Содержание
- 2. I R O N Its density is 7.87 g/cm3 Melting point is 1538 oC Boiling point
- 3. Occurrence of iron Iron is second most abundant metal (6%) in the earth’s crust. But it
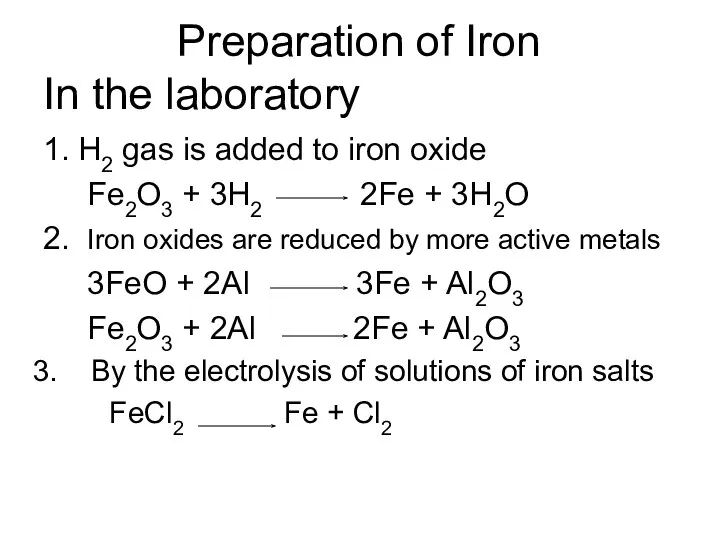
- 4. Preparation of Iron In the laboratory 1. H2 gas is added to iron oxide Fe2O3 +
- 5. Chemical Properties of Iron Iron has 26Fe: [18Ar]4s23d6 electron configuration In compounds, iron takes +2 and
- 6. Reactions of Iron 1) Iron reacts with dilute solutions of strong acids. Fe + 2HCl →
- 7. 2) Iron produces mixed oxides by water 3Fe + 4 H2O →Fe3O4 + 4H2 3) When
- 8. 4) At high temperature, it reacts with halogens. 2Fe(s) + 3Cl2(g) 1200°C 2FeCl3(s) Moisture and oxygen
- 9. Uses Iron is useful in our society today because iron is virtually used in everything :
- 10. IMPORTANT COMPOUNDS OF IRON Iron has +2 and +3 oxidation states in its compounds. Fe2+ ion
- 11. Iron (II) compounds (Ferro Compounds) 1. Iron (II) chloride, FeCl2 It is obtained by passing hydrogen
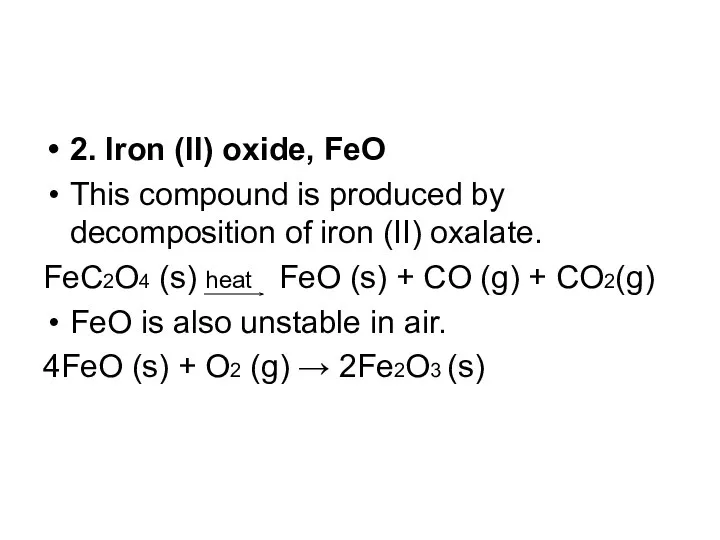
- 12. 2. Iron (II) oxide, FeO This compound is produced by decomposition of iron (II) oxalate. FeC2O4
- 13. Iron (III) Compounds (Ferric Compounds) 1. Iron(III) chloride, FeCl3 When iron is reacted with chlorine gas,
- 14. 2. Iron (III) hydroxide, Fe(OH)3 It is obtained by the reaction of Fe3+ with a base
- 15. 3. Iron (III) oxide, Fe2O3 In nature Fe2O3 is found in hematite and limonite minerals. It
- 16. Iron(II, III) oxide, Fe3O4 Fe3O4, mixed oxide, is obtained by passing heated steam over iron metal
- 18. Скачать презентацию


![Chemical Properties of Iron Iron has 26Fe: [18Ar]4s23d6 electron configuration In](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/1353612/slide-4.jpg)

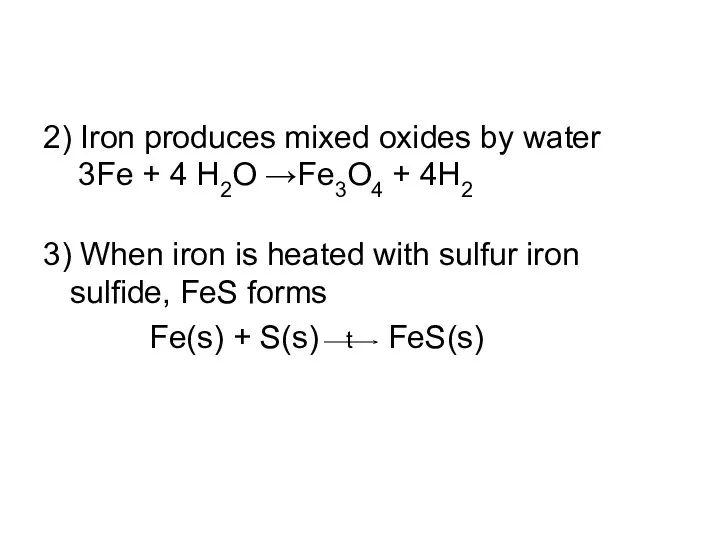








 Термическая переработка газов, нефтяных фракций и остатков нефтепереработки
Термическая переработка газов, нефтяных фракций и остатков нефтепереработки Понятие об адсорбционной влаге
Понятие об адсорбционной влаге Жирорастворимые витамины. (Лекция 12)
Жирорастворимые витамины. (Лекция 12) Презентация по Химии "Значение химии" - скачать смотреть бесплатно
Презентация по Химии "Значение химии" - скачать смотреть бесплатно Газообразное состояние вещества
Газообразное состояние вещества Радиофармацевтическая химия
Радиофармацевтическая химия Влияние нанокомпозитов платина/Нафион на электрокаталитическую активность реакции восстановления водорода и окисления кислорода
Влияние нанокомпозитов платина/Нафион на электрокаталитическую активность реакции восстановления водорода и окисления кислорода Периодический закон Д.И. Менделеева. Строение атома
Периодический закон Д.И. Менделеева. Строение атома Крем
Крем  Кристаллические решетки
Кристаллические решетки Растворы. Основные понятия. Концентрация. Законы Рауля
Растворы. Основные понятия. Концентрация. Законы Рауля Гормоны. Классификация гормонов. Общие свойства. Биохимия стероидных гормонов
Гормоны. Классификация гормонов. Общие свойства. Биохимия стероидных гормонов Nucleic acids
Nucleic acids Коррозия металла
Коррозия металла Викторина. Химический Элементариум
Викторина. Химический Элементариум Полимеры
Полимеры Химическое сопротивление алюминий и его сплавов
Химическое сопротивление алюминий и его сплавов Непредельные, ненасыщенные, ацетиленовые алкины
Непредельные, ненасыщенные, ацетиленовые алкины Химический элемент медь. (11 класс)
Химический элемент медь. (11 класс) Характеристика липидов. Углеводы. Моносахариды. Дисахариды. Полисахариды
Характеристика липидов. Углеводы. Моносахариды. Дисахариды. Полисахариды Radiation dosimetry
Radiation dosimetry Насыщенные (предельные) углеводороды. Алканы (парафины)
Насыщенные (предельные) углеводороды. Алканы (парафины) Энергетический обмен. Биологическое окисление
Энергетический обмен. Биологическое окисление Титан. Структура, применение, свойства (11 класс)
Титан. Структура, применение, свойства (11 класс) Катализатор Уилкинсона
Катализатор Уилкинсона Окислительно- восстановительные процессы
Окислительно- восстановительные процессы Презентация Щелочные металлы
Презентация Щелочные металлы  Марганец и его свойства
Марганец и его свойства