Содержание
- 2. A. Type of polymerization. 6. 1 Introduction Free-radical polymerization Ionic polymerization Complex coordination polymerization
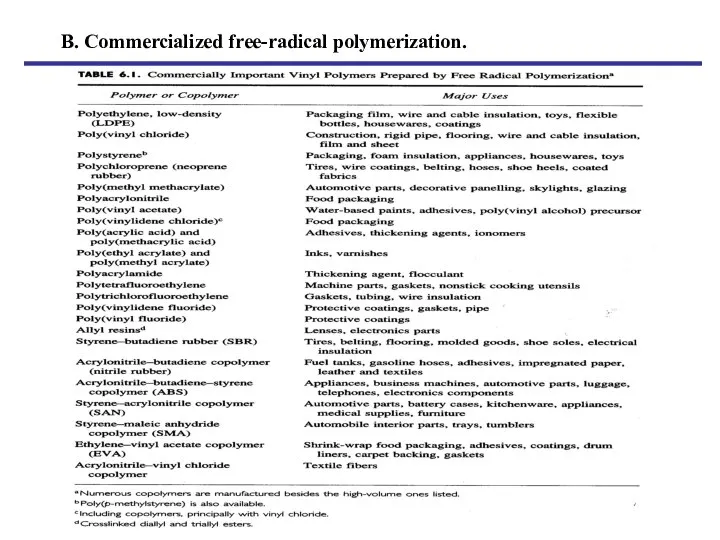
- 3. B. Commercialized free-radical polymerization.
- 4. 6.2 Free Radical Initiators. 6.2.1 Peroxides and Hydroperoxides A. Benzoly peroxide and other peroxides a. Thermal
- 5. d. Other peroxides. Diacetyl peroxide Di-t-butyl peroxide Diacetyl peroxide Di-t-butyl peroxide (half-life:10hours at 120℃) 6.2.1 Peroxides
- 6. e. Promoters : Inducing initiation at lower temperature. (6.9) (6.10) + + + - 6.2.1 Peroxides
- 7. B. Hydroperoxide a. Thermal decomposition hydroperoxide b. Cumyl hydroperoxide. 6.2.1 Peroxides and Hydroperoxides
- 8. A. α,α'-Azobis(isobutyronitrile) (AIBN). a. Decomposition of AIBN. b. Half-life of isobutyronitrile radical : 1.3 hours at
- 9. B. Side reaction : Cage effect. a. Tetramethylsuccinonitrile b. Ketenimine 6.2.2 Azo Compounds.
- 10. A. One electron transfer reaction. a. Making free radical by one electron transfer by redox reaction.
- 11. A. Peroxide and Azo compound. Photolysis and thermalysis. B. Photolabile initiator. 6.2.4 Photoinitiator
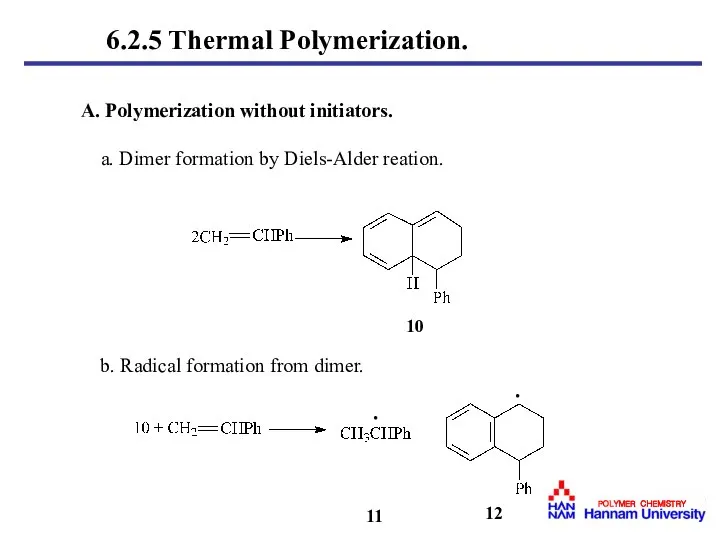
- 12. A. Polymerization without initiators. a. Dimer formation by Diels-Alder reation. 11 12 · b. Radical formation
- 13. 6.2.6 Electrochemical Polymerization. A. Polymerization of electrolysis. a. Cathode reaction : electron transfer to monomer ion
- 14. 6.3 Techniques of Free Radical Polymerization.
- 15. 6.3 Techniques of Free Radical Polymerization. 6.3.1 Bulk A. Reactor charges. a. Monomer. b. Initiator (soluble
- 16. 6.3.2 Suspension. A. Reactor charges. a. Monomer. b. Initiator (soluble in monomer). c. Water or other
- 17. 6.3.3 Solution. A. Reactor charges. a. Monomer (soluble in solvent). b. Initiator (soluble in solvent). c.
- 18. 6.3.4 Emulsion. A. Reactor charges. a. Monomer. b. Redox initiator c. Soap or emulsifier. d. Water.
- 19. 6.3.4 Emulsion. TABLE 6.3. Typical Emulsion Polymerization Recipesa Ingredients, Conditions Ingredients (parts by weight) Water Butadiene
- 20. 6.4 Kinetic and Mechanism of polymerization. A. Mechanism of free-radical polymerization. a. Initiation. 1) Decomposition. Initiator
- 21. b. Propagation. (6.26) 1) Head-to-tail orientation : predominant reaction. Steric and electronic effects. 2) Examples of
- 22. c. Termination. 1) Combination. (6.27) Polystyrene radical. (6.29) 6.4 Kinetic and Mechanism of polymerization.
- 23. 2) Disproportionation. Poly(methyl methacrylate) radical. ① Repulsion of ester group. ② Easy alpha hydrogen abstraction. 3)
- 24. B. Kinetic of free radical polymerization. a. Assumption. 1) The rates of initiation, propagation, and termination
- 25. c. Termination rate ( Rt ) d. Propagation rate ( Rp ) Steady state assumption. kt
- 26. e. Average kinetic chain length ( ) ν B. Kinetic of free radical polymerization.
- 27. f. Gel effect : Trommsdorff effect, Norris-smith effect. 1) Difficult termination reaction because of viscosity. 2)
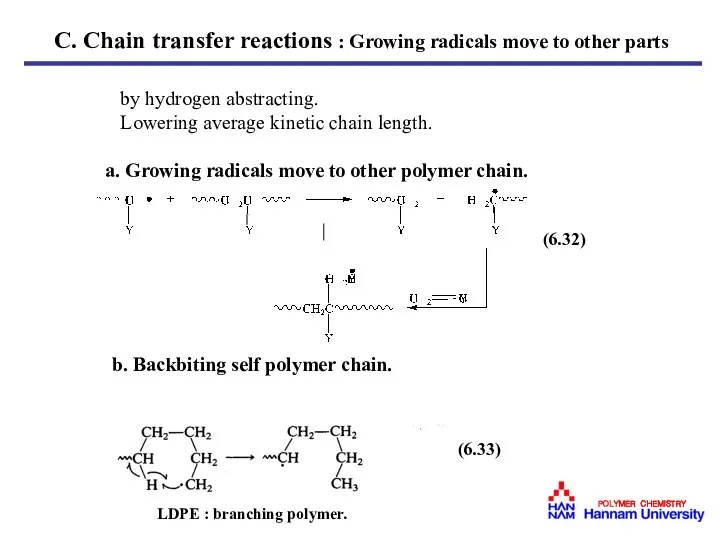
- 28. by hydrogen abstracting. Lowering average kinetic chain length. a. Growing radicals move to other polymer chain.
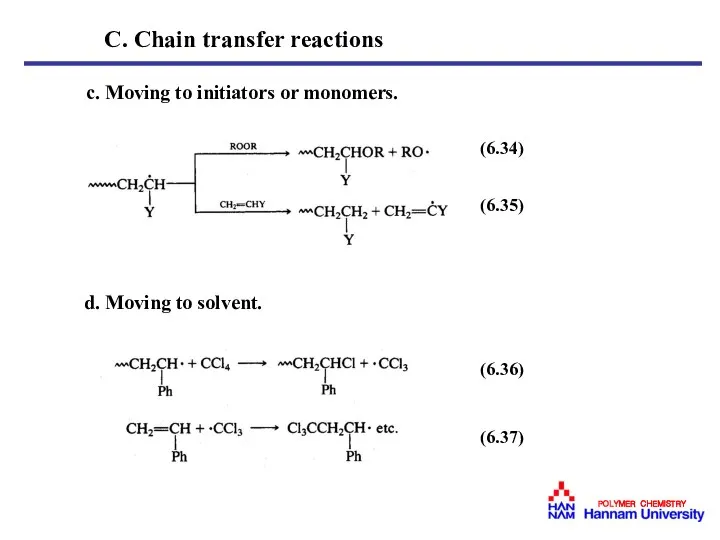
- 29. c. Moving to initiators or monomers. d. Moving to solvent. (6.34) (6.35) (6.36) (6.37) C. Chain
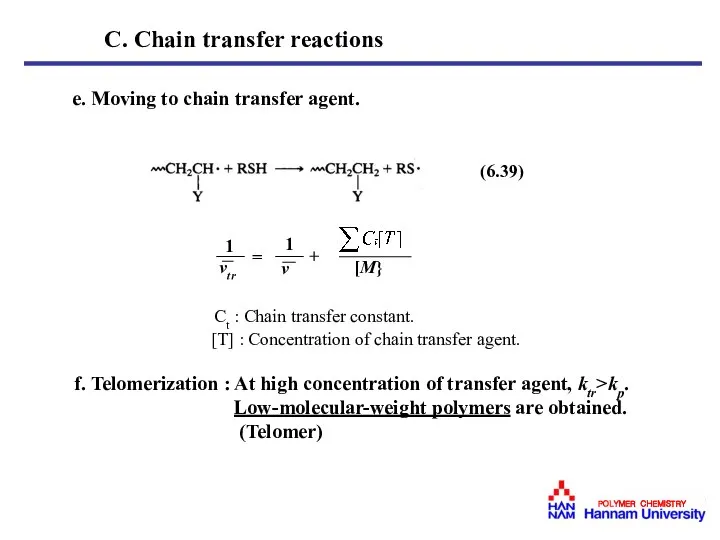
- 30. e. Moving to chain transfer agent. Ct : Chain transfer constant. [T] : Concentration of chain
- 31. a. Copper(I) bypyridyl(bpy) complex: b. TEMPO (18) : 2,2,6,6-tetramethylpiperidinyl-1-oxy. (6.42) (6.43) (6.44) (6.45) D. Leaving free
- 32. c. Synthesis of block copolymers like anionic polymerization. d. Monodisperse polymerization (PI=1.05). E. Kinetics of Emulsion
- 33. 6.5 Stereochemistry of polymerization. A. General consideration. a. Stereoregular polymerization : Ionic and complex coordination polymerization.
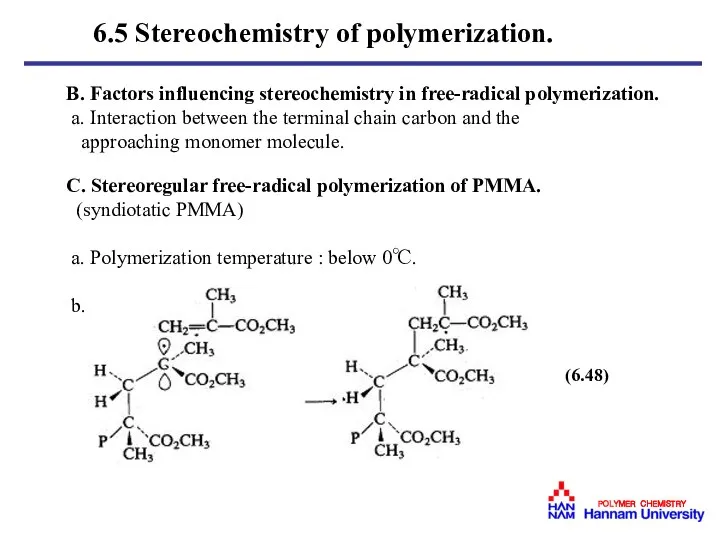
- 34. B. Factors influencing stereochemistry in free-radical polymerization. a. Interaction between the terminal chain carbon and the
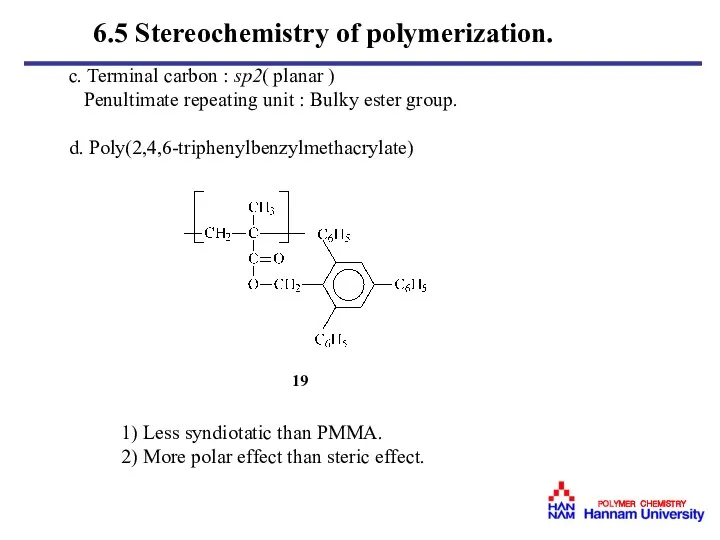
- 35. c. Terminal carbon : sp2( planar ) Penultimate repeating unit : Bulky ester group. d. Poly(2,4,6-triphenylbenzylmethacrylate)
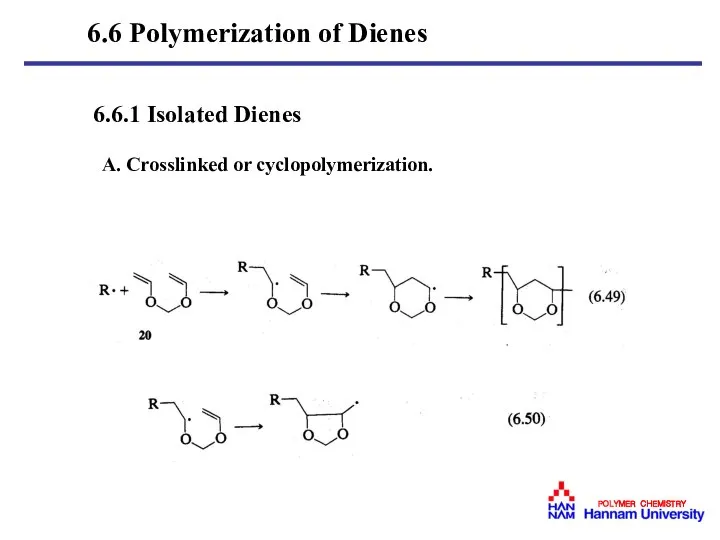
- 36. 6.6.1 Isolated Dienes A. Crosslinked or cyclopolymerization. 6.6 Polymerization of Dienes
- 37. A. Structure of conjugated Diene monoer. Isoprene B. a. 1,2-Addition : Pendent vinyl group. b. Stereochemistry
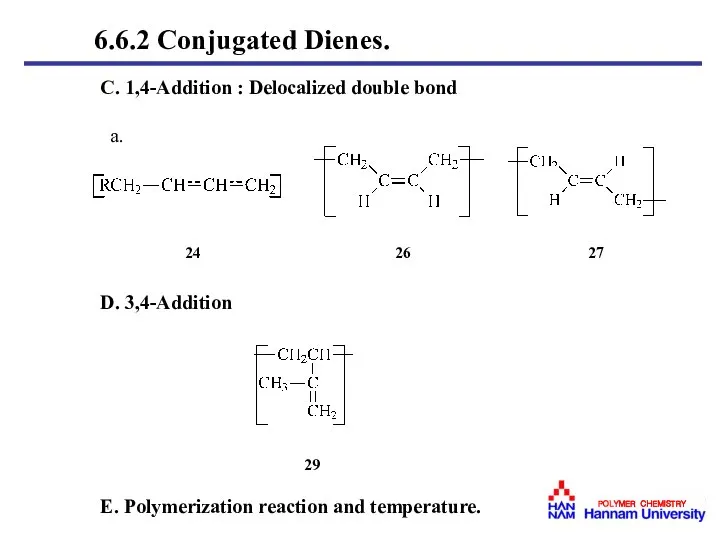
- 38. C. 1,4-Addition : Delocalized double bond a. D. 3,4-Addition E. Polymerization reaction and temperature. 24 26
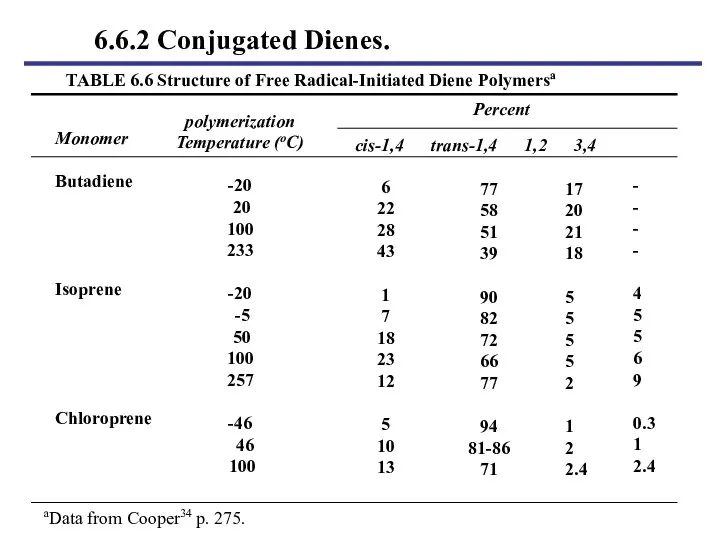
- 39. TABLE 6.6 Structure of Free Radical-Initiated Diene Polymersa polymerization Temperature (oC) -20 20 100 233 -20
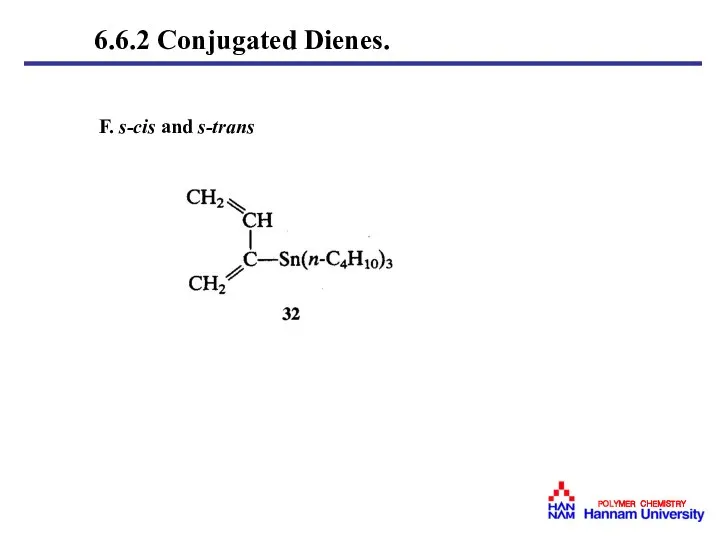
- 40. F. s-cis and s-trans 6.6.2 Conjugated Dienes.
- 41. A. Thermodynamic feasibility. a. ΔGp = ΔHp - TΔSp ΔGp : Gibbs free energy change of
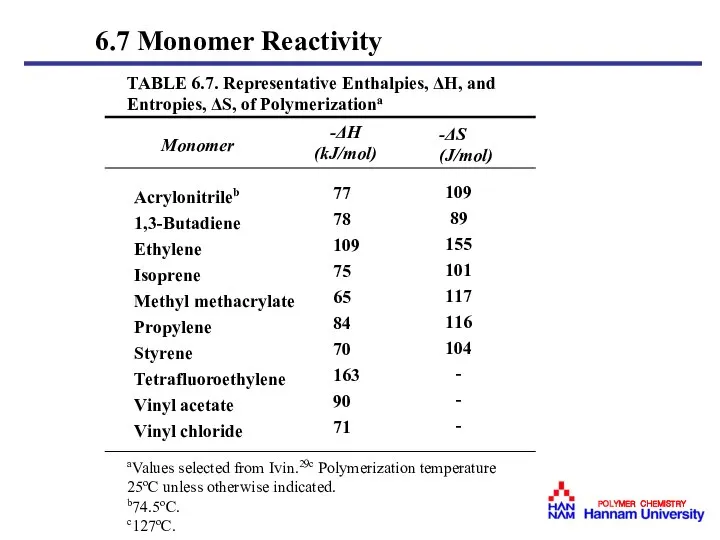
- 42. 6.7 Monomer Reactivity
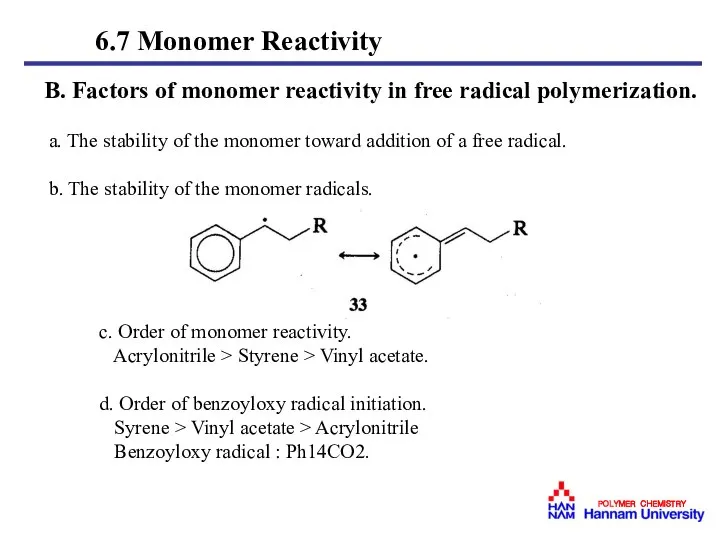
- 43. B. Factors of monomer reactivity in free radical polymerization. a. The stability of the monomer toward
- 44. C. The inverse relationship between monomer stability and polymerization rate. a. Vinyl acetate: not Stable monomer
- 45. D. Ceiling temperature (Tc) a. b. Definition of ceiling temperature. ΔGp = 0 : equal forward
- 46. A. Mechanism of copolymerization. 6.8 Copolymerization.
- 47. a. b. c. let, (reactivity ratio) steady state assumption. d. solving : Copolymer equation or copolymer
- 48. a. r1 = r2 = ∞ : Homopolymer. b. r1 = r2 = 0 : Alternating
- 49. D. Alfrey-price Q-e scheme.
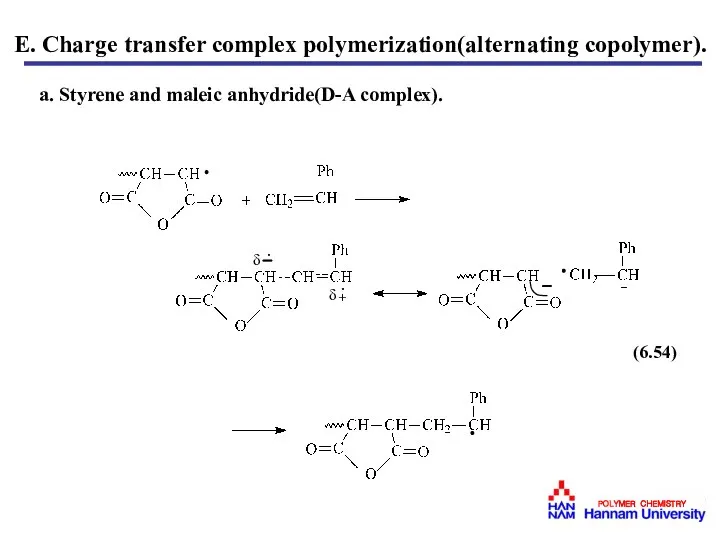
- 50. E. Charge transfer complex polymerization(alternating copolymer). a. Styrene and maleic anhydride(D-A complex).
- 52. Скачать презентацию



































 Адсорбция. Поглощение газов или паров из газовых смесей или растворов твердым веществом
Адсорбция. Поглощение газов или паров из газовых смесей или растворов твердым веществом Полимеры и их использование
Полимеры и их использование Алюминий и его соединения
Алюминий и его соединения Природний газ
Природний газ Процессы проявления и фиксирования
Процессы проявления и фиксирования Алюминий
Алюминий  Йододефицит. Выполнила Кириллова Анастасия.
Йододефицит. Выполнила Кириллова Анастасия.  Физико-химия поверхностных явлений
Физико-химия поверхностных явлений Соединения серы (VI)
Соединения серы (VI)  Когда его называли гением, он морщился: "Какой там гений. Трудился всю жизнь, вот и стал гением". Когда в 26 лет писал курс "
Когда его называли гением, он морщился: "Какой там гений. Трудился всю жизнь, вот и стал гением". Когда в 26 лет писал курс " Планарные потенциометрические сенсоры на основе пилларарена -DNS-162
Планарные потенциометрические сенсоры на основе пилларарена -DNS-162 Исследовательская работа Химические средства в быту
Исследовательская работа Химические средства в быту Тест для подготовки к ЕГЭ по химии 2017
Тест для подготовки к ЕГЭ по химии 2017 Несколько слов о химии
Несколько слов о химии Метод инверсионной вольтамперометрии
Метод инверсионной вольтамперометрии Ферментные препараты
Ферментные препараты Коррозия металлов. Сущность процесса коррозии
Коррозия металлов. Сущность процесса коррозии Марганец. Калий
Марганец. Калий Протолитическая теория кислот и оснований
Протолитическая теория кислот и оснований Белки. Строение и свойства
Белки. Строение и свойства Нафта та продукти її переробки
Нафта та продукти її переробки  Углерод. Вещества, имеющие одинаковое значение степени окисления углерода
Углерод. Вещества, имеющие одинаковое значение степени окисления углерода Презентация по Химии "Современные кондитерские изделия, содержащие красители и консерванты" - скачать смотреть
Презентация по Химии "Современные кондитерские изделия, содержащие красители и консерванты" - скачать смотреть  Физические явления в химии
Физические явления в химии Биологическая роль макро и микроэлементов. (Лекция 17)
Биологическая роль макро и микроэлементов. (Лекция 17) Презентация по Химии "Химия" - скачать смотреть
Презентация по Химии "Химия" - скачать смотреть  Химия в сельском хозяйстве
Химия в сельском хозяйстве Кристаллические решетки
Кристаллические решетки