Содержание
- 2. Chemical kinetics studies the rate and mechanism of chemical processes. v=mole/L∙sec
- 3. Factors affecting the rate of chemical reaction The chemical reaction rate depends on: The nature of
- 4. Factors affecting the rate of chemical reaction The chemical reaction rate depends on: 2. The state
- 5. The dependence of the chemical reaction rate on temperature. J. Van't Hoff formulated a empirical rule:
- 6. THE DEPENDENCE OF THE CHEMICAL REACTION RATE ON TEMPERATURE. Where Vt2 – rate of a chemical
- 7. The mass action law: at a constant temperature chemical reaction rate is proportional to the product
- 8. In case of a chemical reaction occurrence at the interface (gas-liquid, liquid-solid, solid-gas) the surface area
- 9. Kinetic chemical reactions classification according to the molecularity and order of reaction Molecularity of reactions is
- 10. Kinetic classification of chemical reactions Order reaction is defined as the sum of the degrees of
- 11. The rate constant. The rate constant has an expression and a dimension.
- 12. The period of half-transformation In the kinetics the notion of the period of half-transformation t1/2. is
- 13. Methods for determining the order of reaction: The substitution method. The graphical method. Differential method.
- 14. The activation energy. A significant increase of the reaction rate as the temperature increases can be
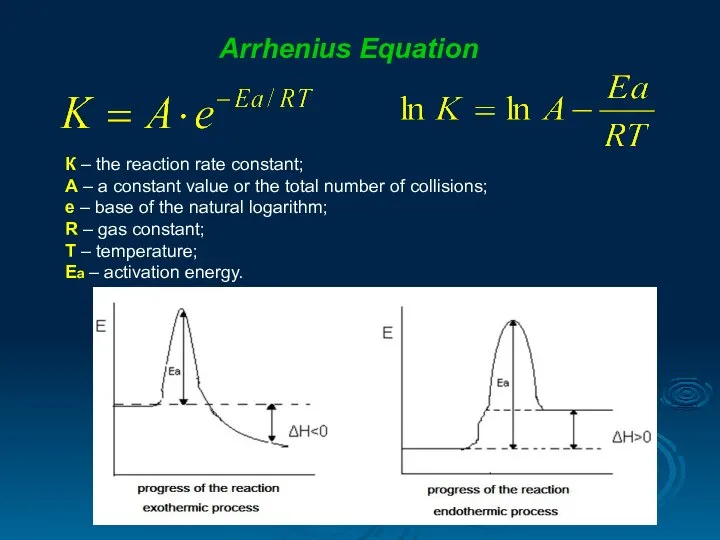
- 15. Arrhenius Equation К – the reaction rate constant; А – a constant value or the total
- 16. Catalytic reactions Catalysis is the process of changing the rate of reaction by catalysts. Reactions taking
- 17. Enzymes Enzymes are protein molecules able to accelerate the course of biochemical reactions. Other than enzymes-proteins
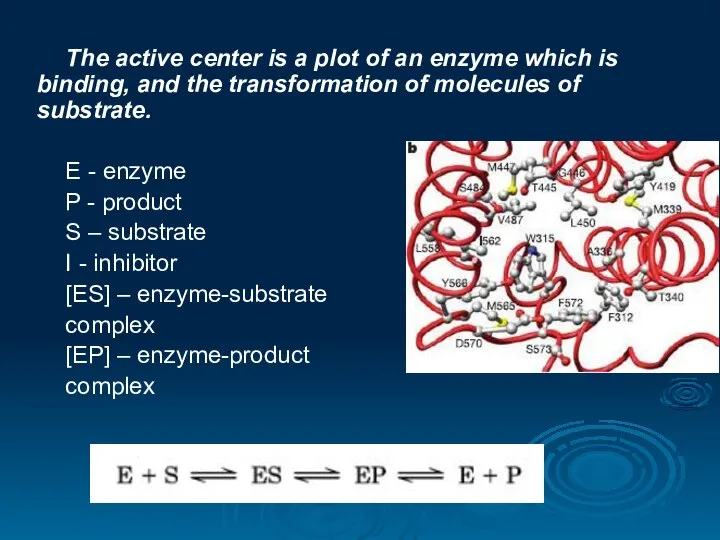
- 18. The active center is a plot of an enzyme which is binding, and the transformation of
- 19. Factors affecting the activity of the enzyme The concentration of the substrate. In 1913 Michaelis and
- 20. Specificity of enzymes: highly specific; law specific; nonspecific. Most of the enzymes are highly specific, since
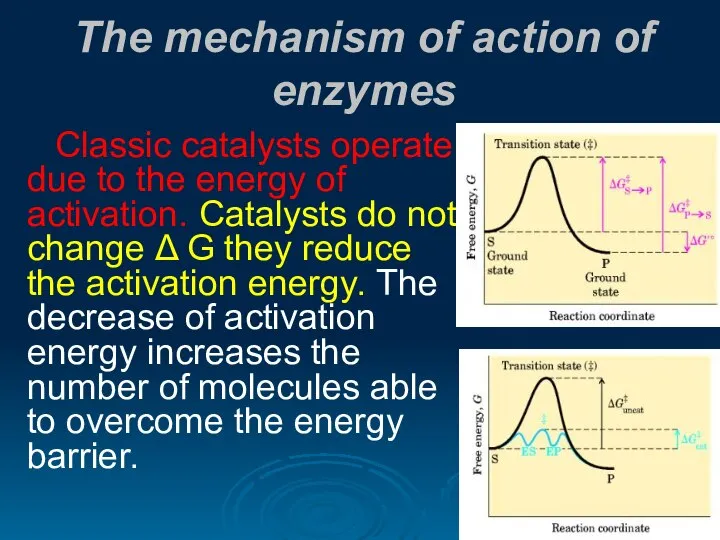
- 21. The mechanism of action of enzymes Classic catalysts operate due to the energy of activation. Catalysts
- 22. The principle of irreversibility of chemical reactions Irreversible reactions lead: to the formation of gaseous substances:
- 23. Reversible chemical reactions. Equilibrium constant. Reactions that proceed in opposite directions are called reversible. V1=K1CH2CI2; V2=K2CHI2
- 24. LE CHATELIER'S PRINCIPLE Shift of the equilibrium based on the principle of Le Chatelier: If the
- 25. LE CHATELIER'S PRINCIPLE 2СО + О2 = 2СО2; ∆Н Equilibrium condition : ∆G =0; and ∆G=∆H-T∆S.
- 26. LE CHATELIER'S PRINCIPLE Acid - base balance of the body is disturbed in violation of the
- 27. The decrease of activation energy is achieved by: 1. Orientation substrates. 2. Theory of steric interactions.
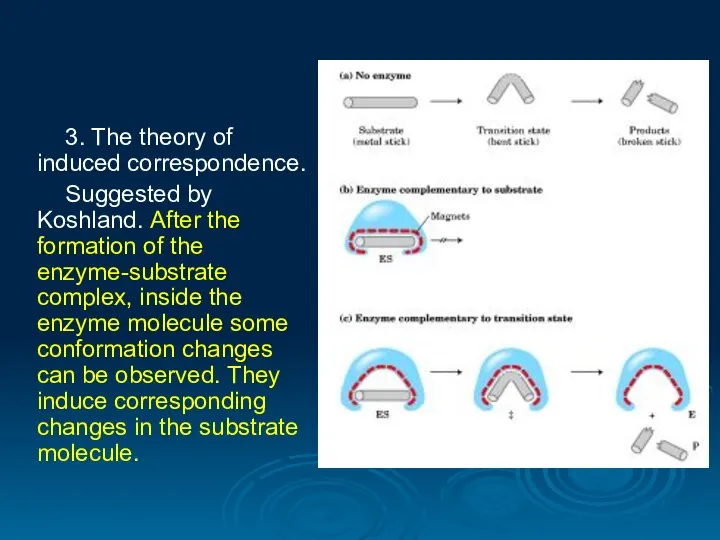
- 28. 3. The theory of induced correspondence. Suggested by Koshland. After the formation of the enzyme-substrate complex,
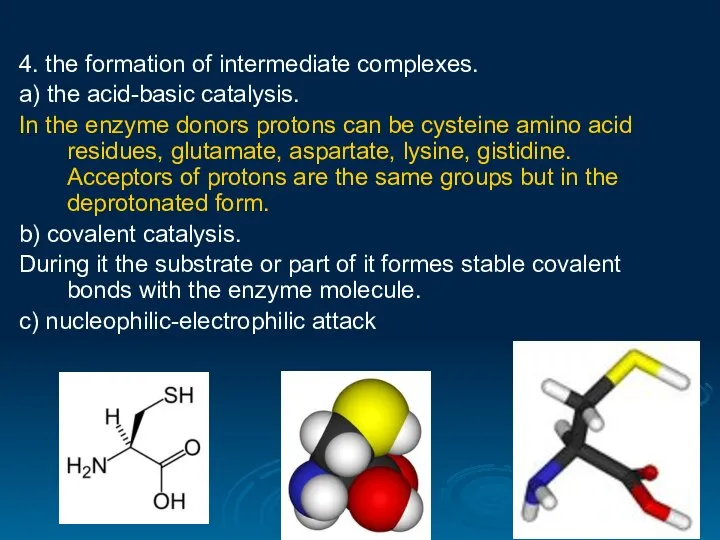
- 29. 4. the formation of intermediate complexes. а) the acid-basic catalysis. In the enzyme donors protons can
- 30. PHOTOCHEMICAL REACTIONS Photochemical reactions occur with the absorption of light energy For example, photosynthesis of the
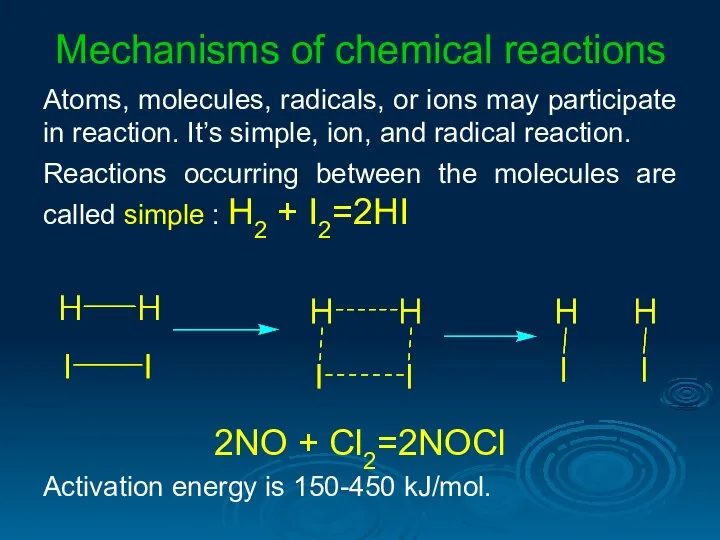
- 31. Mechanisms of chemical reactions Atoms, molecules, radicals, or ions may participate in reaction. It’s simple, ion,
- 33. Скачать презентацию
























 Сложные эфиры. Жиры
Сложные эфиры. Жиры История и методология химии. Лекция 8
История и методология химии. Лекция 8 Презентация по Химии "Презентация Войди в природу другом" - скачать смотреть
Презентация по Химии "Презентация Войди в природу другом" - скачать смотреть  Новогаленовые. Максимально очищенные экстракционные препараты
Новогаленовые. Максимально очищенные экстракционные препараты Полимерные материалы и изделия. (Лекция 16)
Полимерные материалы и изделия. (Лекция 16) Алкены
Алкены Абсорбция, адсорбция, конденсация
Абсорбция, адсорбция, конденсация Жалпы химиялық технология
Жалпы химиялық технология Сплавы серебра
Сплавы серебра Актиноиды. Физические и химические свойства
Актиноиды. Физические и химические свойства Зымыран сұрақтар
Зымыран сұрақтар Алканы
Алканы Спирты в жизни человека
Спирты в жизни человека Изобутан. Получение и применение
Изобутан. Получение и применение Преподаватель: Погребнёв Сергей Сергеевич Лекция № 4. УГЛЕВОДЫ. ЛИПИДЫ.
Преподаватель: Погребнёв Сергей Сергеевич Лекция № 4. УГЛЕВОДЫ. ЛИПИДЫ. Высокомолекулярные соединения (ВМС)
Высокомолекулярные соединения (ВМС) Химико-технологические процессы современных производств. Лекция №3
Химико-технологические процессы современных производств. Лекция №3 Знакомство с химией
Знакомство с химией Закономерности биохимии, токсикологии, изолирования и анализа «металлических» ядов. Первая помощь при отравлении
Закономерности биохимии, токсикологии, изолирования и анализа «металлических» ядов. Первая помощь при отравлении Изучение свойств H2SO4. Практическая работа
Изучение свойств H2SO4. Практическая работа Дисперсные системы
Дисперсные системы Алкалоидтар. Алкалоидтар негіздік қасиет
Алкалоидтар. Алкалоидтар негіздік қасиет Подготовка к контрольной работе
Подготовка к контрольной работе Свойства растворов ВМС
Свойства растворов ВМС Рекомендации по оформлению развёрнутых ответов на задания ЕГЭ
Рекомендации по оформлению развёрнутых ответов на задания ЕГЭ Резеңке. Каучук
Резеңке. Каучук Взаимодействие кислоты с основанием (реакция нейтрализации)
Взаимодействие кислоты с основанием (реакция нейтрализации) Карбоновые кислоты и их функциональные производные
Карбоновые кислоты и их функциональные производные