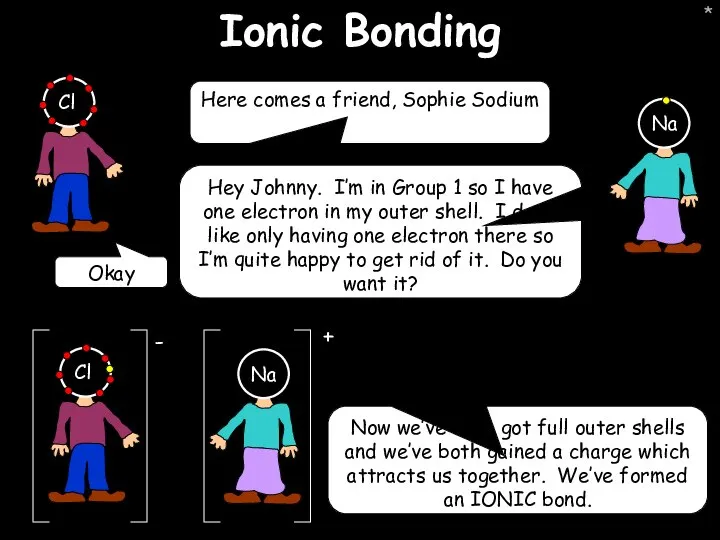
- 21. * Ionic Bonding Here comes a friend, Sophie Sodium Hey Johnny. I’m in Group 1 so
- 22. * Ions An ion is formed when an atom gains or loses electrons and becomes charged:
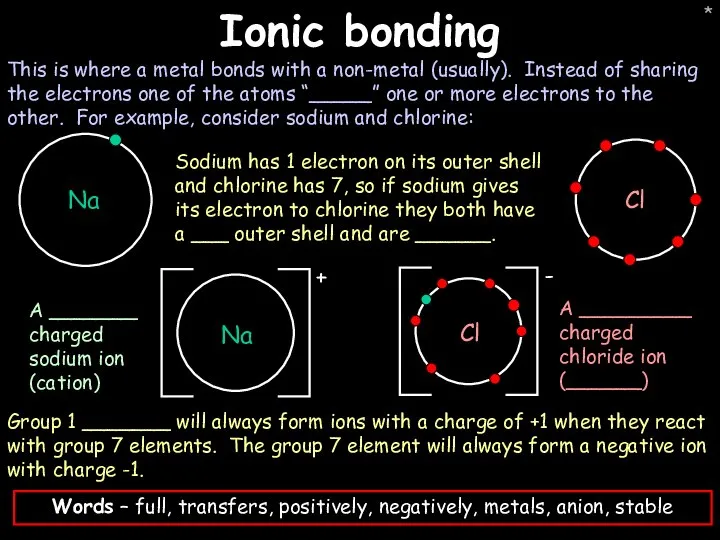
- 23. * Ionic bonding This is where a metal bonds with a non-metal (usually). Instead of sharing
- 24. * Naming compounds
- 25. * Naming compounds
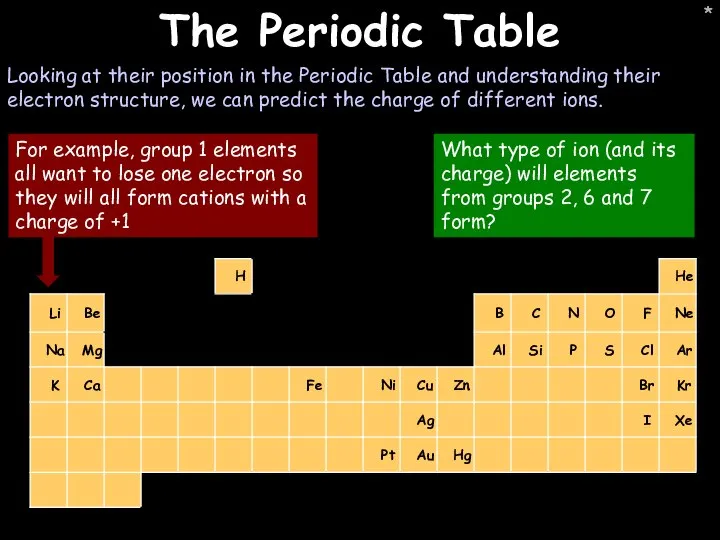
- 26. * * The Periodic Table Looking at their position in the Periodic Table and understanding their
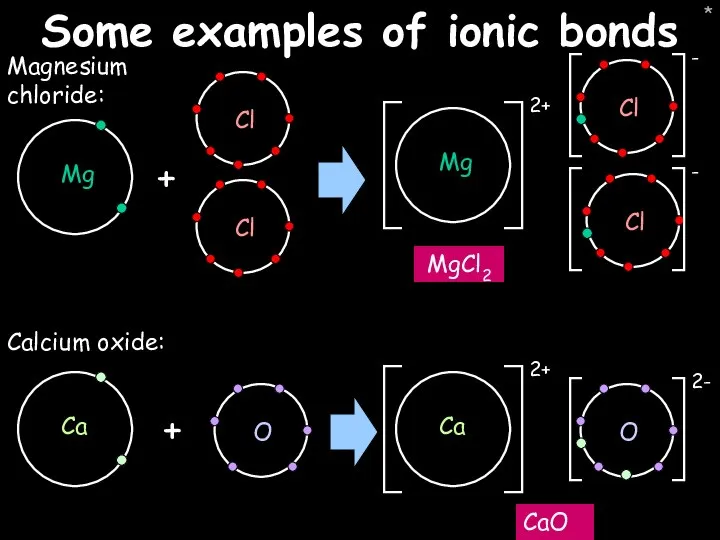
- 27. * Some examples of ionic bonds Magnesium chloride: MgCl2 + Calcium oxide: CaO
- 28. * * Balancing ions Determine the formula of these compounds: Sodium chloride Magnesium oxide Magnesium chloride
- 29. * Giant Ionic Structures When many positive and negative ions are joined they form a “giant
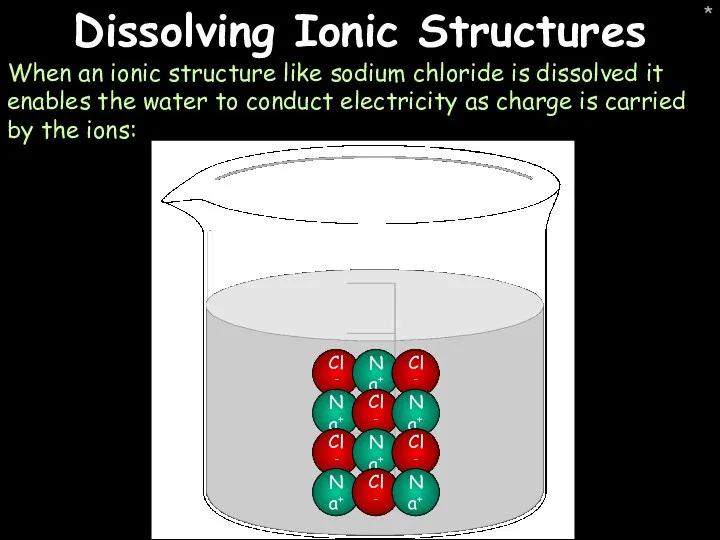
- 30. * Dissolving Ionic Structures When an ionic structure like sodium chloride is dissolved it enables the
- 31. * Solubility rules The following guidelines are useful in working out if a substance will dissolve:
- 32. * * Precipitation Reactions A precipitation reaction occurs when an insoluble solid is made by mixing
- 33. * Precipitates Some metal compounds form precipitates, i.e. an insoluble solid that is formed when sodium
- 34. * Barium Sulfate Barium sulfate can be used as part of a “barium meal” to X-ray
- 35. * Flame tests Compounds containing lithium, sodium, potassium, calcium and barium ions can be recognised by
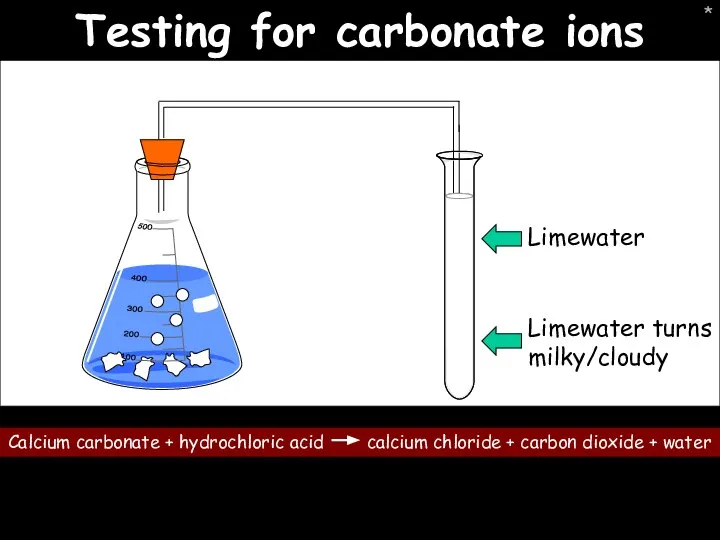
- 36. * Testing for carbonate ions
- 37. * Testing for chloride and sulfate ions Test 1: Chloride ions Add a few drops of
- 38. * Spectroscopy Spectroscopy is kind of like a flame test but using a “spectroscope” to see
- 39. * Topic 3 – Covalent Compounds and Separation Techniques
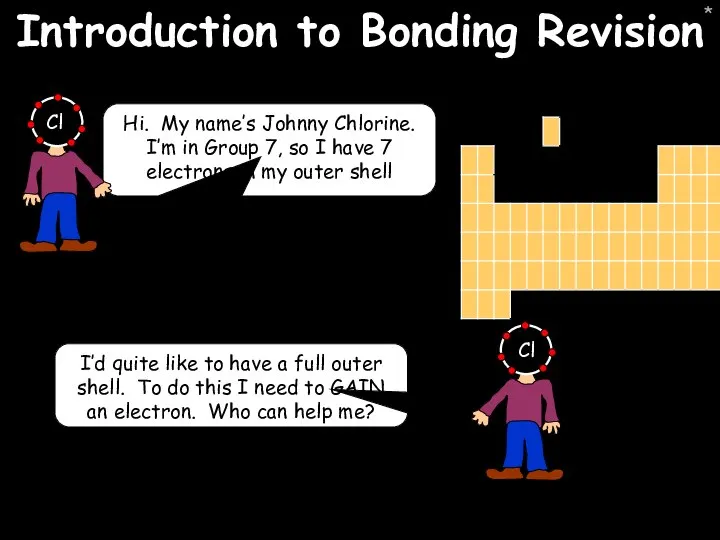
- 40. * Introduction to Bonding Revision Hi. My name’s Johnny Chlorine. I’m in Group 7, so I
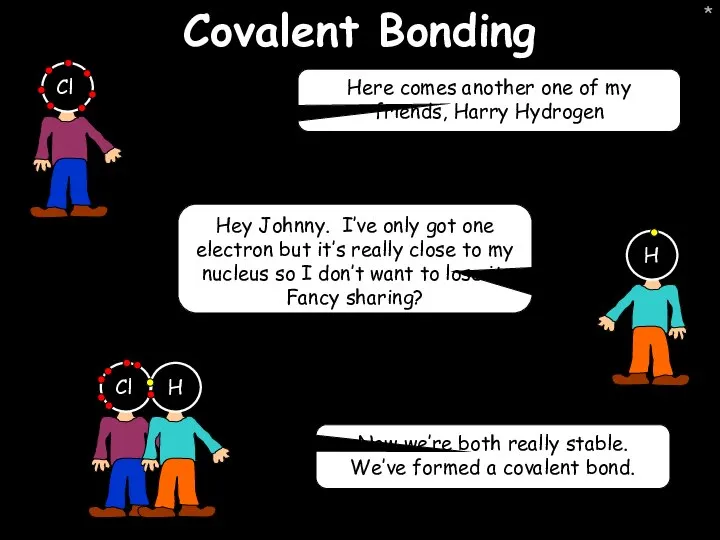
- 41. * Covalent Bonding Here comes another one of my friends, Harry Hydrogen Hey Johnny. I’ve only
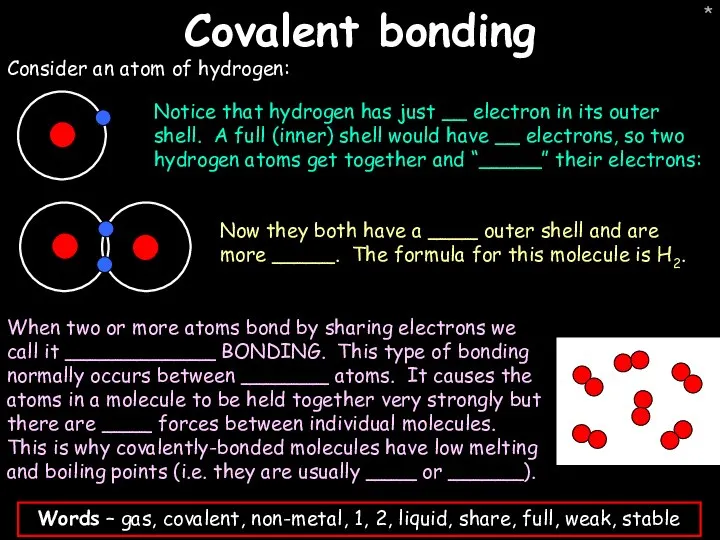
- 42. * Covalent bonding Consider an atom of hydrogen:
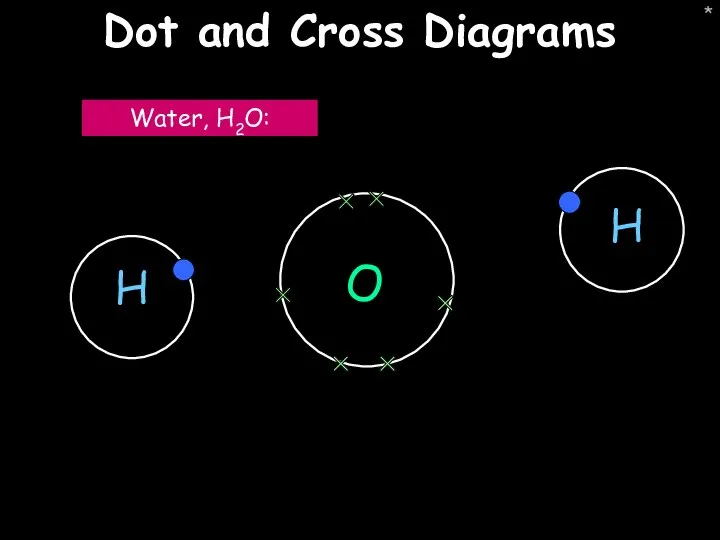
- 43. * Dot and Cross Diagrams O Water, H2O:
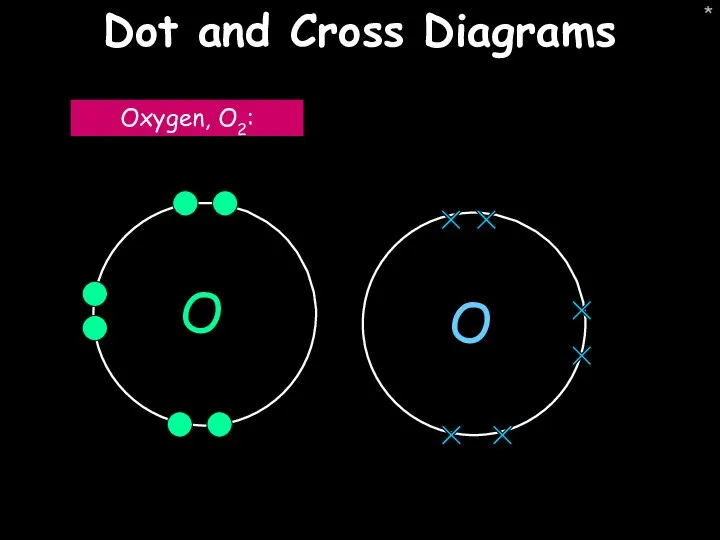
- 44. * Dot and Cross Diagrams Oxygen, O2: O O
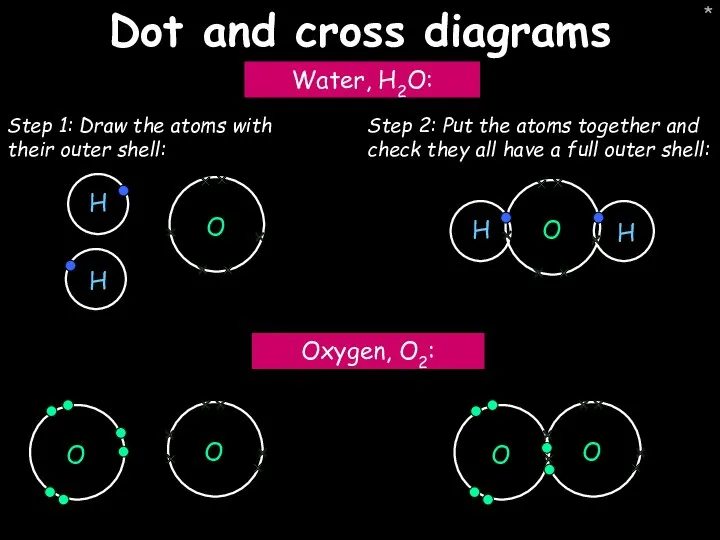
- 45. * Dot and cross diagrams Water, H2O: Oxygen, O2: Step 1: Draw the atoms with their
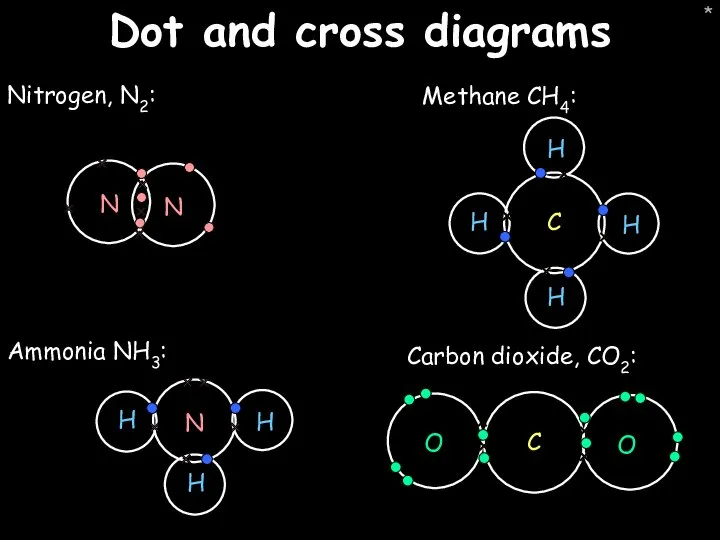
- 46. * Dot and cross diagrams Nitrogen, N2: Carbon dioxide, CO2: Ammonia NH3: Methane CH4:
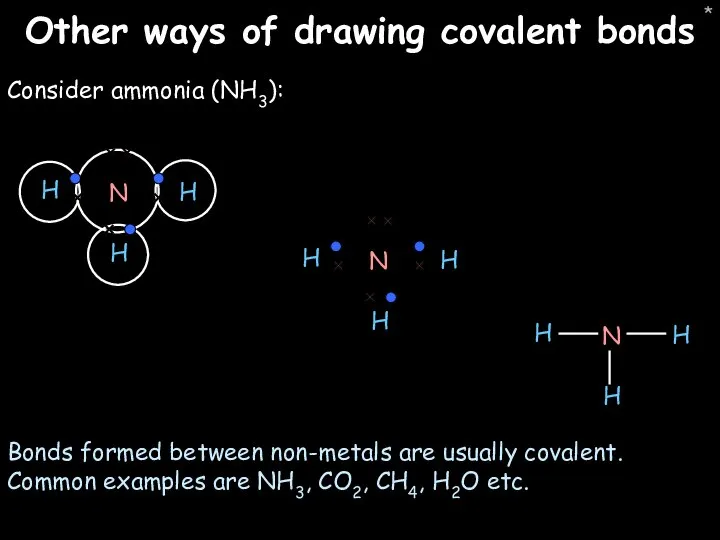
- 47. * Other ways of drawing covalent bonds Consider ammonia (NH3): Bonds formed between non-metals are usually
- 48. * Properties of covalent molecules Recall our model of a simple covalent compound like hydrogen, H2:
- 50. Скачать презентацию























 Stereochemistry.Isomers are different compounds
Stereochemistry.Isomers are different compounds ПЕРВОНАЧАЛЬНЫЕ ХИМИЧЕСКИЕ ПОНЯТИЯ ОБОБЩАЮЩИЙ УРОК
ПЕРВОНАЧАЛЬНЫЕ ХИМИЧЕСКИЕ ПОНЯТИЯ ОБОБЩАЮЩИЙ УРОК  Сера
Сера Металлы. Кроссворд
Металлы. Кроссворд Урок - лекция с контролем в 10 классе
Урок - лекция с контролем в 10 классе  Определение массовой концентрации общего железа с сульфосалициловой кислотой. (Лабораторная работа 2)
Определение массовой концентрации общего железа с сульфосалициловой кислотой. (Лабораторная работа 2) Особенности КИМ ЕГЭ 2015 года по химии Составитель: Снастина М. Г. Москва 2014 г.
Особенности КИМ ЕГЭ 2015 года по химии Составитель: Снастина М. Г. Москва 2014 г.  Презентация Соли
Презентация Соли Вуглеводи
Вуглеводи Понятие о качественных реакциях. 9 класс
Понятие о качественных реакциях. 9 класс Адам мен жануар организміне бордың әсері
Адам мен жануар организміне бордың әсері Жиры. Классификация жиров
Жиры. Классификация жиров Азокрасители. Цвет, классификация
Азокрасители. Цвет, классификация Химические аспекты выбора полимеров-предшественников для получения углеродных волокон
Химические аспекты выбора полимеров-предшественников для получения углеродных волокон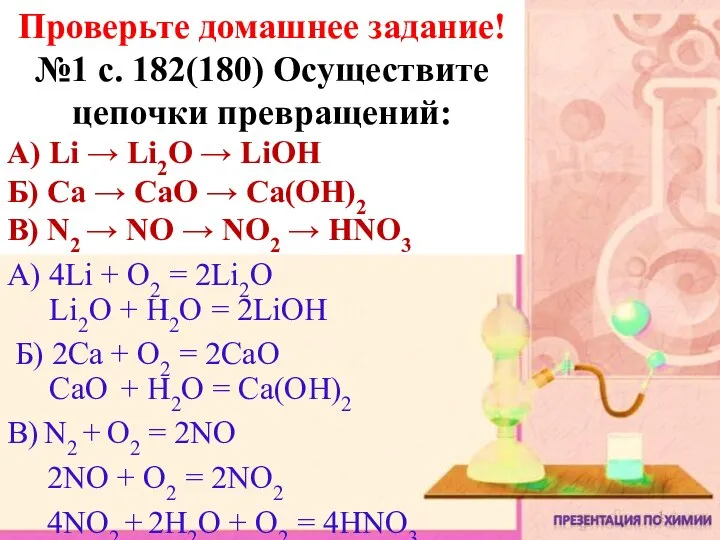 Реакции замещения
Реакции замещения Химическая термодинамика. Лекция 2
Химическая термодинамика. Лекция 2 Химический источник электрического тока
Химический источник электрического тока Выполнила ученица 9б класса Коробатова Алена Учитель: Дьяченко Людмила Александровна
Выполнила ученица 9б класса Коробатова Алена Учитель: Дьяченко Людмила Александровна Фосфор красный, белый и черный
Фосфор красный, белый и черный Общая характеристика элементов IIIA-группы. Бор. Алюминий
Общая характеристика элементов IIIA-группы. Бор. Алюминий Индиго-краситель
Индиго-краситель Сера, ее физические и химические свойства
Сера, ее физические и химические свойства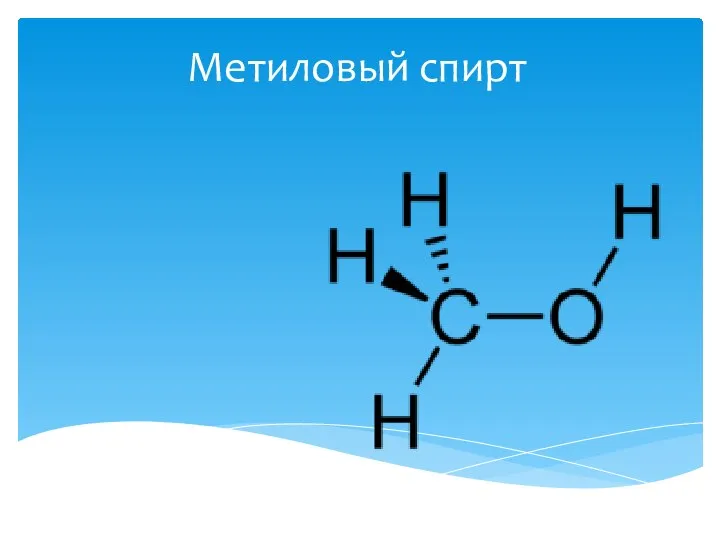 Метиловый спирт
Метиловый спирт Требование к оформлению отчета лабораторной работы
Требование к оформлению отчета лабораторной работы Значение окислительно-восстановительных реакций в жизни человека
Значение окислительно-восстановительных реакций в жизни человека Координационные соединения и их применение в медицине, фармакологии
Координационные соединения и их применение в медицине, фармакологии Нефть Общие сведения
Нефть Общие сведения Сульфаттар және эфирсульфаттар
Сульфаттар және эфирсульфаттар