Содержание
- 2. Acids and alkalis Solutions can be sorted by whether they are: acid, alkali or neutral. When
- 3. When the oxide of some non-metals dissolve in water they make an acid. Acids have a
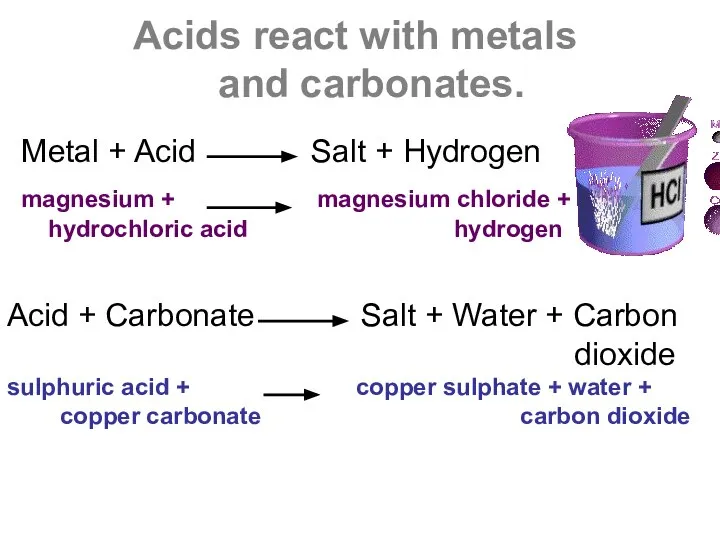
- 4. Acids react with metals and carbonates. Metal + Acid Salt + Hydrogen magnesium + magnesium chloride
- 5. Acids Lemon juice contains citric acid, and vinegar contains ethanoic acid. Some strong acids are hydrochloric
- 6. Neutralisation Acids and alkalis react with each other. The alkali cancels out the acid in the
- 7. Salts The salt made depends on the acid and alkali used. The salt contains the metal
- 8. Alkalis When the oxides of some metals dissolve in water they make an alkali solution. Alkalis
- 9. Alkalis Alkalis are present in many cleaning substances in use in our homes. Kitchen cleaners are
- 10. Indicators They change colour in acid or alkaline solutions. Different indicators change to different colours. Indicators
- 11. Litmus Test Litmus is an indicator. It changes colour in acid and alkaline solutions. Litmus is
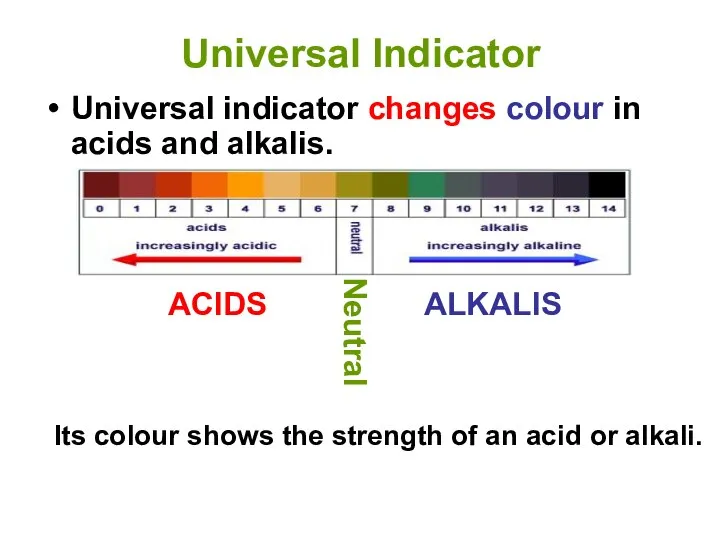
- 12. Universal Indicator Universal indicator changes colour in acids and alkalis. Its colour shows the strength of
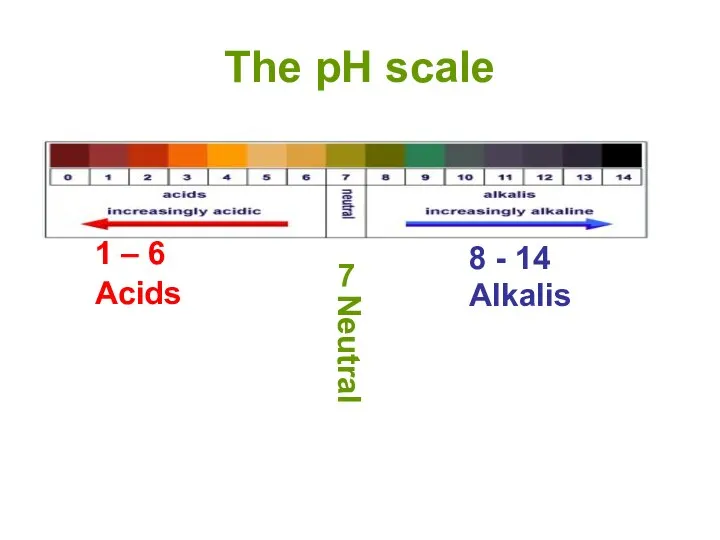
- 13. The pH scale 1 – 6 8 - 14 Alkalis 7 Neutral Acids
- 14. Applications of Neutralisation Indigestion: Our stomach carries around hydrochloric acid. Too much of this leads to
- 16. Скачать презентацию










 Алканы. Этан - C₂H₆
Алканы. Этан - C₂H₆ Кремний и его соединения
Кремний и его соединения Исследовательская работа Химические средства в быту
Исследовательская работа Химические средства в быту Арены. 10 класс
Арены. 10 класс Химический состав клетки. Неорганические вещества клетки
Химический состав клетки. Неорганические вещества клетки Химия-технологиялық процестеріндегі негізгі заңдылықтары. Технологиялық процестерідегі тепетеңдік
Химия-технологиялық процестеріндегі негізгі заңдылықтары. Технологиялық процестерідегі тепетеңдік Пресс - конференция «Карбоновые кислоты в природе, химические свойства, применение.»
Пресс - конференция «Карбоновые кислоты в природе, химические свойства, применение.» Презентация Строение и свойства циклоалканов.
Презентация Строение и свойства циклоалканов.  Органічні сполуки в побуті
Органічні сполуки в побуті Гидроксид магния. Mg(OH)2
Гидроксид магния. Mg(OH)2 Гидролиз. Применение
Гидролиз. Применение Определение формул органических веществ
Определение формул органических веществ Обменные реакции в растворах электролитов
Обменные реакции в растворах электролитов Кислотные и основные свойства органических соединений
Кислотные и основные свойства органических соединений Атомы. Молекулы. Ионы
Атомы. Молекулы. Ионы Карбоновые кислоты
Карбоновые кислоты Способы получения полимеров (Лекция 3)
Способы получения полимеров (Лекция 3) Гранулометрический метод контроля
Гранулометрический метод контроля Закономерности изменения металлических и неметаллических свойств элементов в периодах и группах (8 класс)
Закономерности изменения металлических и неметаллических свойств элементов в периодах и группах (8 класс) Төменгі фосфориттерден азот – фосфорлы тыңайтқыштар алу технологиясын әзірлеу
Төменгі фосфориттерден азот – фосфорлы тыңайтқыштар алу технологиясын әзірлеу Негізгі топша элементтерінің химиясы
Негізгі топша элементтерінің химиясы Эндогенная серия. Магматическая группа
Эндогенная серия. Магматическая группа Алканы. Предельные углеводороды. Парафины. Насыщенные углеводороды
Алканы. Предельные углеводороды. Парафины. Насыщенные углеводороды Химиялық кинетика
Химиялық кинетика Ток в жидкостях. Закон электролиза. Гальваностегия. Гальванопластика
Ток в жидкостях. Закон электролиза. Гальваностегия. Гальванопластика Тканые армирующие материалы
Тканые армирующие материалы Обмен липидов
Обмен липидов Презентация по Химии "Презентация Химический состав растений" - скачать смотреть
Презентация по Химии "Презентация Химический состав растений" - скачать смотреть