Содержание
- 2. Chemical changes The formation of new substances takes place with different chemical properties is called chemical
- 4. Different types of chemical reaction 30-09-2015
- 5. In a chemical reaction, the substances which react together are called reactants whereas the new substances
- 6. Different types of chemical reaction Combination reactions Decomposition reactions Displacement reactions Double-displacement reactions Oxidation-reduction reactions Precipitation
- 7. What is combination reaction? A reaction in which two or more substances (elements or compounds) combine
- 8. Examples: H2(g) + Cl2(g) 2HCl(l) Mg(s) + Cl2(g) MgCl2(s) Fe(s) + S(s) FeS (s) MgO(s) +
- 9. Synthesis Reaction: The combination reaction in which a compound is formed from its constituent elements is
- 10. What is decomposition reaction? A reaction in which a substance is broken down into two or
- 11. Various types of decomposition reactions Thermal decomposition reaction Decomposition caused by heating Electrolytic decomposition (electrolysis) reaction
- 12. Examples: ZnCO3(g) ZnO(s) + CO2(g) 2H2O (l) 2H2(g) + O2(g) CaCO3(s) CaO (s) + CO2 (g)
- 13. What is displacement reaction? A reaction in which one part (an atom or a group of
- 14. Examples: Zn(s) + 2HCl(dil) ZnCl2(aq) + H2(g) 2KBr(aq) + Cl2(aq) 2KCl(aq) + Br2(aq) CuSO4(aq) + Zn(s)
- 15. What is double-displacement reaction? A reaction in which the two reacting ionic compounds exchange their corresponding
- 16. Examples: AgNO3(aq) + NaCl(aq) AgCl(s) + NaNO3(aq) Ni(NO3)2 (aq) + 2NaOH (aq) Ni(OH)2(s) + NaNO3(aq) 2KBr(aq)
- 17. Oxidation-Reduction Reaction What do you mean by oxidation-reduction reaction? Oxidation reaction: any process involving addition of
- 18. Oxidising agent : The substance which brings about oxidation of other substances is called an oxidising
- 19. Reducing agent: The substance which brings about reduction of other substance is called a reducing agent.
- 20. Different types of chemical reaction 30-09-2015
- 21. What is precipitation reaction? The reaction in which one of the products formed is an insoluble
- 22. Examples: AgNO3(aq) + KCl(aq) AgCl(s) + KNO3(aq) Pb(NO3)2 (aq) + 2KI(aq) PbI2(s) + 2KNO3(aq) BaCl2(aq) +
- 23. What do you mean by exothermic and endothermic reaction? Reaction which is accompanied by evolution of
- 24. Example: H2O(s) + heat H2O(l) (endothermic) C(s) + H2O(v) + heat CO(g) + H2(g) (endothermic) CH4(g)
- 26. Скачать презентацию























 Структурно-механiчнi (реологiчнi) властивостi харчових мас
Структурно-механiчнi (реологiчнi) властивостi харчових мас Миметик глицина на основе производного Гераниола
Миметик глицина на основе производного Гераниола Noţiuni generale despre enzime
Noţiuni generale despre enzime Вредные вещества
Вредные вещества Алканы и алкены
Алканы и алкены Фосфорні добрива. Технології збагачення фосфоровмісної сировини
Фосфорні добрива. Технології збагачення фосфоровмісної сировини Растворы неэлектролитов. Закон Вант- Гоффа и Рауля. Растворы электролитов. Теория электронной диссоциации
Растворы неэлектролитов. Закон Вант- Гоффа и Рауля. Растворы электролитов. Теория электронной диссоциации Периодический закон. Периодическая система элементов. (Лекция 2)
Периодический закон. Периодическая система элементов. (Лекция 2) Осмий. Нахождение в природе
Осмий. Нахождение в природе Каучук, резина и другие
Каучук, резина и другие Первые русские женщины-химики
Первые русские женщины-химики Anionic Polymerization
Anionic Polymerization Способ вызвать быструю, но контролируемую коррозию железа
Способ вызвать быструю, но контролируемую коррозию железа Алкены
Алкены Материальный баланс аппарата
Материальный баланс аппарата Азот и фосфор
Азот и фосфор Межклеточная сигнализация. Сигнальные молекулы. Гормоны. (Тема 3)
Межклеточная сигнализация. Сигнальные молекулы. Гормоны. (Тема 3) Изменения свойств химических элементов и их соединений в группах и периодах
Изменения свойств химических элементов и их соединений в группах и периодах Темір туралы
Темір туралы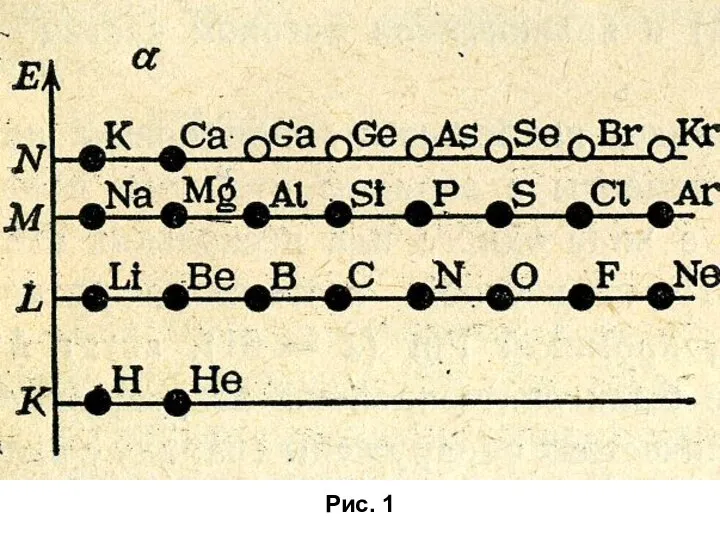 Строение атома и его электронной оболочки
Строение атома и его электронной оболочки Контроль качества эфирных масел
Контроль качества эфирных масел Орагническая химия. Теория А.М. Бутлерова
Орагническая химия. Теория А.М. Бутлерова Типовые процессы технологии неорганических веществ. (Тема 3)
Типовые процессы технологии неорганических веществ. (Тема 3) Жалпы химиялық технология
Жалпы химиялық технология Основные понятия и законы химии
Основные понятия и законы химии Оцет Яковлев Олексій 11-В
Оцет Яковлев Олексій 11-В  Кондуктометрия и кондуктометрическое титрование
Кондуктометрия и кондуктометрическое титрование Изотермический распад переохлажденного аустенита
Изотермический распад переохлажденного аустенита