Содержание
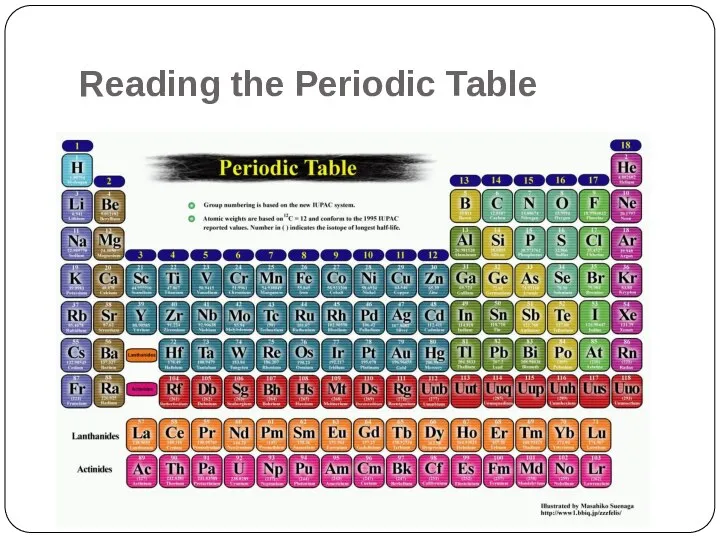
- 2. Reading the Periodic Table
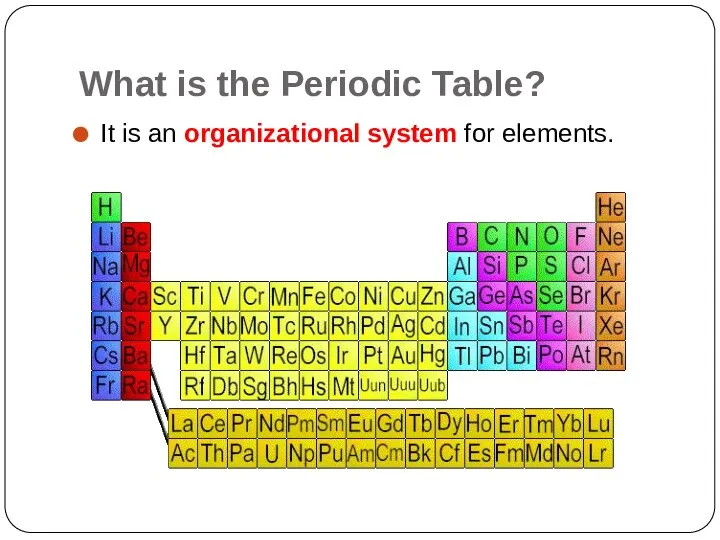
- 3. What is the Periodic Table? It is an organizational system for elements.
- 4. Who created it? The quest for a systematic arrangement of the elements started with the discovery
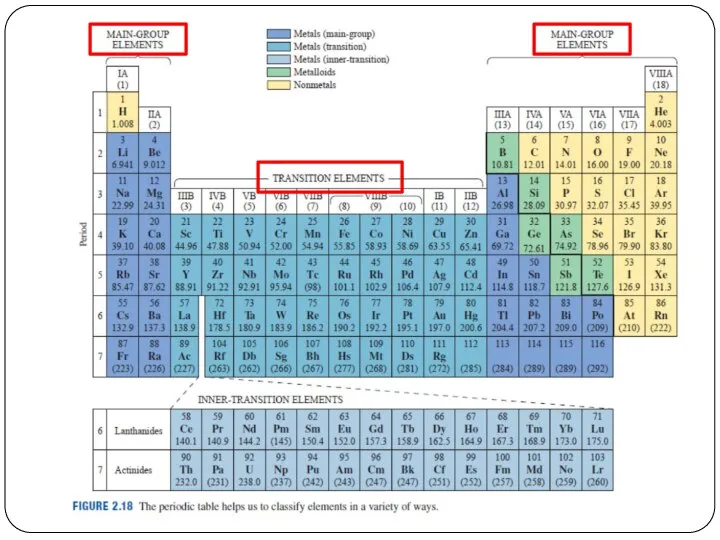
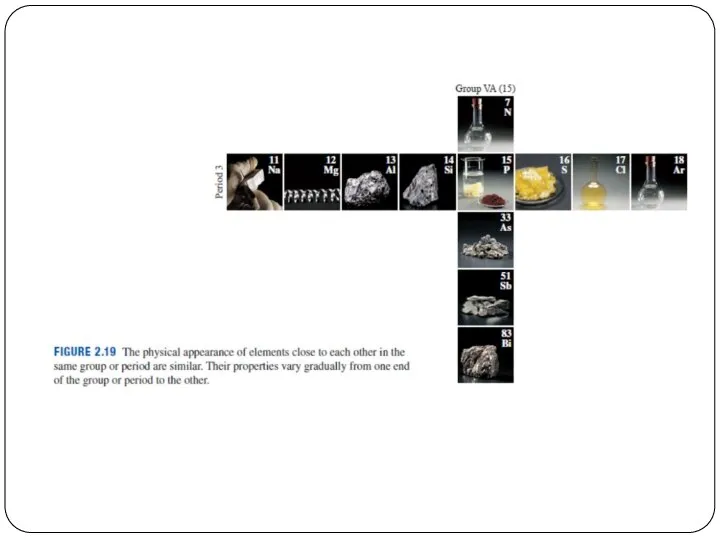
- 5. Periods = Rows 一排 The horizontal rows of the periodic table are called periods. Elements in
- 6. Periods = Rows Atomic mass increases from left to right across a period. Metals are on
- 7. Groups = Columns縱列 The vertical columns of the periodic table are called groups. Elements in the
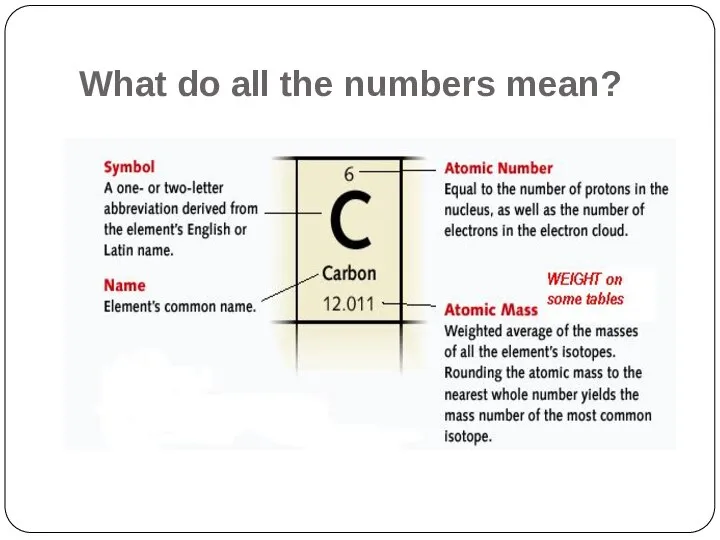
- 8. What do all the numbers mean?
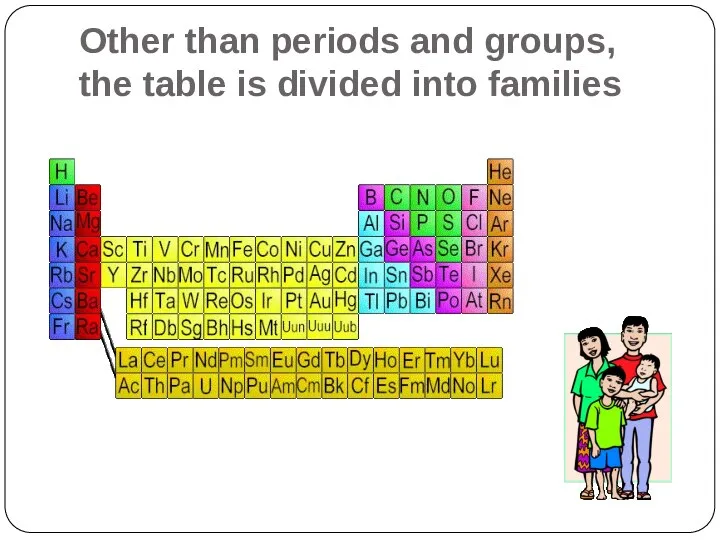
- 11. Other than periods and groups, the table is divided into families
- 12. Hydrogen Hydrogen belongs to a family of its own. Hydrogen is a diatomic (H2), reactive gas.
- 13. Alkali metals 1st column on the periodic table (Group 1) not including hydrogen. Their low ionization
- 14. Alkaline earth metals Second column on the periodic table (Group 2). They are very reactive metals,
- 15. Transition metals The transition elements are located in groups IB to VIIIB of the periodic table.
- 16. Rare earth elements The rare earth metals are found in group 3 of the periodic table,
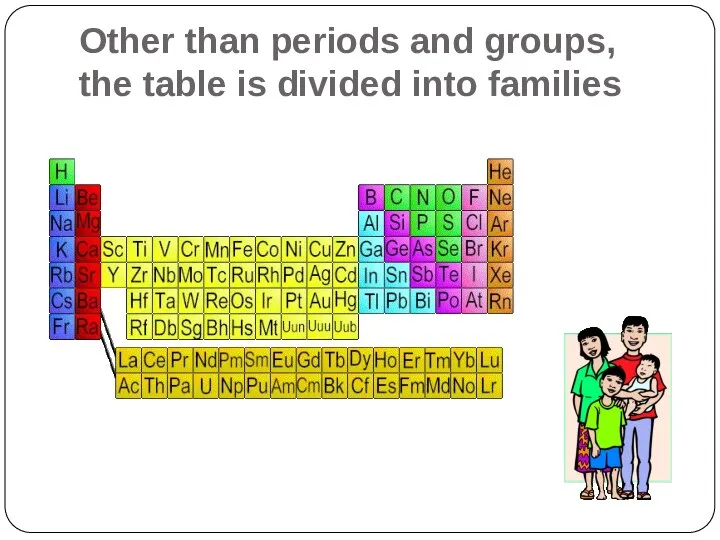
- 17. Other than periods and groups, the table is divided into families
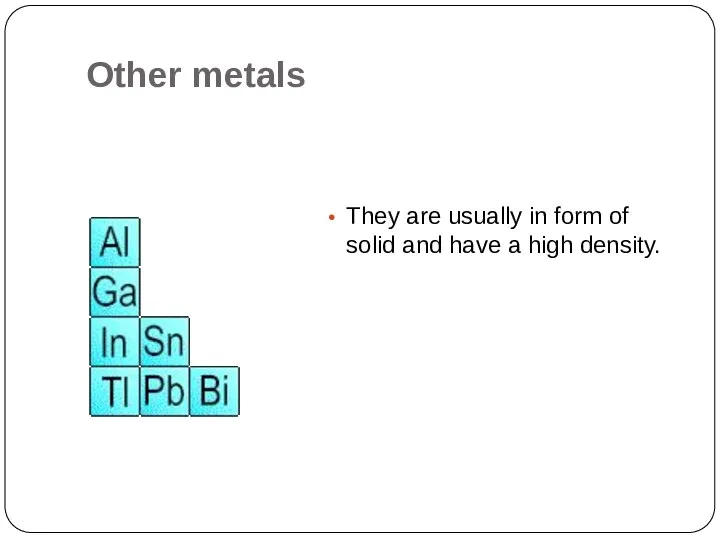
- 18. Other metals They are usually in form of solid and have a high density.
- 19. The electronegativities and ionization energies of the metalloids are between those of the metals and nonmetals,
- 20. The nonmetals are located on the upper right side of the periodic table. Nonmetals have high
- 21. The halogens are located in Group VIIA of the periodic table, and are a particular class
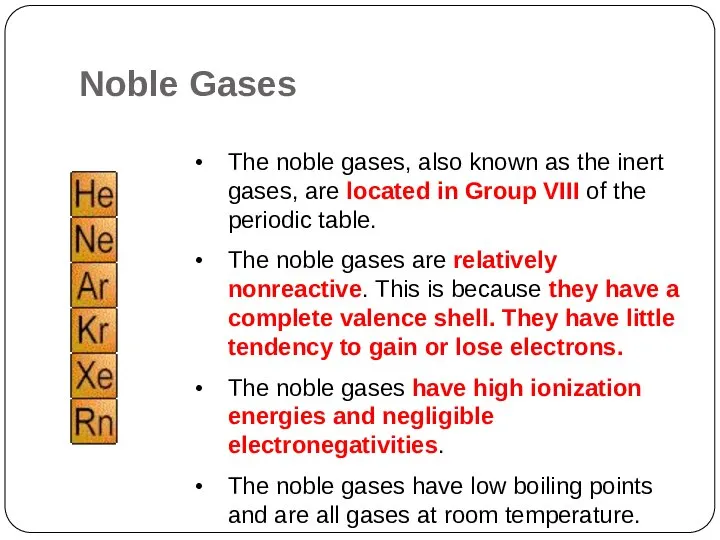
- 22. The noble gases, also known as the inert gases, are located in Group VIII of the
- 28. Скачать презентацию
















 Белки плазмы крови, их биологическая роль. Методы разделения белков плазмы крови. Диспротеинемии. Выполнила студентка 6 курса МБФ
Белки плазмы крови, их биологическая роль. Методы разделения белков плазмы крови. Диспротеинемии. Выполнила студентка 6 курса МБФ Презентация Лекарства дома
Презентация Лекарства дома Презентация Основные классы неорганических соединений
Презентация Основные классы неорганических соединений  Карбоновые кислоты и их функциональные производные. Хроматографические методы исследования
Карбоновые кислоты и их функциональные производные. Хроматографические методы исследования Пептиды. Тема 3
Пептиды. Тема 3 Аттестационная работа. Методическая разработка программы для 5 классов: Мир химии
Аттестационная работа. Методическая разработка программы для 5 классов: Мир химии Получение калийной селитры
Получение калийной селитры Углерод. Положение в периодической системе и строение атома
Углерод. Положение в периодической системе и строение атома Предмет и задачи металлургии благородных металлов; свойства и минералы благородных металлов
Предмет и задачи металлургии благородных металлов; свойства и минералы благородных металлов Самоорганизация на межфазных границах, метод Ленгмюра-Блоджетт. Объекты исследования коллоидной химии
Самоорганизация на межфазных границах, метод Ленгмюра-Блоджетт. Объекты исследования коллоидной химии Аттестационная работа. Развитие творческих возможностей учащихся через организацию исследовательской работы по химии
Аттестационная работа. Развитие творческих возможностей учащихся через организацию исследовательской работы по химии Презентация по Химии "Влияние пищевых добавок на здоровье человека" - скачать смотреть
Презентация по Химии "Влияние пищевых добавок на здоровье человека" - скачать смотреть  Дисахариды Ширяева София ХБ-4 г.Липецк 2012
Дисахариды Ширяева София ХБ-4 г.Липецк 2012 Амины
Амины Углеводы Органические соединения, содержащие в составе молекул наряду с карбонильной несколько гидроксильных групп, их пр
Углеводы Органические соединения, содержащие в составе молекул наряду с карбонильной несколько гидроксильных групп, их пр Вода. Химические и физические свойства
Вода. Химические и физические свойства Обобщающий урок по теме: «Основные классы неорганических соединений. Генетическая связь между классами неорганических соединени
Обобщающий урок по теме: «Основные классы неорганических соединений. Генетическая связь между классами неорганических соединени Классификация и краткие характеристики основных групп токсикантов
Классификация и краткие характеристики основных групп токсикантов Химия вокруг нас Работа учеников 9 класса «В» МБОУ «СОШ №25» г.Набережные Челны Давлиев Камиль и Юманьков Антон
Химия вокруг нас Работа учеников 9 класса «В» МБОУ «СОШ №25» г.Набережные Челны Давлиев Камиль и Юманьков Антон  Химический процесс: энергетика и равновесие
Химический процесс: энергетика и равновесие Галерея русских химиков
Галерея русских химиков Развитие учебной мотивации на уроках химии через систему урочной и внеурочной деятельности
Развитие учебной мотивации на уроках химии через систему урочной и внеурочной деятельности Биохимические функции железа
Биохимические функции железа Олійно-жирова промисловість Підготувала учениця 11-Б класу Халімон Заріна
Олійно-жирова промисловість Підготувала учениця 11-Б класу Халімон Заріна  Производство серной кислоты контактным способом
Производство серной кислоты контактным способом Химическая термодинамика
Химическая термодинамика Речь без пословицы - всё равно что еда без соли Речь без пословицы - всё равно что еда без соли · Пуд соли съешь, прежде чем познаешь человека · &nbs
Речь без пословицы - всё равно что еда без соли Речь без пословицы - всё равно что еда без соли · Пуд соли съешь, прежде чем познаешь человека · &nbs Катализ в химической технологии
Катализ в химической технологии