Содержание
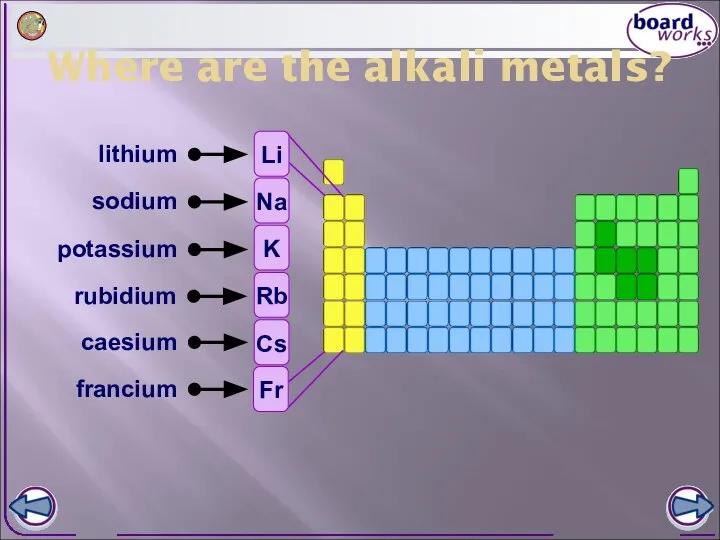
- 2. Where are the alkali metals? lithium sodium potassium rubidium caesium francium
- 3. Why are they called the ‘alkali metals’? The alkali metals are so reactive that, as elements,
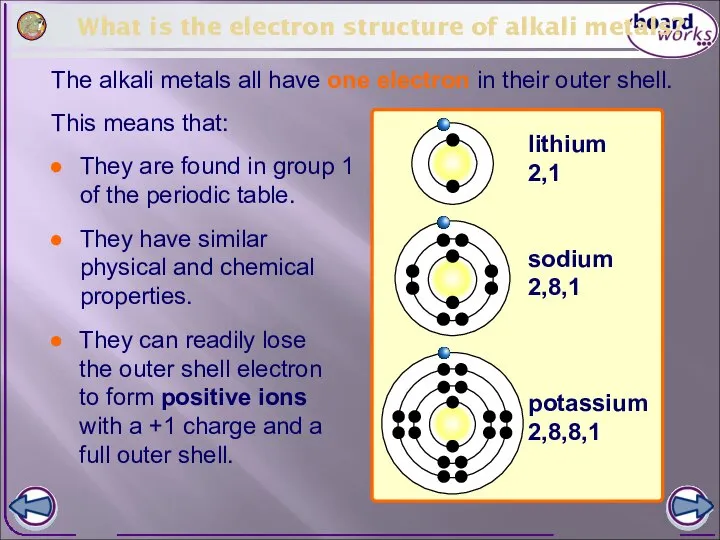
- 4. What is the electron structure of alkali metals? The alkali metals all have one electron in
- 5. What are the properties of the alkali metals? The characteristic properties of the alkali metals are:
- 6. What is the trend in density? The alkali metals generally become more dense going down the
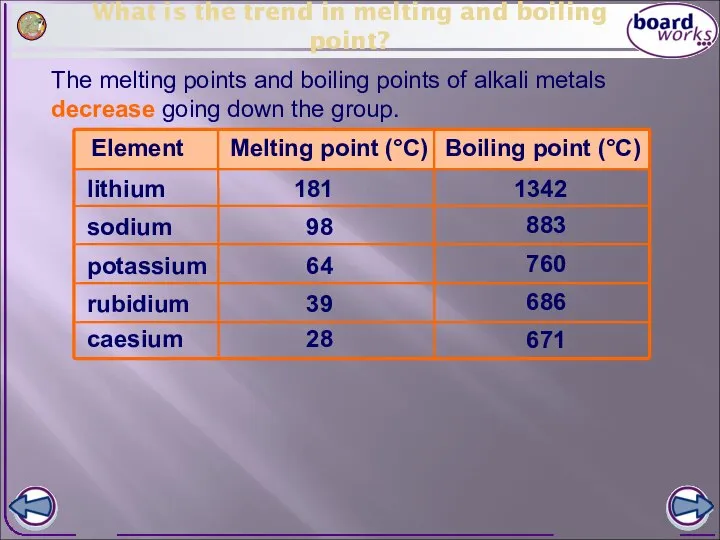
- 7. What is the trend in melting and boiling point? The melting points and boiling points of
- 8. How do the alkali metals react with oxygen? All alkali metals react with oxygen in the
- 9. What are the word and chemical equations for the reaction that causes sodium to tarnish? What
- 10. How do the alkali metals react with water?
- 11. All the alkali metals react vigorously with water. What does the reaction with water produce? It
- 12. This reaction creates alkaline hydroxide ions. The general equation for the reaction between an alkali metal
- 13. How does lithium react with water? Lithium is the least reactive of the alkali metals. When
- 14. How does sodium react with water? When added to water, sodium fizzes more than lithium, and
- 15. How does potassium react with water? When added to water, the potassium moves across the surface
- 16. How do alkali metals react with water? Which of the alkali metals will react most strongly
- 17. How does electron structure affect reactivity? The reactivity of alkali metals increases going down the group.
- 18. Alkali metals burst into flames when heated and added to chlorine. They form metal chlorides. How
- 20. Скачать презентацию














 Аварії з викидом хімічних небезпечних речовин та їх класифікація
Аварії з викидом хімічних небезпечних речовин та їх класифікація Бескислородные керамические материалы
Бескислородные керамические материалы Основания. Определение. Номенклатура
Основания. Определение. Номенклатура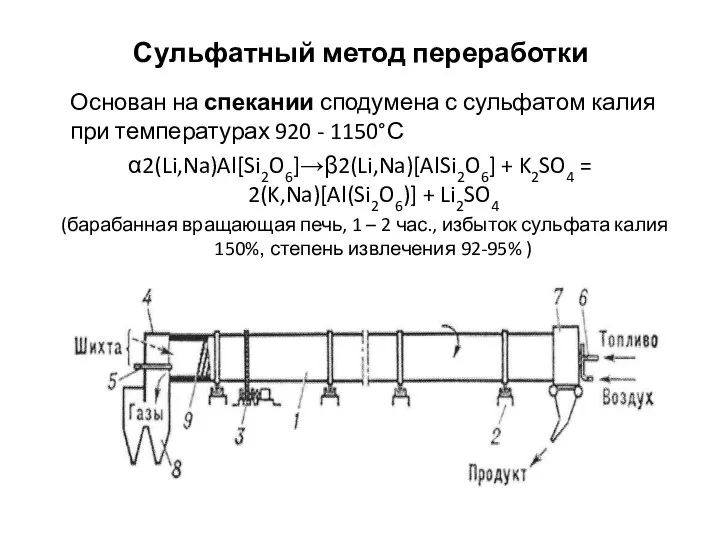 Сульфатный метод переработки
Сульфатный метод переработки Азот
Азот Кадмий (Cadmium)
Кадмий (Cadmium) Терпены и стероиды
Терпены и стероиды Гетерогенное ионное равновесие. Реакции обмена
Гетерогенное ионное равновесие. Реакции обмена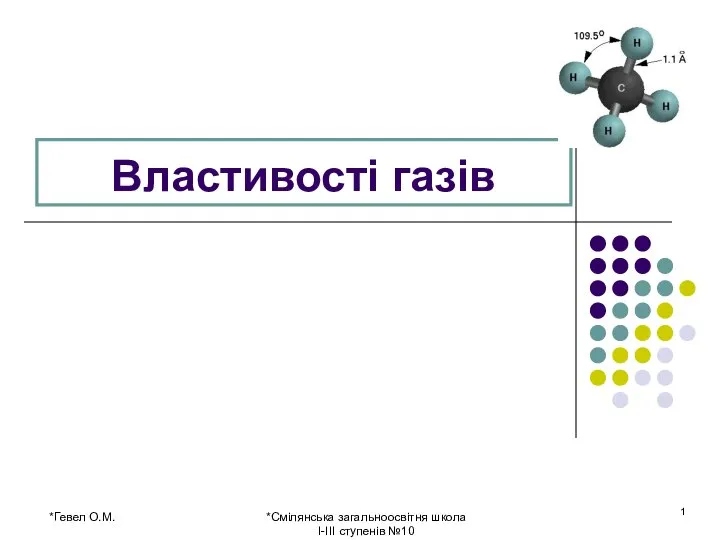 Властивості газів
Властивості газів Растворы. Процесс растворения
Растворы. Процесс растворения Стеклянные товары. Основные сырьевые материалы для производства стекла
Стеклянные товары. Основные сырьевые материалы для производства стекла Химико-технологические процессы современных производств. Лекция №3
Химико-технологические процессы современных производств. Лекция №3 Растворы. Первичные понятия
Растворы. Первичные понятия Констукционные и функциональные волокнистые композиты
Констукционные и функциональные волокнистые композиты Исследовательский проект. Коллаген
Исследовательский проект. Коллаген Бордос сұйықтығын дайындау және оның сапасын тексеру
Бордос сұйықтығын дайындау және оның сапасын тексеру Презентация по Химии "Фосфор" - скачать смотреть бесплатно_
Презентация по Химии "Фосфор" - скачать смотреть бесплатно_ Установка пиролиза широкой фракции, легких углеводородов
Установка пиролиза широкой фракции, легких углеводородов Коллоидная химия
Коллоидная химия Природные соединения кремния
Природные соединения кремния Строение и свойства комплексных соединений
Строение и свойства комплексных соединений Превращения в нашей жизни: физические и химические
Превращения в нашей жизни: физические и химические Evolution of Isoconversional Methods
Evolution of Isoconversional Methods Металлы
Металлы «Предмет органической химии. Особенности органических веществ». По учебнику О. С. Габриеляна. 9 класс
«Предмет органической химии. Особенности органических веществ». По учебнику О. С. Габриеляна. 9 класс Чугуны. Структура и свойства. Классификация и применение
Чугуны. Структура и свойства. Классификация и применение Гидрокаталитические процессы
Гидрокаталитические процессы Підготувала: Учениця 10-Б класу Іщенко Інна
Підготувала: Учениця 10-Б класу Іщенко Інна