Содержание
- 2. Chemical Formulas A molecular formula shows the exact number of atoms of each element in the
- 4. If you know the name of an ingredient, you can write a chemical formula, and the
- 14. Ionic compounds consist of a combination of cations and anions (metal + nonmetal). the formula is
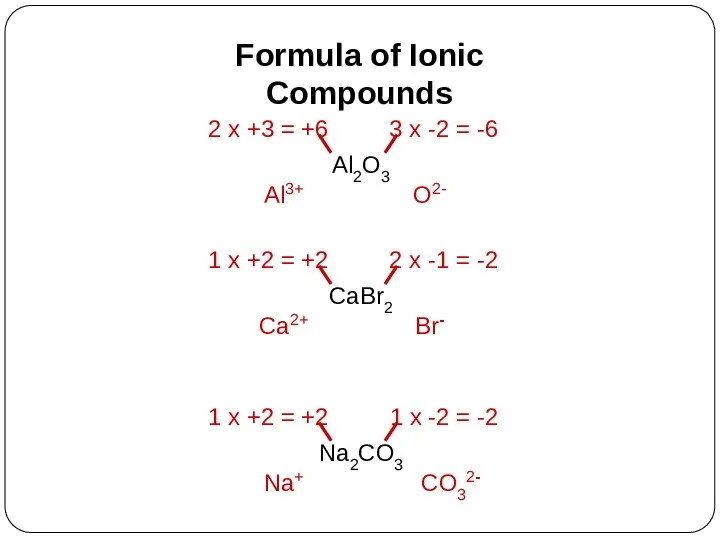
- 17. Formula of Ionic Compounds Al2O3 Al3+ O2- CaBr2 Ca2+ Br- Na2CO3 Na+ CO32-
- 22. Chemical Nomenclature Ionic Compounds often a metal + nonmetal anion (nonmetal), add “ide” to element name
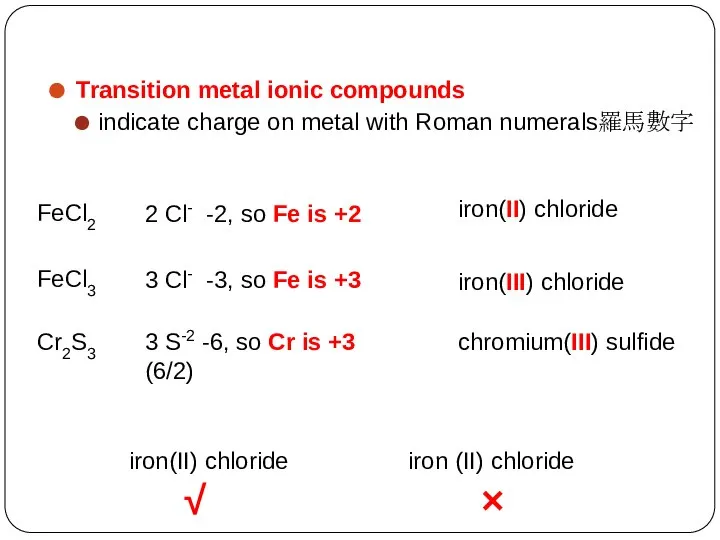
- 23. Transition metal ionic compounds indicate charge on metal with Roman numerals羅馬數字 FeCl2 2 Cl- -2, so
- 31. Molecular compounds nonmetals or nonmetals + metalloids common names H2O, NH3, CH4, C60 element further left
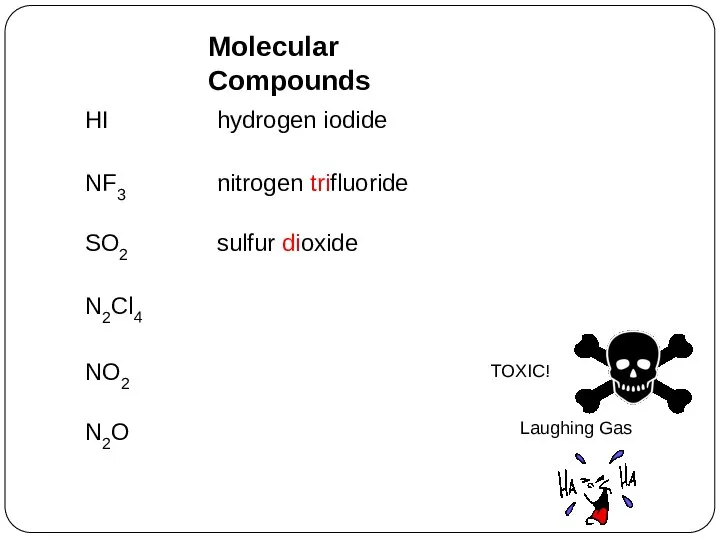
- 32. HI hydrogen iodide NF3 nitrogen trifluoride SO2 sulfur dioxide N2Cl4 NO2 N2O Molecular Compounds
- 35. Скачать презентацию





























 Дифузія у побуті
Дифузія у побуті Химическая связь. Электроотрицательность. Ковалентная связь
Химическая связь. Электроотрицательность. Ковалентная связь АБС-пластик. Свойства
АБС-пластик. Свойства Реакции окисления и восстановления органических соединений
Реакции окисления и восстановления органических соединений Основные сведения о композитах. Понятие о композитах
Основные сведения о композитах. Понятие о композитах Тема урока: Составление химических формул бинарных соединений по степени окисления атомов
Тема урока: Составление химических формул бинарных соединений по степени окисления атомов Пластмаси. Властивості пластмас. Склад пластичних мас. Види пластмаси
Пластмаси. Властивості пластмас. Склад пластичних мас. Види пластмаси Способы получения полимеров (Лекция 3)
Способы получения полимеров (Лекция 3) Окислительно-восстановительные реакции
Окислительно-восстановительные реакции Исследование возможности создания порохов на основе 1,1диамино-2,2-динитроэтилена
Исследование возможности создания порохов на основе 1,1диамино-2,2-динитроэтилена Робота учня 11-В класу: Андрійка Андрія
Робота учня 11-В класу: Андрійка Андрія  Виды присадок к моторным топливам
Виды присадок к моторным топливам Презентация Автор: Кудрявцева Марина Анатольевна Учитель химии МОУ «СОШ №12»
Презентация Автор: Кудрявцева Марина Анатольевна Учитель химии МОУ «СОШ №12»  Фазовые равновесия. Физическая и коллоидная химия
Фазовые равновесия. Физическая и коллоидная химия Теория растворов (лекция 1)
Теория растворов (лекция 1) Buxarlanma ve kondensasiya
Buxarlanma ve kondensasiya Рівняння характеристики крупності
Рівняння характеристики крупності Химический элемент фосфор. Фосфор в составе ДНК и РНК
Химический элемент фосфор. Фосфор в составе ДНК и РНК вУГЛЕВОДНІ Презентація швеця максима
вУГЛЕВОДНІ Презентація швеця максима  Шыны тәріздес фосфат
Шыны тәріздес фосфат Титан. Металл XXI века
Титан. Металл XXI века Реактопласти підготував учень 11-в класу Котенко Роман
Реактопласти підготував учень 11-в класу Котенко Роман  Кремний. Нахождение в природе. Свойства
Кремний. Нахождение в природе. Свойства Кальций. Маңызды химиялық формулалар
Кальций. Маңызды химиялық формулалар Галогены. Основные химические свойства. Качественные реакции
Галогены. Основные химические свойства. Качественные реакции Характеристика органических соединений
Характеристика органических соединений Радикальная полимеризация (Лекция 5)
Радикальная полимеризация (Лекция 5) Ароматические углеводороды (арены)
Ароматические углеводороды (арены)