Содержание
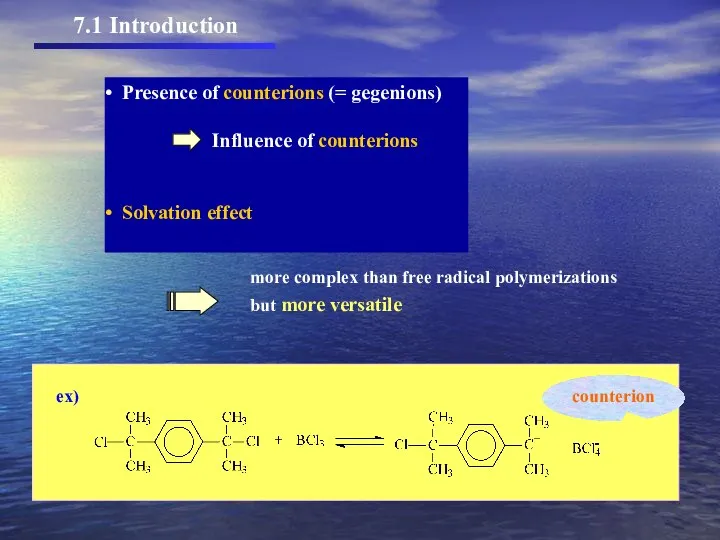
- 2. 7.1 Introduction Presence of counterions (= gegenions) Influence of counterions Solvation effect more complex than free
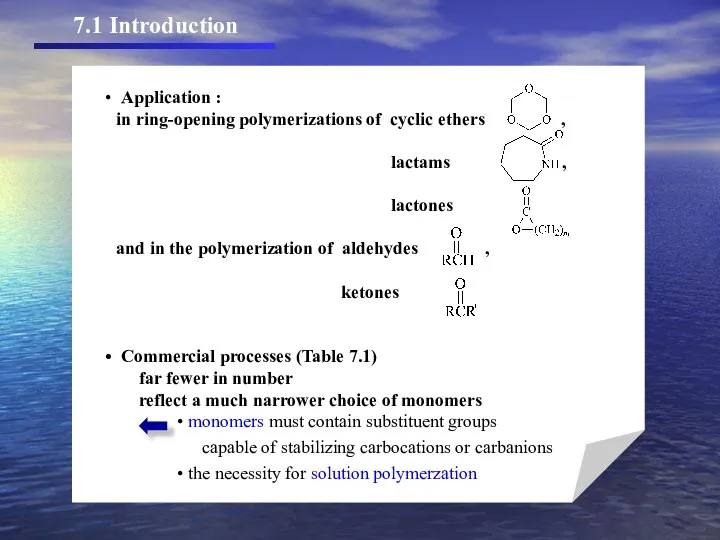
- 3. 7.1 Introduction
- 4. TABLE 7.1. Commercially Important Polymers Prepared by Ionic Polymerization Polymer or Copolymer Cationica Polyisobutylene and polybuteneb
- 5. 7.2.1 Cationic initiators 7.2.2 Mechanism, kinetics, and reactivity in cationic polymerization 7.2.3 Stereochemistry of cationic polymerization
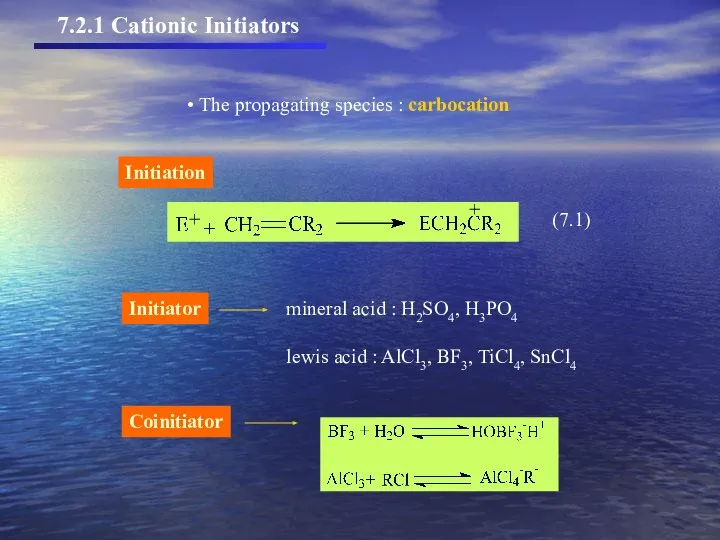
- 6. 7.2.1 Cationic Initiators The propagating species : carbocation Coinitiator
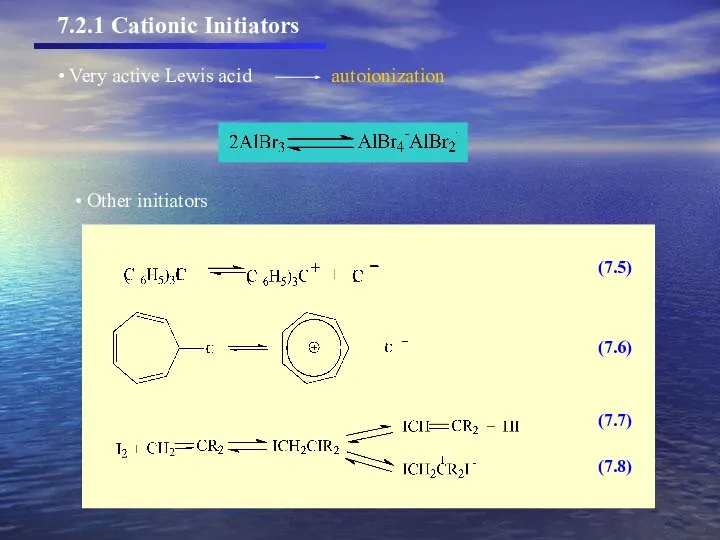
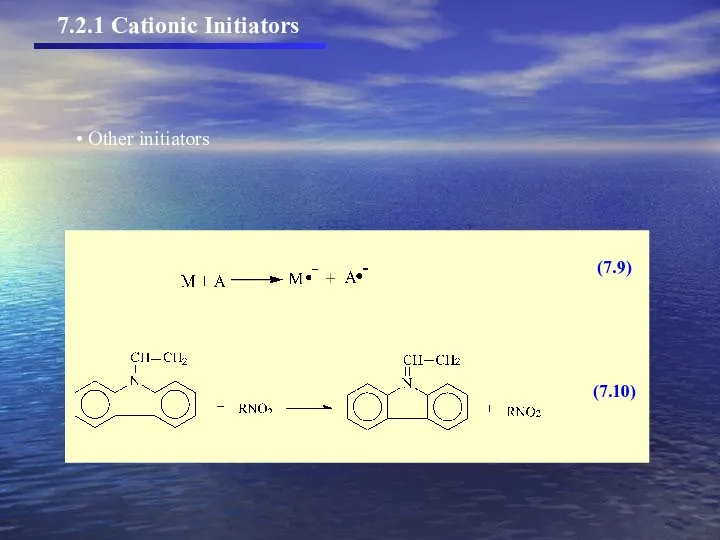
- 7. (7.5) (7.6) (7.7) (7.8) Other initiators 7.2.1 Cationic Initiators
- 8. Other initiators 7.2.1 Cationic Initiators
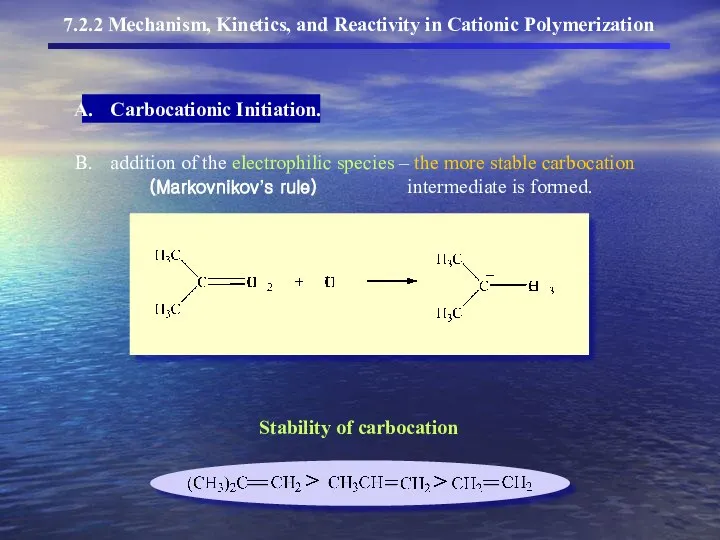
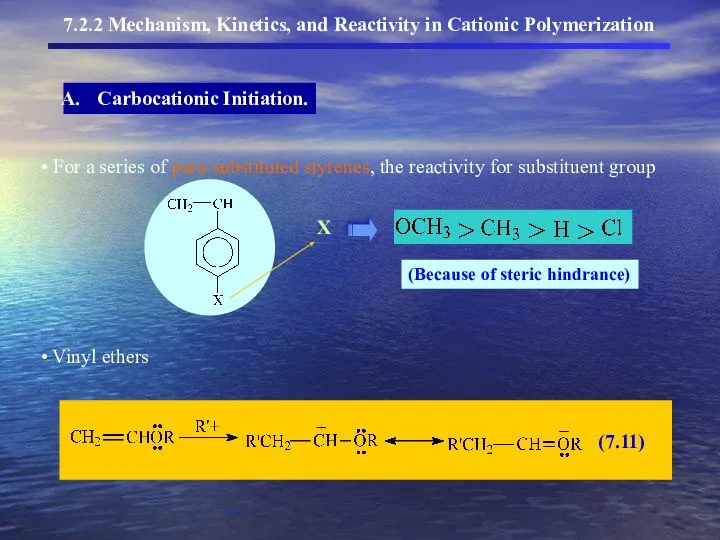
- 9. 7.2.2 Mechanism, Kinetics, and Reactivity in Cationic Polymerization Carbocationic Initiation. addition of the electrophilic species –
- 10. 7.2.2 Mechanism, Kinetics, and Reactivity in Cationic Polymerization Carbocationic Initiation.
- 11. B. Propagation Step 7.2.2 Mechanism, Kinetics, and Reactivity in Cationic Polymerization
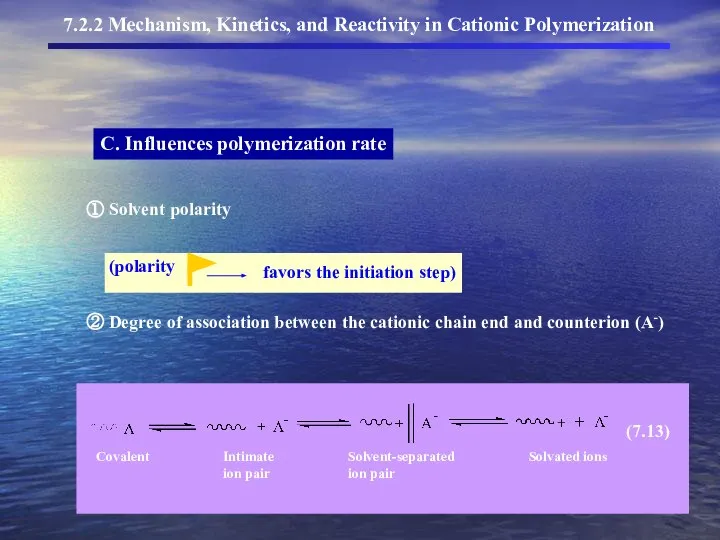
- 12. C. Influences polymerization rate
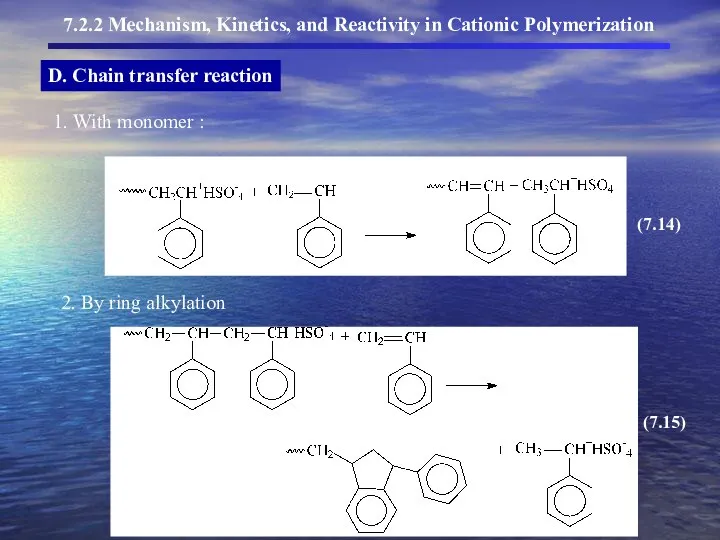
- 13. D. Chain transfer reaction
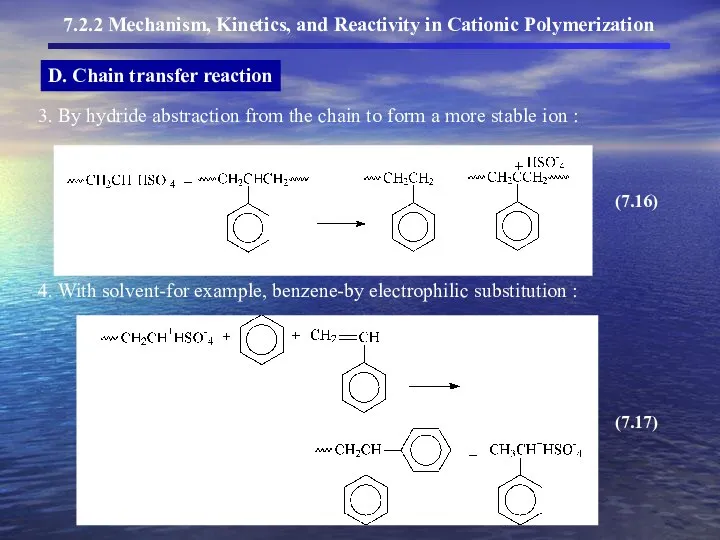
- 14. D. Chain transfer reaction
- 15. E. Termination reaction
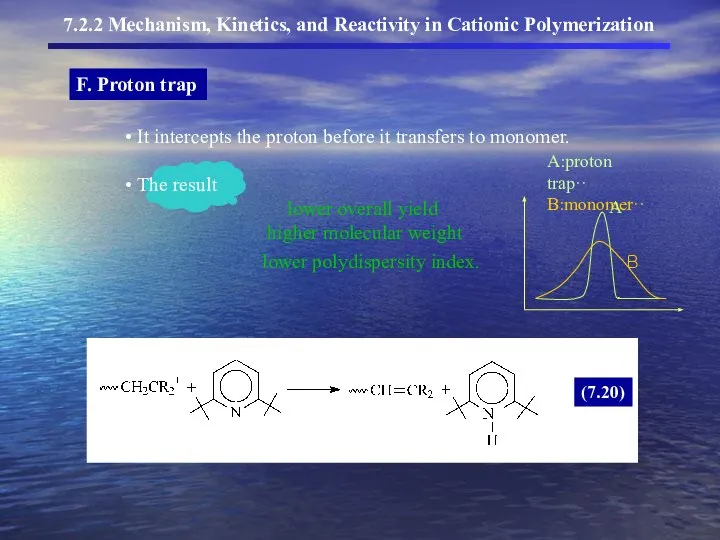
- 16. F. Proton trap
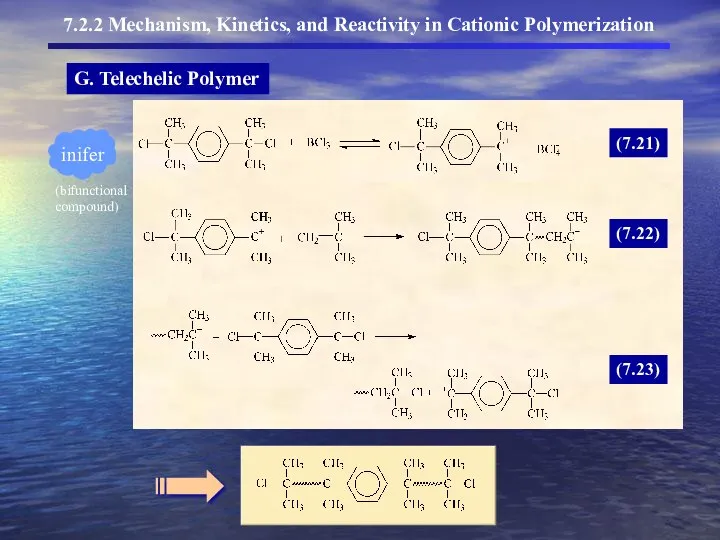
- 17. G. Telechelic Polymer
- 18. H. Pseudocationic Polymerization
- 19. I. To prepare living polymers under cationic conditions.
- 20. I. To prepare living polymers under cationic conditions.
- 21. J. Kinetics
- 23. Substituting for in , one obtains In the absence of any chain transfer, (the kinetic chain
- 24. K. Difference between free radical and cationic processes.
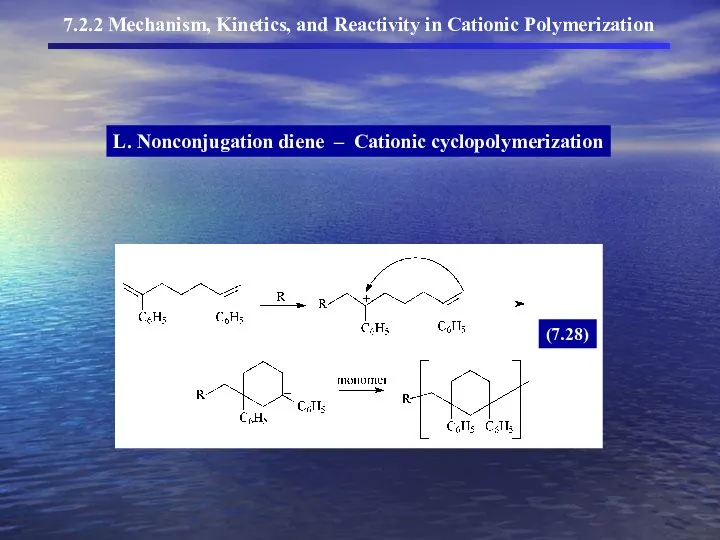
- 25. L. Nonconjugation diene – Cationic cyclopolymerization 7.2.2 Mechanism, Kinetics, and Reactivity in Cationic Polymerization
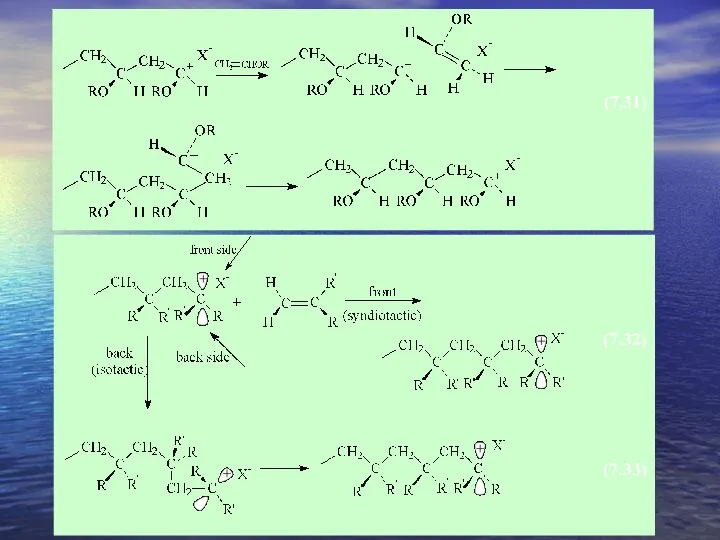
- 26. Cationic Polymerization lead to stereoregular structures. ex) vinyl ether α - methylstyrene Vinyl ether observation resulting
- 27. EX) t-butyl vinyl ether forms isotactic polymer in nonpolar solvents. forms mainly syndiotactic polymer in polar
- 28. In polar solvents both ions 1) be strongly solvated 2) the chain end – exist as
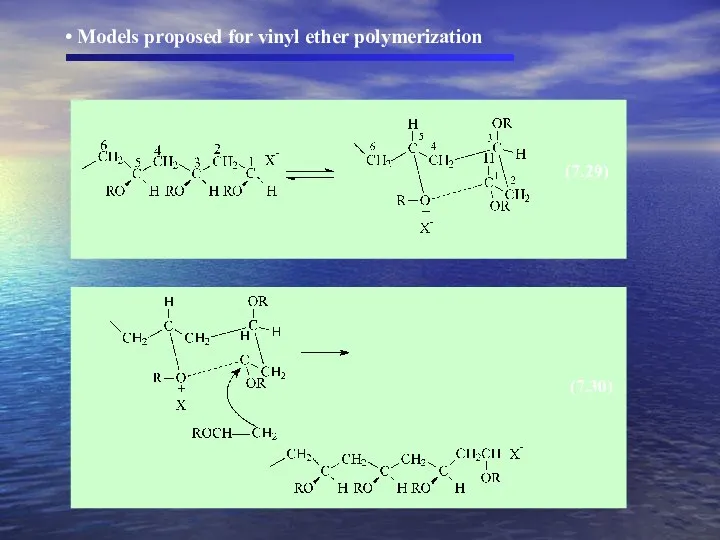
- 29. (7.29) (7.30) Models proposed for vinyl ether polymerization
- 31. 7.2.4 Cationic Copolymerization A. Copolymerization equation - the situation is complication by counterion effects. B. Reactivity
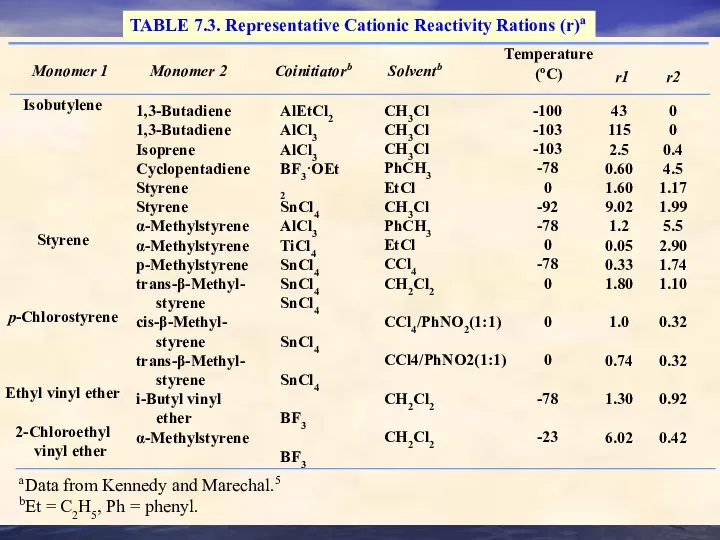
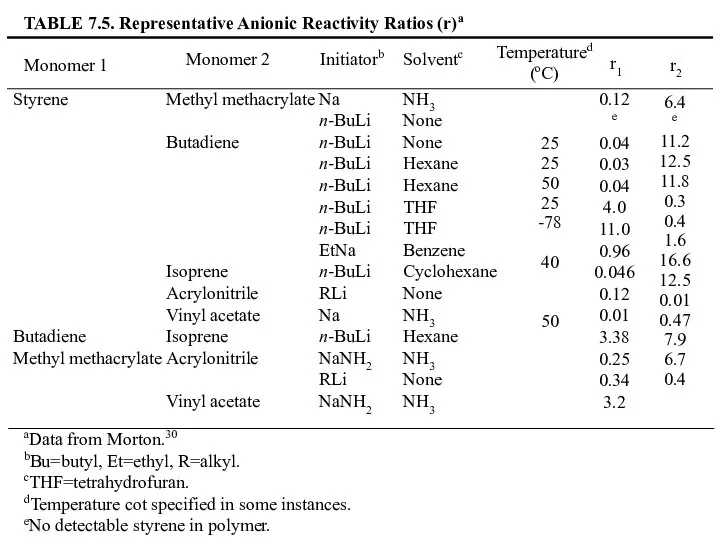
- 32. TABLE 7.3. Representative Cationic Reactivity Rations (r)a Monomer 1 Monomer 2 Coinitiatorb Solventb Temperature (oC) r1
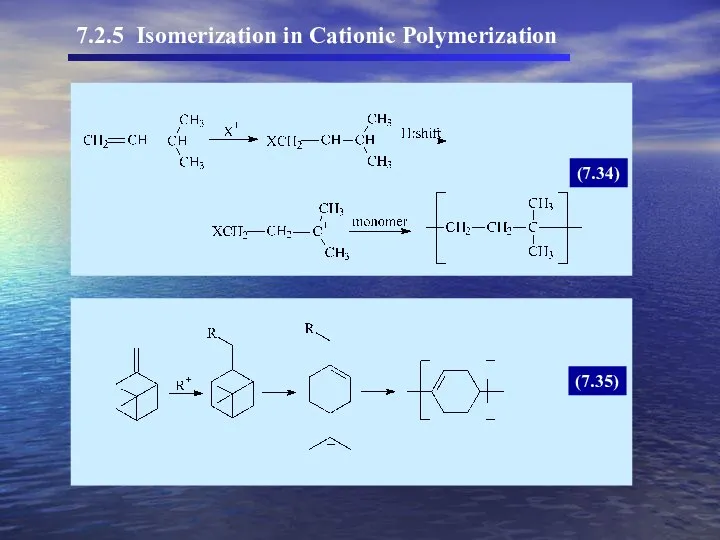
- 33. 7.2.5 Isomerization in Cationic Polymerization (7.34) (7.35)
- 34. 7.3 Anionic Polymerization 7.3.1 Anionic initiators 7.3.2 Mechanism, kinetics, and reactivity in anionic polymerization 7.3.3 Stereochemistry
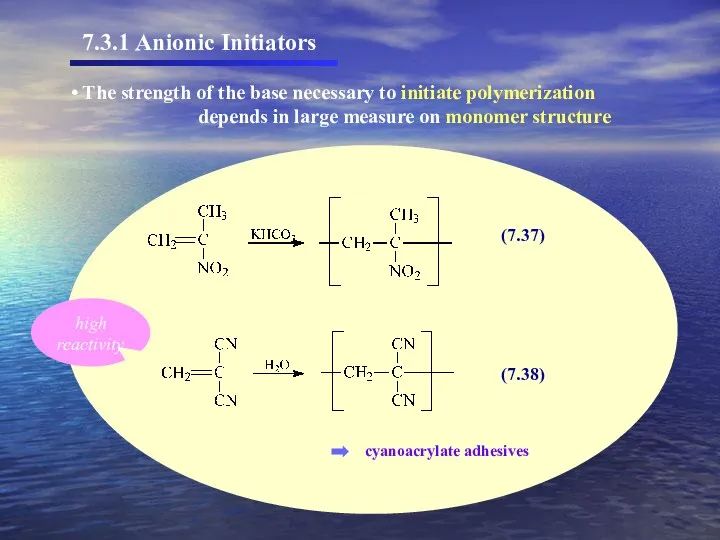
- 35. (7.36) Propagating chain - carbanion Examples – nitro, cyano, carboxyl, vinyl, and phenyl. Monomers having substituent
- 36. The strength of the base necessary to initiate polymerization depends in large measure on monomer structure
- 37. Two basic types that react by addition of a negative ion that undergo electron transfer. ①
- 38. 7.3.1 Anionic Initiators
- 39. 7.3.2 Mechanism, kinetics, and reactivity in anionic polymerization A. Mechanism을 변화시킬 수 있는 요인 a. solvent
- 40. b. Type of cation (counterion) c. Temperature B. The rate of initiation - initiator 와 monomer의
- 41. D. Kinetic 7.3.2 Mechanism, kinetics, and reactivity in anionic polymerization Because the second step is slow
- 42. Substituting in Rp we obtain The average kinetic chain length, is expressed as Assuming a steady
- 43. E. Other types of transfer reactions 7.3.2 Mechanism, kinetics, and reactivity in anionic polymerization
- 44. 7.3.2 Mechanism, kinetics, and reactivity in anionic polymerization
- 45. 7.3.2 Mechanism, kinetics, and reactivity in anionic polymerization G. Important factor in propagation rate. a. Association
- 46. 7.3.2 Mechanism, kinetics, and reactivity in anionic polymerization G. Important factor in propagation rate.
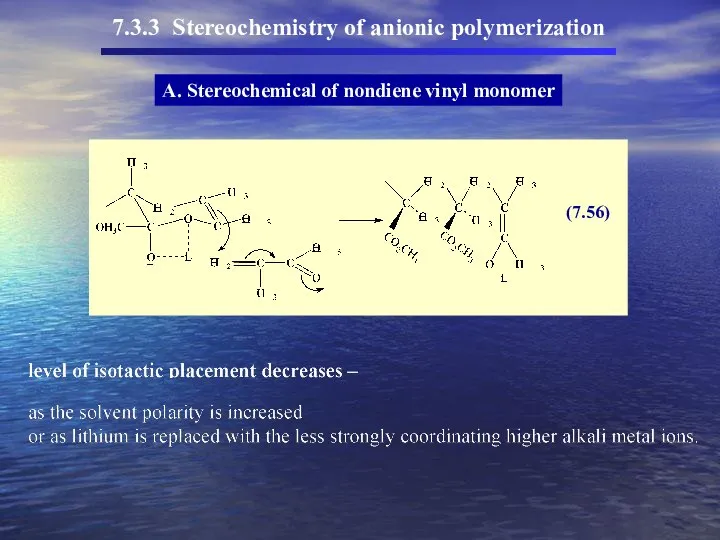
- 47. 7.3.3 Stereochemistry of anionic polymerization A. Stereochemical of nondiene vinyl monomer With soluble anionic initiators (homogeneous
- 48. 7.3.3 Stereochemistry of anionic polymerization A. Stereochemical of nondiene vinyl monomer
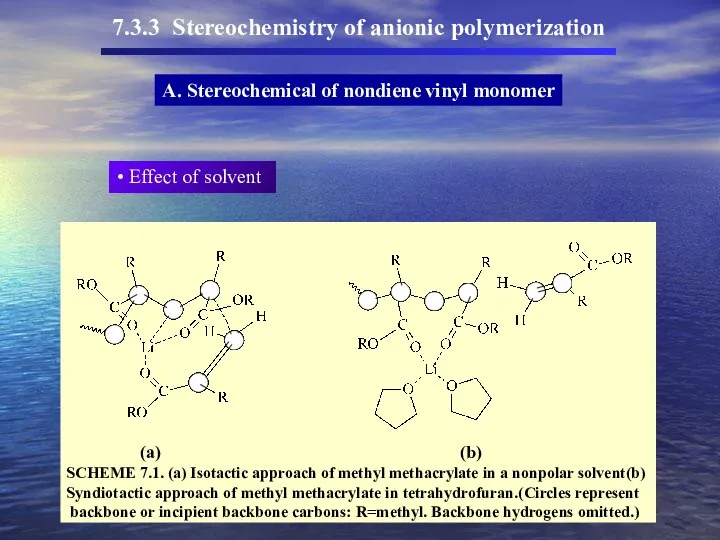
- 49. 7.3.3 Stereochemistry of anionic polymerization A. Stereochemical of nondiene vinyl monomer Effect of solvent
- 50. B. Stereochemical of Dienes 7.3.3 Stereochemistry of anionic polymerization catalyst, solvent의 영향 isoprene 1,3-butadiene Li-based initiator/nonpolar
- 51. formation of cis-polyisoprene – lithium’s ability forming a six-membered ring transition state – “lock” the isoprene
- 52. 7.3.4 Anionic Copolymerization ④ contrasts between homogeneous and heterogeneous polymerization systems.
- 54. 7.3.4 Anionic Copolymerization formation of block copolymers by the living polymer method.
- 55. ABA triblock polymers – Greatest commercial success ex) styrene-butadiene-styrene star-block (radial) – much lower melt viscosities,
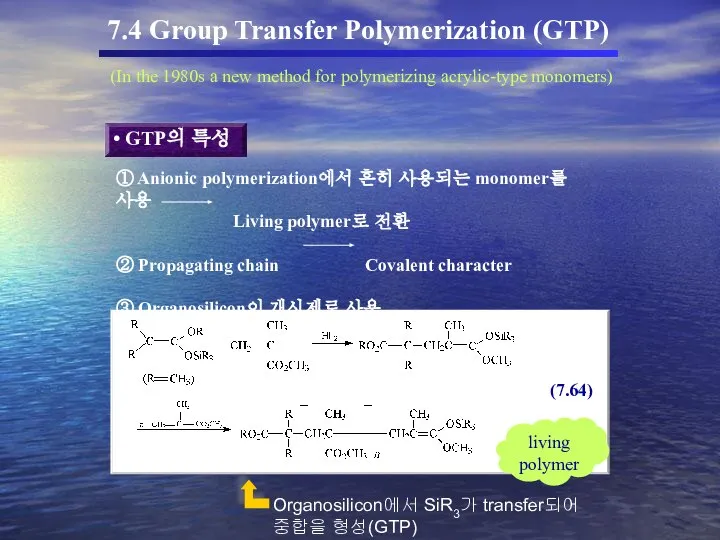
- 56. 7.4 Group Transfer Polymerization (GTP) (In the 1980s a new method for polymerizing acrylic-type monomers) GTP의
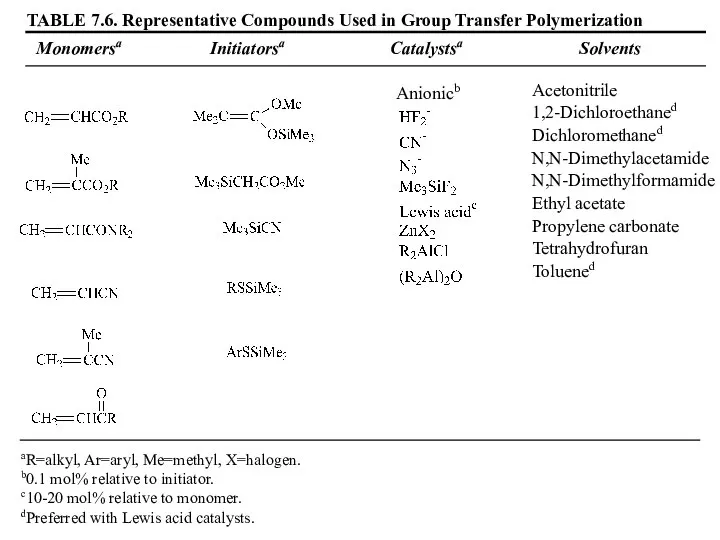
- 58. 7.4 Group Transfer Polymerization (GTP) 두 개의 작용기를 갖는 개시제 사용 사슬의 양끝에서 성장
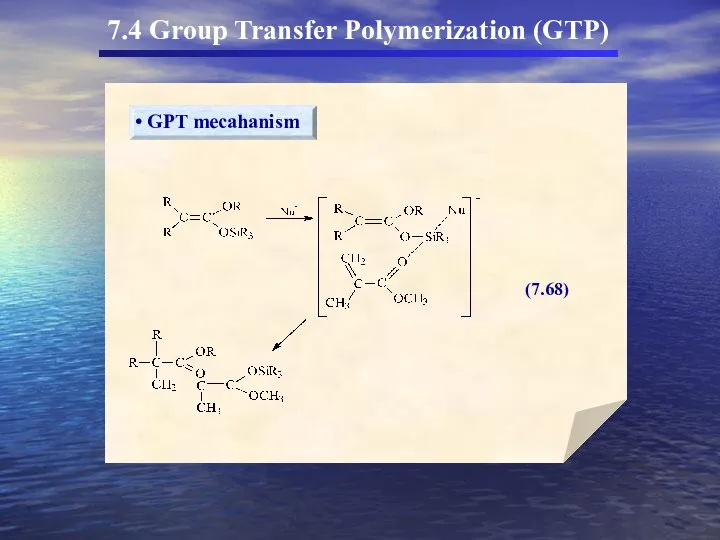
- 59. 7.4 Group Transfer Polymerization (GTP)
- 60. 7.4 Group Transfer Polymerization (GTP)
- 62. Скачать презентацию



































 Свойства белков
Свойства белков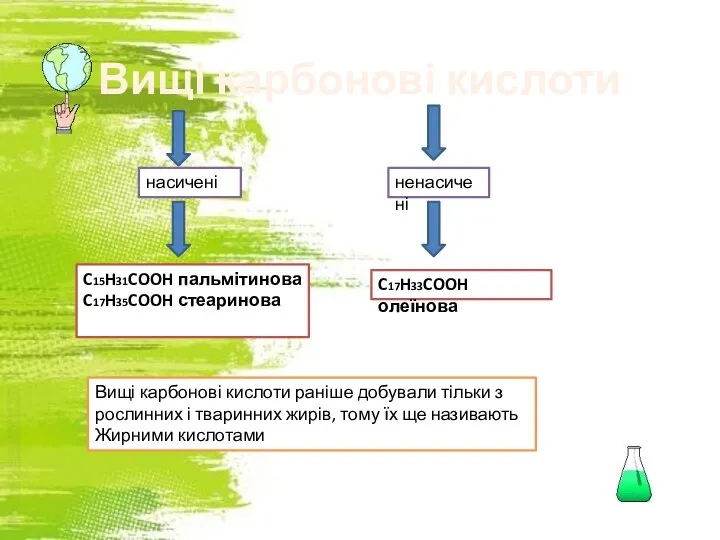 Вищі карбонові кислоти C17H33COOH олеїнова
Вищі карбонові кислоти C17H33COOH олеїнова  Альдегиды и кетоны
Альдегиды и кетоны Литология. Кремнистые породы
Литология. Кремнистые породы Харчові добавки Виконала: Учениця 10-Б класу Єфіменко Злата
Харчові добавки Виконала: Учениця 10-Б класу Єфіменко Злата  Аналитическая химия
Аналитическая химия ИОННАЯ СВЯЗЬ. ВЕЩЕСТВА ИОННОГО НЕМОЛЕКУЛЯРНОГО) СТРОЕНИЯ.
ИОННАЯ СВЯЗЬ. ВЕЩЕСТВА ИОННОГО НЕМОЛЕКУЛЯРНОГО) СТРОЕНИЯ. Минерал гранат
Минерал гранат Презентация по Химии "ЭЛЕКТРОЛИТИЧЕСКАЯ ДИССОЦИАЦИЯ" - скачать смотреть бесплатно
Презентация по Химии "ЭЛЕКТРОЛИТИЧЕСКАЯ ДИССОЦИАЦИЯ" - скачать смотреть бесплатно Современное состояние и пути совершенствования стандартизации лекарственных средств
Современное состояние и пути совершенствования стандартизации лекарственных средств Презентация по Химии "Вода как растворитель" - скачать смотреть
Презентация по Химии "Вода как растворитель" - скачать смотреть  Элементы V группы главной подгруппы
Элементы V группы главной подгруппы Химия и сельское хозяйство
Химия и сельское хозяйство Псевдогалогены и их соединения
Псевдогалогены и их соединения Белки. Классификация белков. (Тема 2)
Белки. Классификация белков. (Тема 2) Основы химической коррозии
Основы химической коррозии Показатели КЩС крови
Показатели КЩС крови Ֆլավանոիդներ պարունակող դեղաբույսեր եվ հումք
Ֆլավանոիդներ պարունակող դեղաբույսեր եվ հումք Химическая промышленность
Химическая промышленность Всасывание продуктов гидролиза липидов в тонком кишечнике. Ресинтез жиров. Образование смешанных мицелл
Всасывание продуктов гидролиза липидов в тонком кишечнике. Ресинтез жиров. Образование смешанных мицелл Инструментальные методы исследования клеточных метаболитов и макромолекул
Инструментальные методы исследования клеточных метаболитов и макромолекул Александр Михайлович Бутлеров
Александр Михайлович Бутлеров Химические свойства алкинов
Химические свойства алкинов Конкурс интерактивных презентаций «Интерактивная мозаика» автор: Кудрявцева Ирина Александровна МОУ «Средняя общеобразова
Конкурс интерактивных презентаций «Интерактивная мозаика» автор: Кудрявцева Ирина Александровна МОУ «Средняя общеобразова Переосаждение. (Глава 4. § 20. Задача 12)
Переосаждение. (Глава 4. § 20. Задача 12) Соединения галогенов
Соединения галогенов Подача питательной воды в парогенератор при запроектной аварии
Подача питательной воды в парогенератор при запроектной аварии Хромопротеиды. Патология пигментного обмена. Взаимосвязь обменов
Хромопротеиды. Патология пигментного обмена. Взаимосвязь обменов