Содержание
- 2. INTRODUCTION This Powerpoint show is one of several produced to help students understand selected topics at
- 3. CONTENTS Prior knowledge Electron pair repulsion theory The regular molecular shapes Shapes of molecules with lone
- 4. Before you start it would be helpful to… know the definition of a covalent bond know
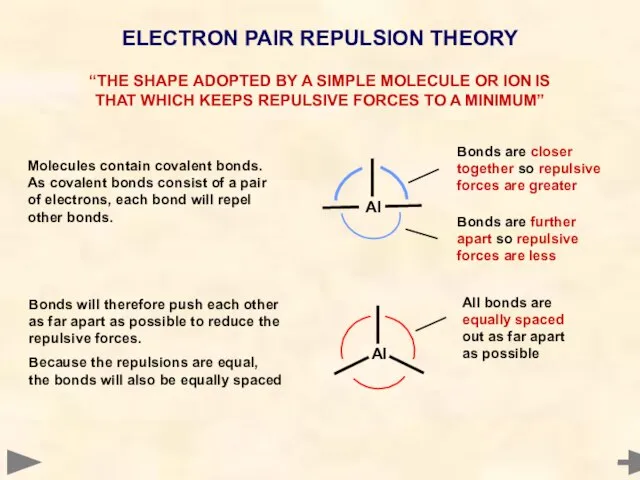
- 5. ELECTRON PAIR REPULSION THEORY “THE SHAPE ADOPTED BY A SIMPLE MOLECULE OR ION IS THAT WHICH
- 6. ELECTRON PAIR REPULSION THEORY “THE SHAPE ADOPTED BY A SIMPLE MOLECULE OR ION IS THAT WHICH
- 7. REGULAR SHAPES Molecules, or ions, possessing ONLY BOND PAIRS of electrons fit into a set of
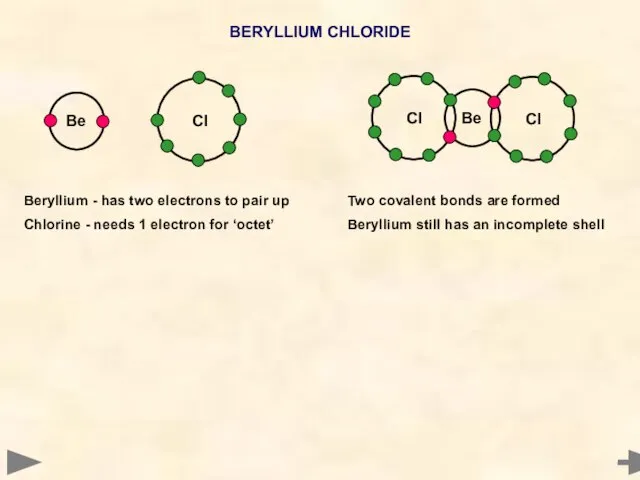
- 8. BERYLLIUM CHLORIDE Beryllium - has two electrons to pair up Chlorine - needs 1 electron for
- 9. BERYLLIUM CHLORIDE BOND PAIRS 2 LONE PAIRS 0 BOND ANGLE... SHAPE... 180° LINEAR Beryllium - has
- 10. ADDING ANOTHER ATOM - ANIMATION
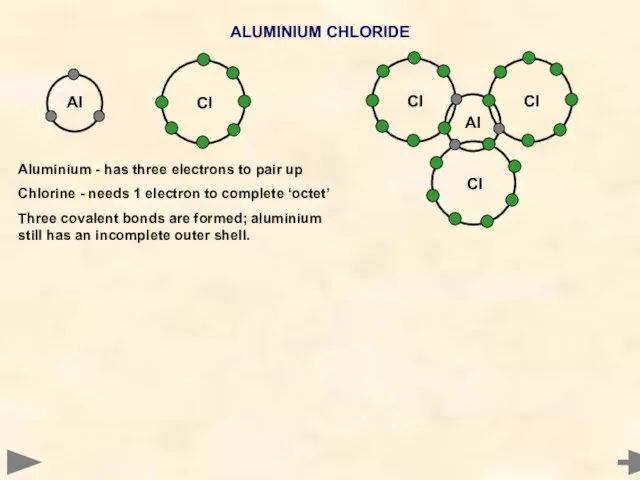
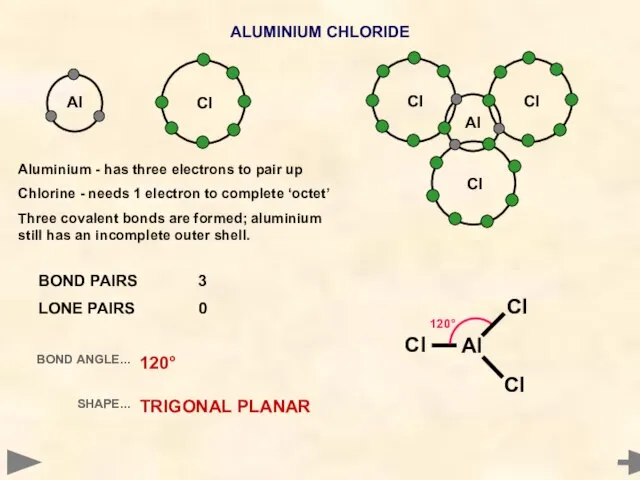
- 11. Al ALUMINIUM CHLORIDE Aluminium - has three electrons to pair up Chlorine - needs 1 electron
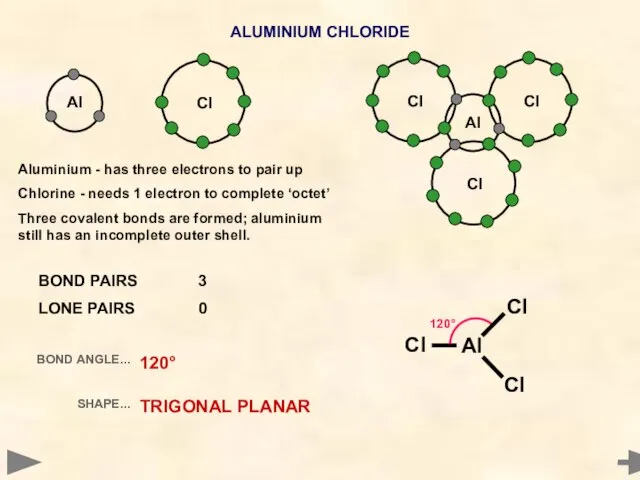
- 12. Al ALUMINIUM CHLORIDE Cl Cl Al 120° Cl BOND PAIRS 3 LONE PAIRS 0 BOND ANGLE...
- 13. Al ALUMINIUM CHLORIDE Cl Cl Al 120° Cl BOND PAIRS 3 LONE PAIRS 0 BOND ANGLE...
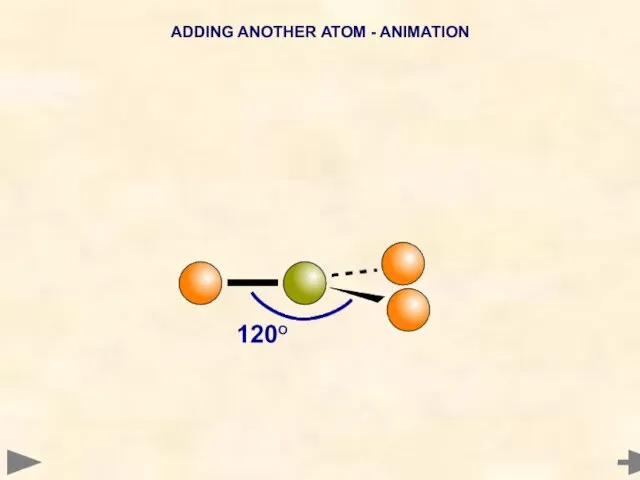
- 14. ADDING ANOTHER ATOM - ANIMATION
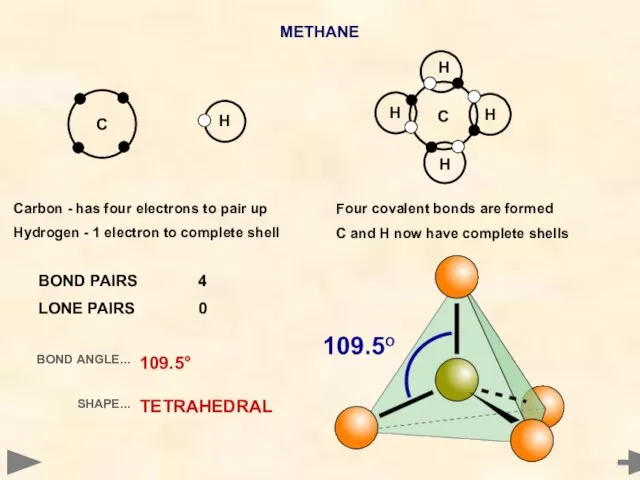
- 15. METHANE Carbon - has four electrons to pair up Hydrogen - 1 electron to complete shell
- 16. METHANE BOND PAIRS 4 LONE PAIRS 0 BOND ANGLE... SHAPE... 109.5° TETRAHEDRAL Carbon - has four
- 17. METHANE BOND PAIRS 4 LONE PAIRS 0 BOND ANGLE... SHAPE... 109.5° TETRAHEDRAL Carbon - has four
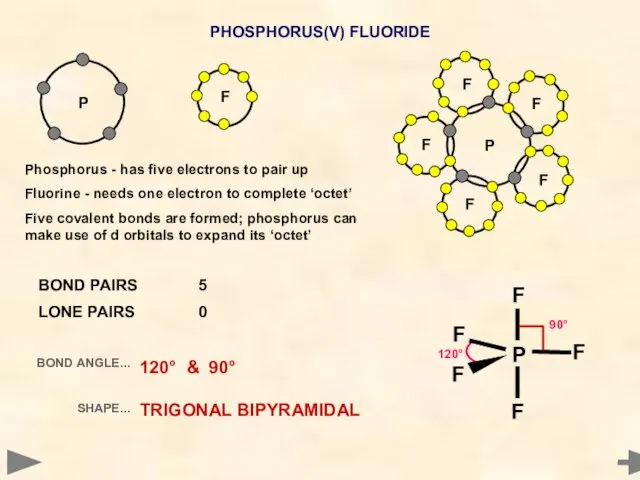
- 18. PHOSPHORUS(V) FLUORIDE P Phosphorus - has five electrons to pair up Fluorine - needs one electron
- 19. PHOSPHORUS(V) FLUORIDE P BOND PAIRS 5 LONE PAIRS 0 BOND ANGLE... SHAPE... 120° & 90° TRIGONAL
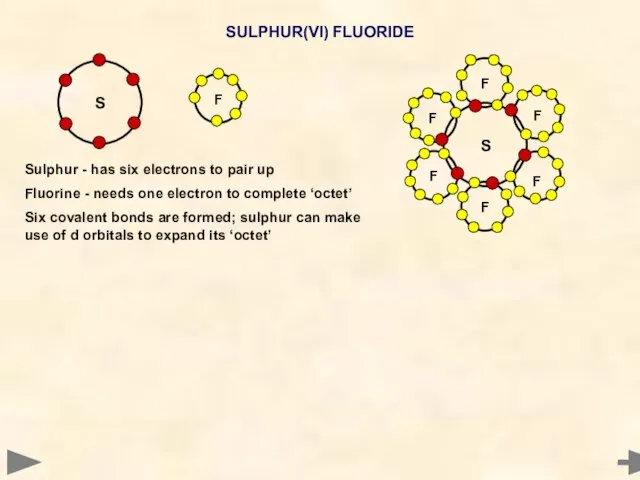
- 20. SULPHUR(VI) FLUORIDE Sulphur - has six electrons to pair up Fluorine - needs one electron to
- 21. SULPHUR(VI) FLUORIDE BOND PAIRS 6 LONE PAIRS 0 BOND ANGLE... SHAPE... 90° OCTAHEDRAL Sulphur - has
- 22. SULPHUR(VI) FLUORIDE BOND PAIRS 6 LONE PAIRS 0 BOND ANGLE... SHAPE... 90° OCTAHEDRAL Sulphur - has
- 23. IRREGULAR SHAPES If a molecule, or ion, has lone pairs on the central atom, the shapes
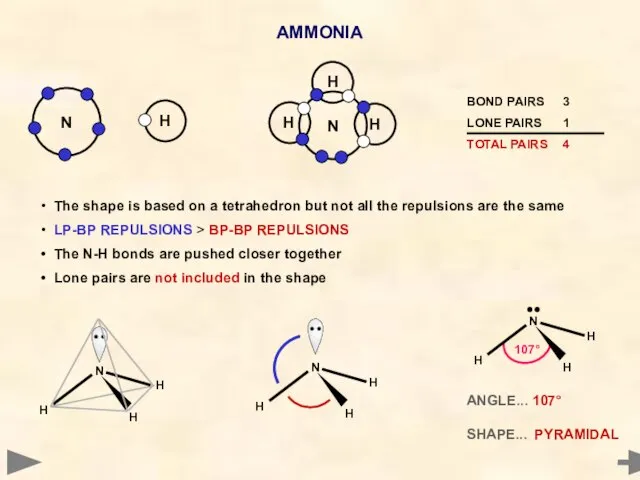
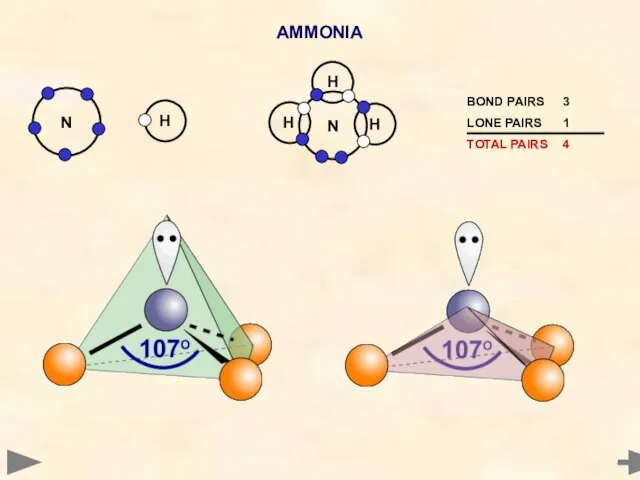
- 24. AMMONIA Nitrogen has five electrons in its outer shell It cannot pair up all five -
- 25. AMMONIA ANGLE... 107° SHAPE... PYRAMIDAL H H N H The shape is based on a tetrahedron
- 26. AMMONIA
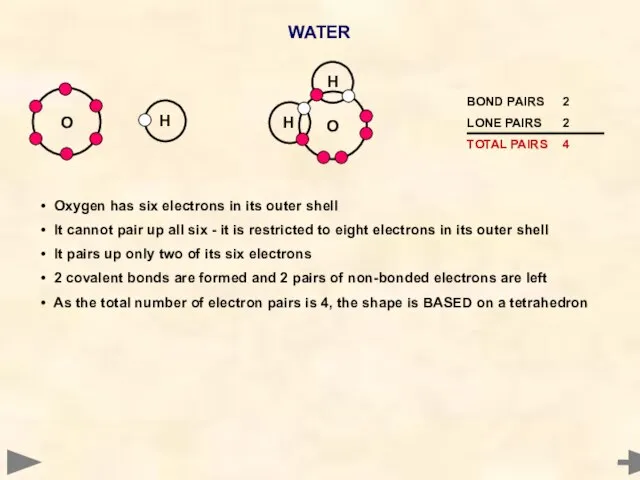
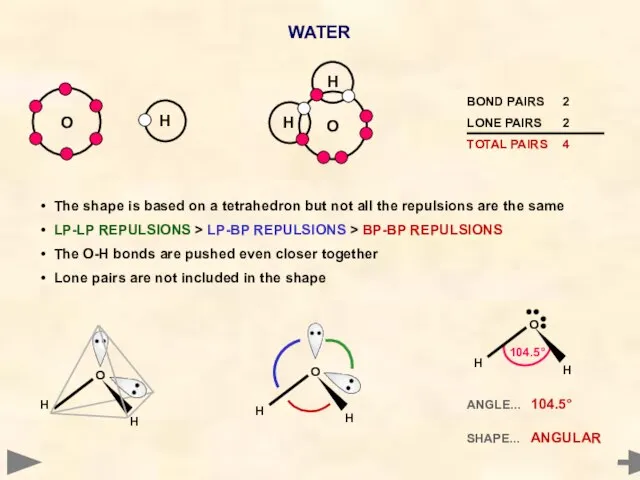
- 27. WATER Oxygen has six electrons in its outer shell It cannot pair up all six -
- 28. ANGLE... 104.5° SHAPE... ANGULAR H O H H O H 104.5° H O H The shape
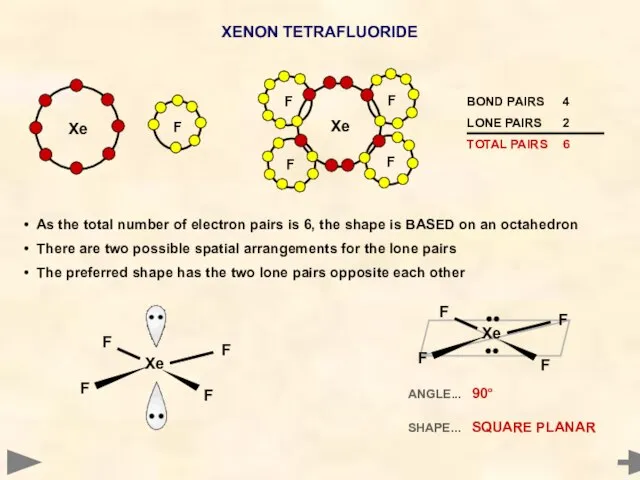
- 29. XENON TETRAFLUORIDE Xenon has eight electrons in its outer shell It pairs up four of its
- 30. XENON TETRAFLUORIDE F F F F Xe ANGLE... 90° SHAPE... SQUARE PLANAR As the total number
- 31. CALCULATING THE SHAPE OF IONS The shape of a complex ion is calculated in the same
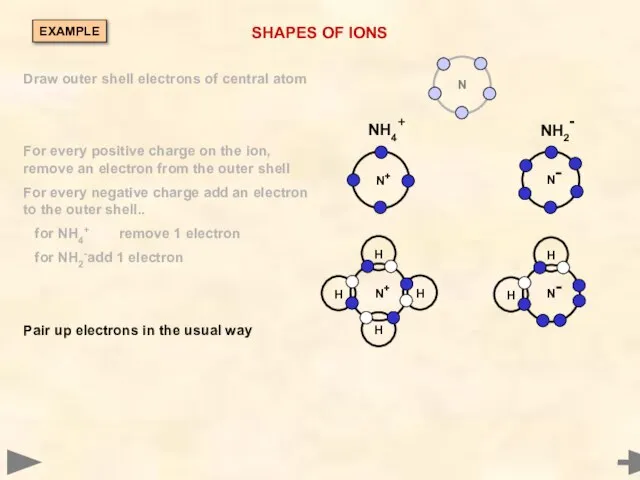
- 32. SHAPES OF IONS Draw outer shell electrons of central atom EXAMPLE
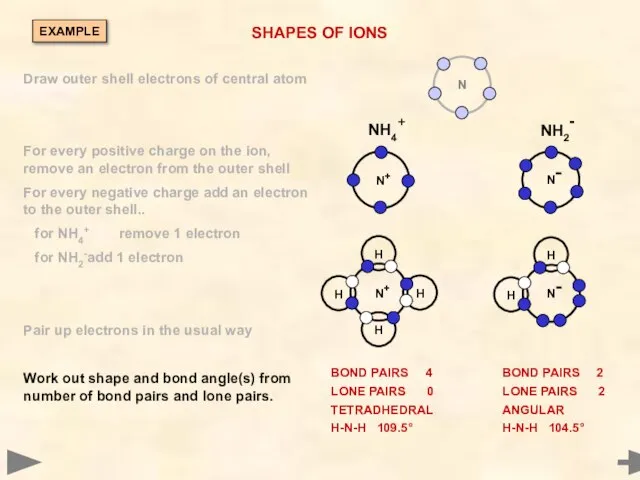
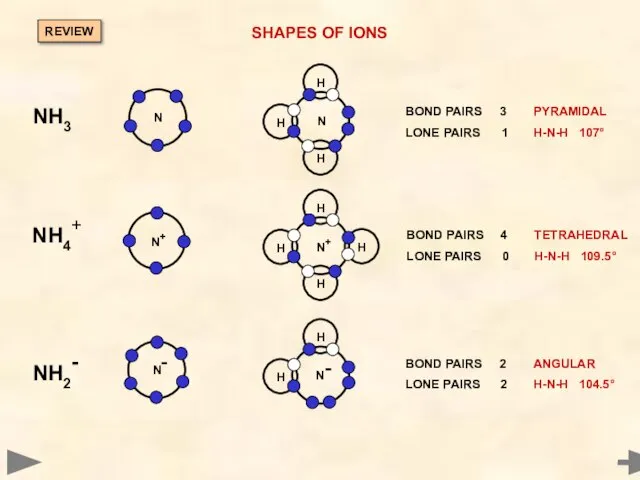
- 33. SHAPES OF IONS NH4+ NH2- Draw outer shell electrons of central atom For every positive charge
- 34. SHAPES OF IONS NH4+ NH2- Draw outer shell electrons of central atom For every positive charge
- 35. SHAPES OF IONS NH4+ NH2- BOND PAIRS 4 LONE PAIRS 0 TETRADHEDRAL H-N-H 109.5° BOND PAIRS
- 36. SHAPES OF IONS BOND PAIRS 3 PYRAMIDAL LONE PAIRS 1 H-N-H 107° BOND PAIRS 4 TETRAHEDRAL
- 37. MOLECULES WITH DOUBLE BONDS C O O Carbon - needs four electrons to complete its shell
- 38. MOLECULES WITH DOUBLE BONDS C O O Carbon - needs four electrons to complete its shell
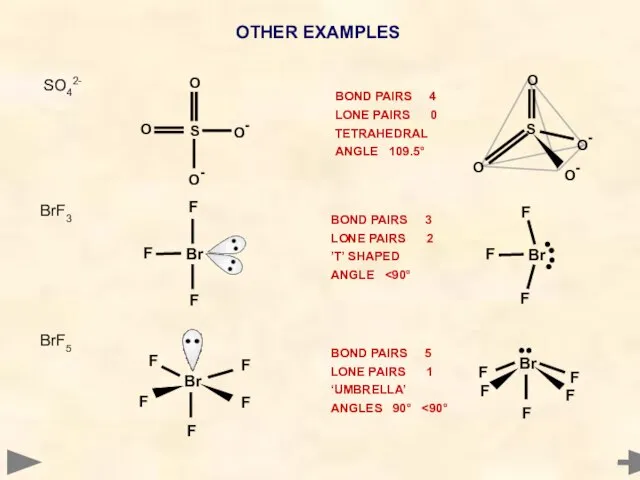
- 39. OTHER EXAMPLES BrF5 BOND PAIRS 5 LONE PAIRS 1 ‘UMBRELLA’ ANGLES 90° F F F F
- 40. ANSWERS ON NEXT PAGE TEST QUESTIONS For each of the following ions/molecules, state the number of
- 41. TEST QUESTIONS 3 bp 0 lp 120º trigonal planar boron pairs up all 3 electrons in
- 42. REVISION CHECK What should you be able to do? Recall the theory of Electron Pair Repulsion
- 43. You need to go over the relevant topic(s) again Click on the button to return to
- 44. WELL DONE! Try some past paper questions
- 46. Скачать презентацию

























 Альдегиды и кетоны
Альдегиды и кетоны Машиностроительные материалы
Машиностроительные материалы Изучение свойств пластичных масс для лепки
Изучение свойств пластичных масс для лепки Нанотехнологии. Методы получения наноматериалов. (Лекция 2)
Нанотехнологии. Методы получения наноматериалов. (Лекция 2) Химический диктант как метод формирующего оценивания
Химический диктант как метод формирующего оценивания Тағамдық қоспалардың функционалдық жүктелуі және олардың сипаттамасы
Тағамдық қоспалардың функционалдық жүктелуі және олардың сипаттамасы Сравнительный анализ Фармакопейных статей для субстанций, представленных в мировых Фармакопеях
Сравнительный анализ Фармакопейных статей для субстанций, представленных в мировых Фармакопеях Решение задач на нахождение молекулярной формулы органических веществ
Решение задач на нахождение молекулярной формулы органических веществ Дисахариды и полисахариды
Дисахариды и полисахариды Атомның электрондық конфегурациясы
Атомның электрондық конфегурациясы Скорость химической реакции
Скорость химической реакции Гниение аминокислот в кишечнике
Гниение аминокислот в кишечнике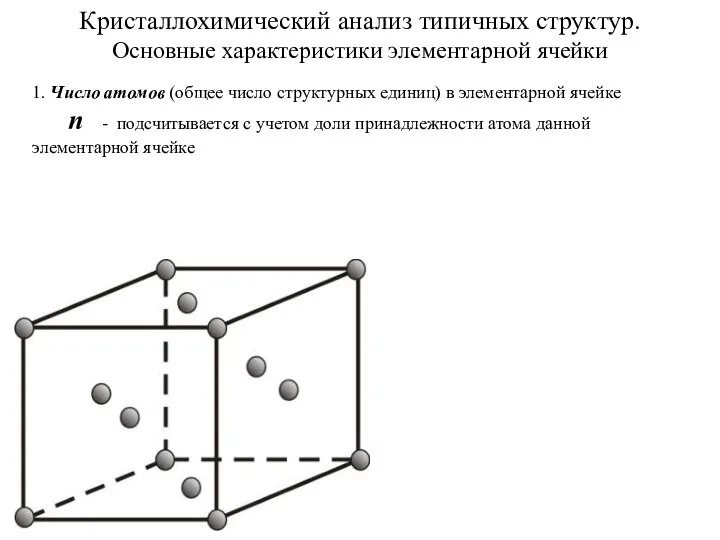 Кристаллохимический анализ типичных структур. Основные характеристики элементарной ячейки
Кристаллохимический анализ типичных структур. Основные характеристики элементарной ячейки Растворы. Термодинамическая теория растворов
Растворы. Термодинамическая теория растворов Элементы группы VII B
Элементы группы VII B Экологический проект Тема: Лабораторное исследование химического состава чипсов Работу выполнила Купавцева Татьяна
Экологический проект Тема: Лабораторное исследование химического состава чипсов Работу выполнила Купавцева Татьяна  Изучение фотокатализа в химии
Изучение фотокатализа в химии Основное уравнение молекулярно-кинетической теории
Основное уравнение молекулярно-кинетической теории Яблочная кладовая Автор работы: Быстрова Анастасия, ученица 9«А» класса Руководитель: Баранова Алевтина Владимировна 2010
Яблочная кладовая Автор работы: Быстрова Анастасия, ученица 9«А» класса Руководитель: Баранова Алевтина Владимировна 2010 Презентация по Химии "Белки" - скачать смотреть _
Презентация по Химии "Белки" - скачать смотреть _ Металлы. Общая характеристика
Металлы. Общая характеристика Своя игра «Знаешь ли ты химические элементы?»
Своя игра «Знаешь ли ты химические элементы?» Свойства металла. Подготовительная группа 2
Свойства металла. Подготовительная группа 2 Адсорбционные равновесия и процессы на подвижных и неподвижных границах раздела фаз
Адсорбционные равновесия и процессы на подвижных и неподвижных границах раздела фаз Закон сохранения массы веществ Омельянчук Т.Е. учитель химии МАОУ ДСОШ №2 г. Домодедово
Закон сохранения массы веществ Омельянчук Т.Е. учитель химии МАОУ ДСОШ №2 г. Домодедово Основы МКТ
Основы МКТ Жири. Жири в природі. Біологічна роль жирів. Жири. Жири в природі. Біологічна роль жирів.
Жири. Жири в природі. Біологічна роль жирів. Жири. Жири в природі. Біологічна роль жирів.  ДИВІНИЛОВИЙ КАУЧУК Підготували Реутенко А., Даниленко А., 11-А
ДИВІНИЛОВИЙ КАУЧУК Підготували Реутенко А., Даниленко А., 11-А