Содержание
- 2. A repeating pattern of chemical properties in elements is called periodicity. What is the periodicity?
- 3. The periodicity in properties of elements can be explained by the the repetition of outermost shell
- 4. Atomic number and electrons The properties of elements are influenced by the number and arrangement of
- 5. Electron shells Electrons are arranged in shells around an atom’s nucleus. This electron arrangement is written
- 6. Electrons in period 1 Elements in period 1 only have electrons in the first shell. Why
- 7. Elements in period 2 all have a complete first shell. Аt second shell the number of
- 8. 2,8,1 2,8,2 2,8,3 2,8,4 2,8,5 2,8,6 2,8,7 2,8,8 Elements in period 3 have complete first and
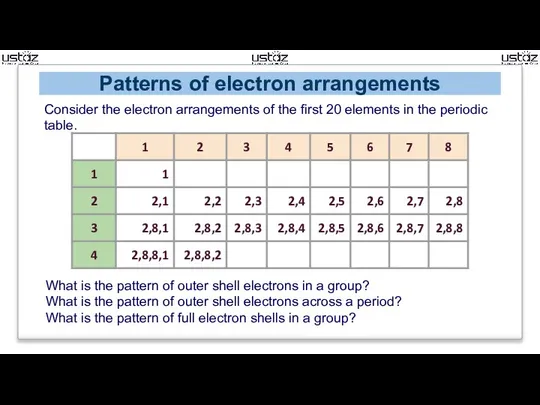
- 9. Patterns of electron arrangements Consider the electron arrangements of the first 20 elements in the periodic
- 10. Electron trends in the periodic table Trends down a group: By the start of new period
- 11. Electron trends in the periodic table The number of a group is the same as the
- 12. What is the electronic configuration?
- 13. As you know, all electrons are distributed among the shells and subshells. The arrangement of electrons
- 14. s s p s p s d p s d p s f d p s
- 15. s s p s p s d p s d p s f d p s
- 16. 1s 2s p 3s p 4s d p 5s d p 6s f d p 7s
- 17. 1s2 2s2 p 3s2 p 4s2 d p 5s2 d p 6s2 f d p 7s2
- 18. 1s2 2s2 2p 3s2 3p 4s2 d 4p 5s2 d 5p 6s2 f d 6p 7s2
- 19. 1s2 2s2 2p6 3s2 3p6 4s2 d 4p6 5s2 d 5p6 6s2 f d 6p6 7s2
- 20. 1s2 2s2 2p6 3s2 3p6 4s2 3d 4p6 5s2 4d 5p6 6s2 f 5d 6p6 7s2
- 21. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 f 5d10 6p6 7s2
- 22. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f 5d10 6p6 7s2
- 23. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d10 6p6 7s2
- 24. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d10 6p6 7s2
- 25. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d10 6p6 7s2
- 26. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d10 6p6 7s2
- 27. 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d10 6p6 7s2
- 28. According to their electronic configuration ending, elements can also be classified as s-block, p-block, d-block and
- 29. Task 2. Work in pairs. Create the sentences from mixed-up words and share your answer with
- 30. Answer: Task 2 A repeating pattern of chemical properties in elements is called periodicity. All the
- 31. Task 3. Find the mistake. Here 4 sentences. In each sentences 2 words are changed their
- 32. Task 3. Find the mistake. Here 4 sentences. In each sentences 2 words are changed their
- 33. Task 4. Electron trends in the periodic table. Write 4 sentences and make best matching of
- 34. Task 4. Electron trends in the periodic table. Write 4 sentences and make best matching of
- 36. Скачать презентацию
































 Элементарные процессы роста кристаллов (лекция 2)
Элементарные процессы роста кристаллов (лекция 2) c4bf82f888b84ff1b75efb1a64c84b0f
c4bf82f888b84ff1b75efb1a64c84b0f Получение и реакции пиразина
Получение и реакции пиразина Химическое сопротивление алюминий и его сплавов
Химическое сопротивление алюминий и его сплавов Энергетический обмен
Энергетический обмен Правила поведения зимой на воде
Правила поведения зимой на воде  Классы неорганических соединений Оксиды Кислоты Соли Основания
Классы неорганических соединений Оксиды Кислоты Соли Основания Основы промышленной экологии. Занятие 2
Основы промышленной экологии. Занятие 2 Пластмассалар, пластикалық материалдар
Пластмассалар, пластикалық материалдар Ізотопи і нукліди
Ізотопи і нукліди Номенклатура органических веществ - алканов
Номенклатура органических веществ - алканов Своя игра «Знаешь ли ты химические элементы?»
Своя игра «Знаешь ли ты химические элементы?» Химическая промышленность и химические технологии
Химическая промышленность и химические технологии Л3.12 Композицитонные материалы
Л3.12 Композицитонные материалы Выделение нефтегазоматеринских пород по данным пиролиза. Построение геохимического разреза
Выделение нефтегазоматеринских пород по данным пиролиза. Построение геохимического разреза Полимеры. Классификация, применение
Полимеры. Классификация, применение Значение органической химии Кондрашов Алексей | 9 А класс
Значение органической химии Кондрашов Алексей | 9 А класс  Средние породы. Типичный андезитовый вулкан
Средние породы. Типичный андезитовый вулкан Игра «Химические элементы» (формулы и названия)
Игра «Химические элементы» (формулы и названия) Агрохимия растений
Агрохимия растений Штучні і синтетичні волокна
Штучні і синтетичні волокна  Строение и функции отдельных коферментов
Строение и функции отдельных коферментов Релаксационные свойства полимеров
Релаксационные свойства полимеров Роль металлов в нашей жизни
Роль металлов в нашей жизни Конструкционные и специальные материалы холодильной техники
Конструкционные и специальные материалы холодильной техники Предмет химии. Вещества
Предмет химии. Вещества Гидролиз солей.
Гидролиз солей.  Использование дрожжей в пищевой промышленности Федеральное государственное образовательное учреждение среднего профессионал
Использование дрожжей в пищевой промышленности Федеральное государственное образовательное учреждение среднего профессионал