Содержание
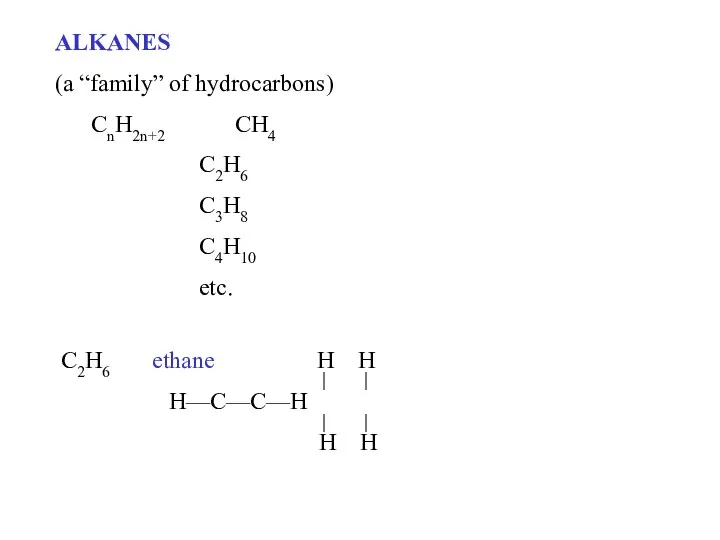
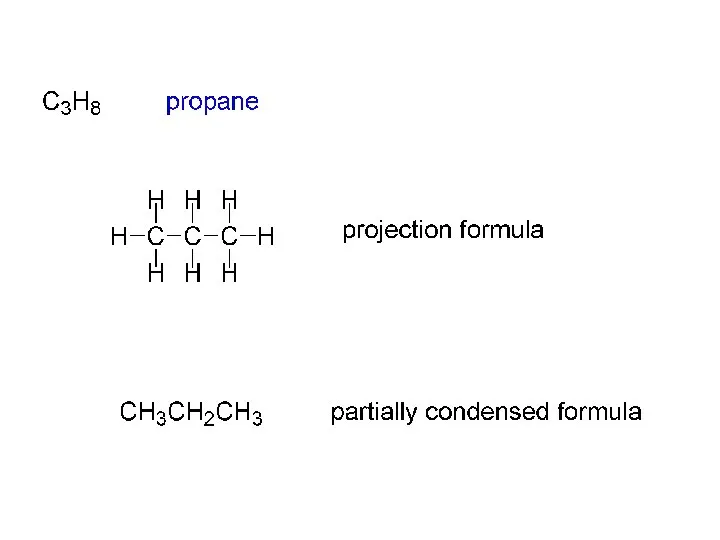
- 2. ALKANES (a “family” of hydrocarbons) CnH2n+2 CH4 C2H6 C3H8 C4H10 etc. C2H6 ethane H H H—C—C—H
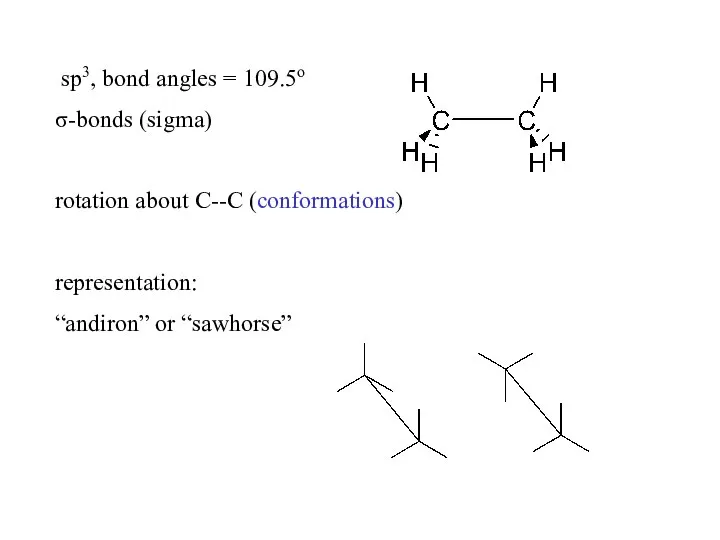
- 3. sp3, bond angles = 109.5o σ-bonds (sigma) rotation about C--C (conformations) representation: “andiron” or “sawhorse”
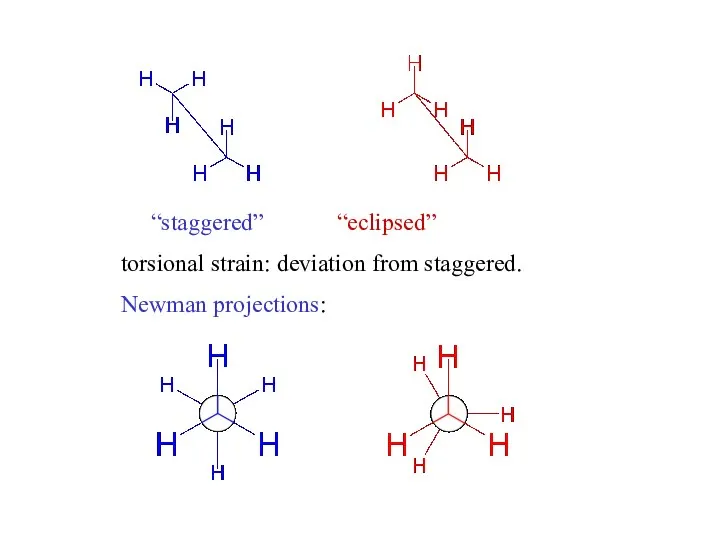
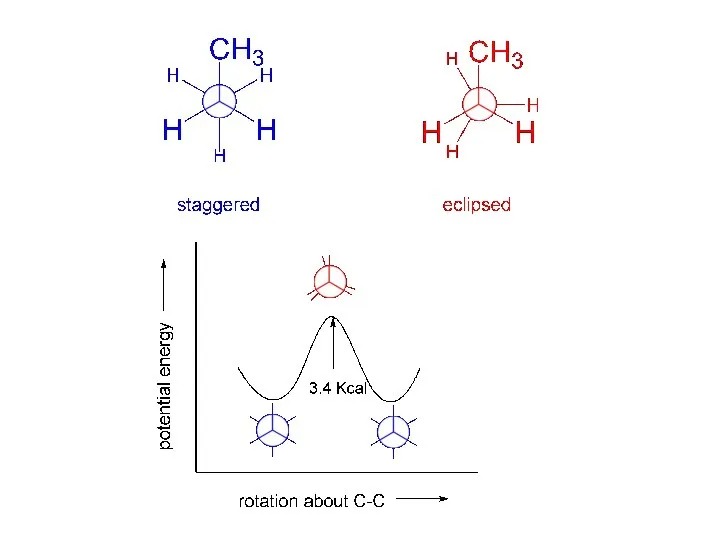
- 4. “staggered” “eclipsed” torsional strain: deviation from staggered. Newman projections:
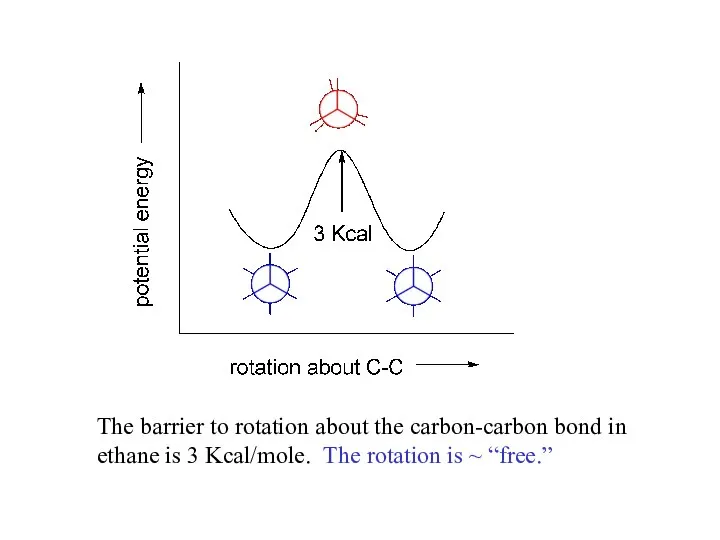
- 5. The barrier to rotation about the carbon-carbon bond in ethane is 3 Kcal/mole. The rotation is
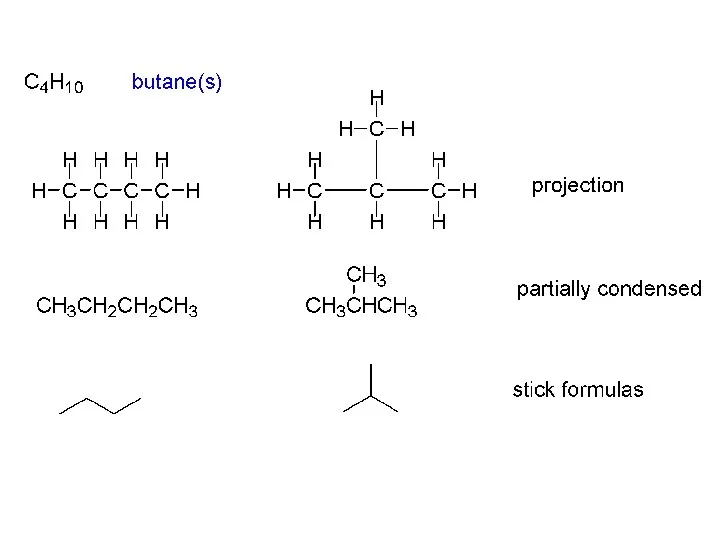
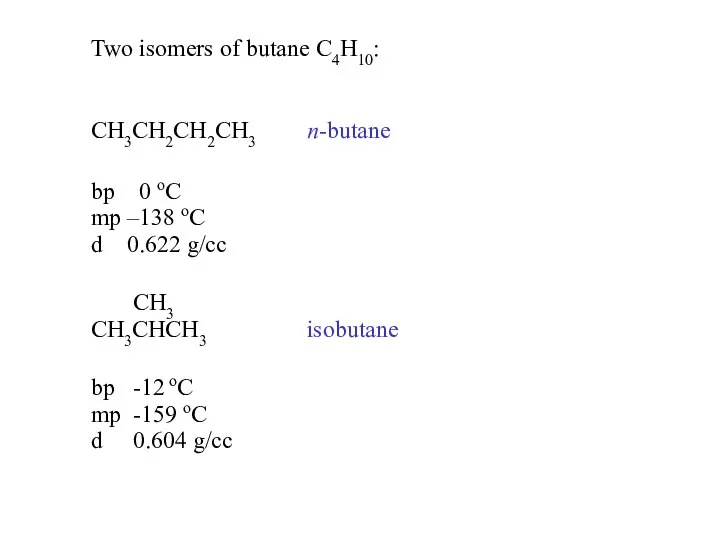
- 9. Two isomers of butane C4H10: CH3CH2CH2CH3 n-butane bp 0 oC mp –138 oC d 0.622 g/cc
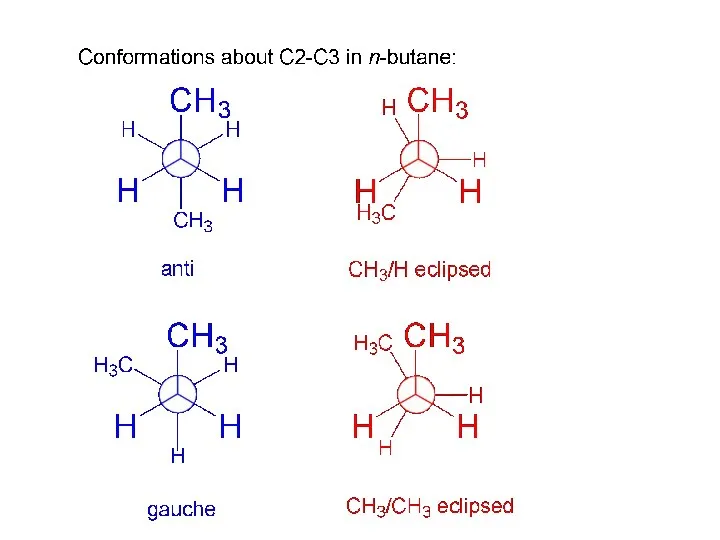
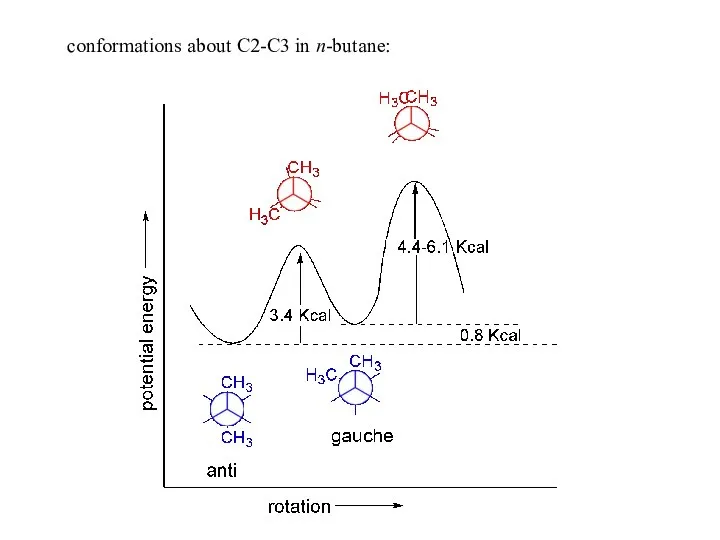
- 11. conformations about C2-C3 in n-butane:
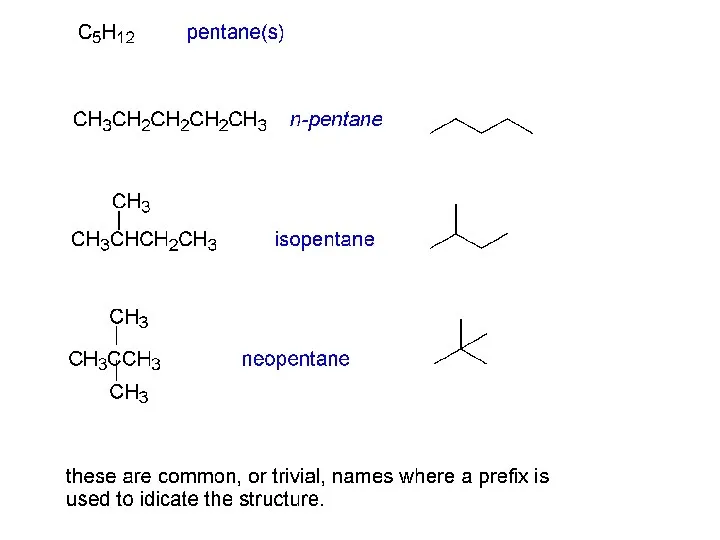
- 13. Alkane name isomers CH4 methane 1 C2H6 ethane 1 C3H8 propane 1 C4H10 butanes 2 C5H12
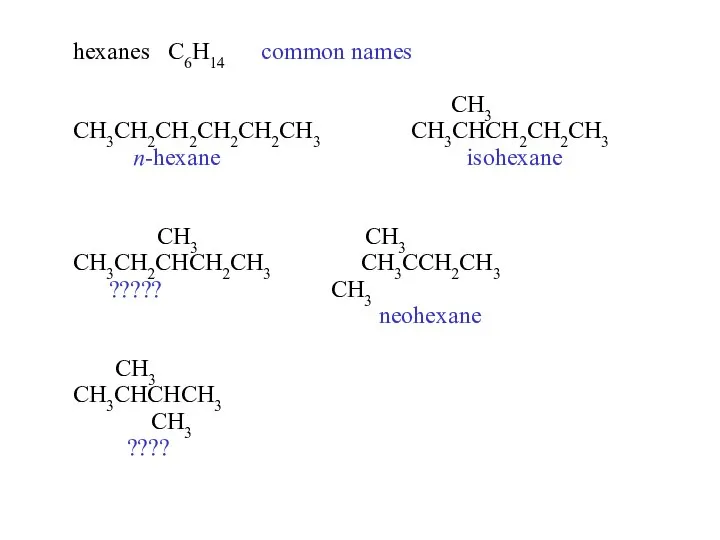
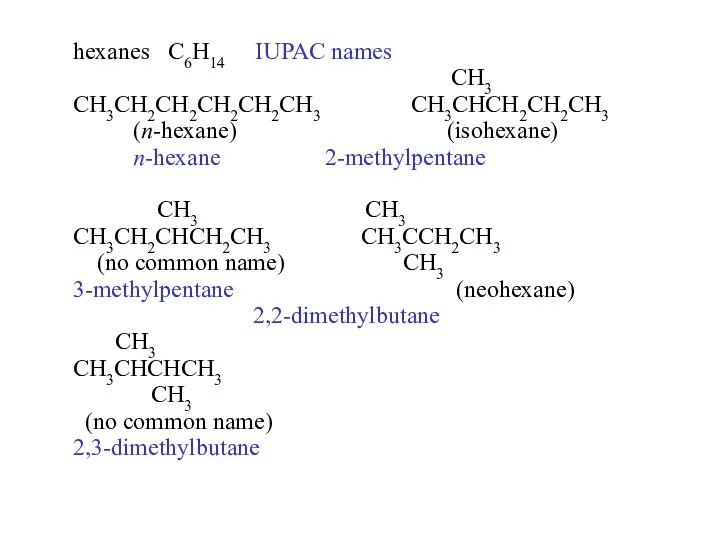
- 14. hexanes C6H14 common names CH3 CH3CH2CH2CH2CH2CH3 CH3CHCH2CH2CH3 n-hexane isohexane CH3 CH3 CH3CH2CHCH2CH3 CH3CCH2CH3 ????? CH3 neohexane
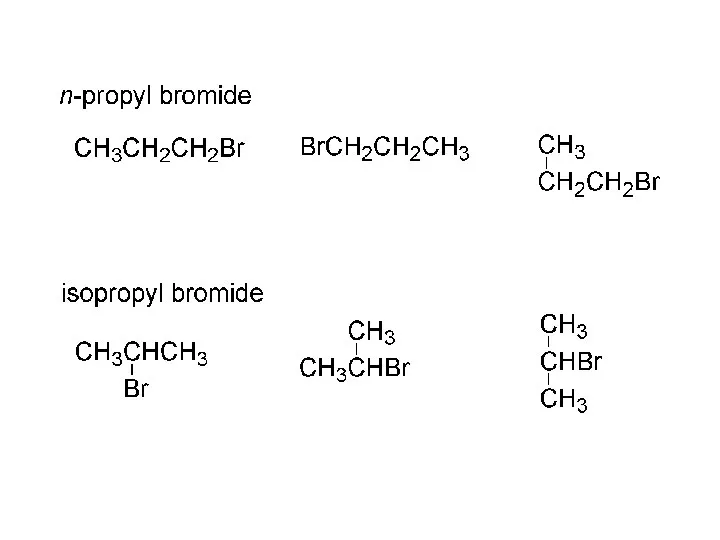
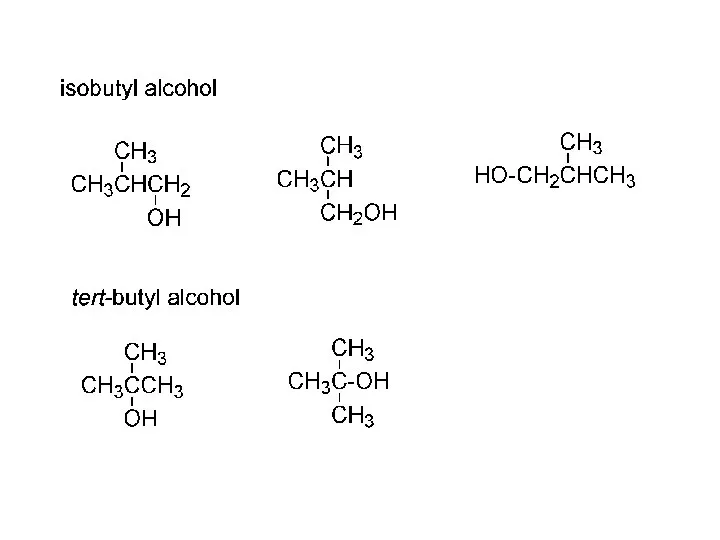
- 15. IUPAC nomenclature (Geneva, 1920) names of radicals (alkyl groups): CH3- “methyl” CH3Cl methyl chloride CH3OH methyl
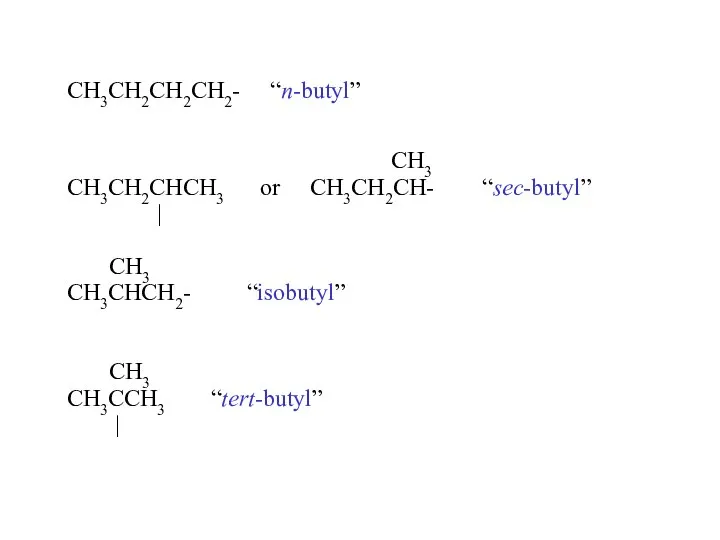
- 16. CH3CH2CH2CH2- “n-butyl” CH3 CH3CH2CHCH3 or CH3CH2CH- “sec-butyl” | CH3 CH3CHCH2- “isobutyl” CH3 CH3CCH3 “tert-butyl” |
- 20. Web problems to help with naming and recognizing organic radicals: Click here or copy and paste
- 21. IUPAC rules for naming alkanes: parent chain = longest continuous carbon chain ? “alkane”. branches on
- 22. hexanes C6H14 IUPAC names CH3 CH3CH2CH2CH2CH2CH3 CH3CHCH2CH2CH3 (n-hexane) (isohexane) n-hexane 2-methylpentane CH3 CH3 CH3CH2CHCH2CH3 CH3CCH2CH3 (no
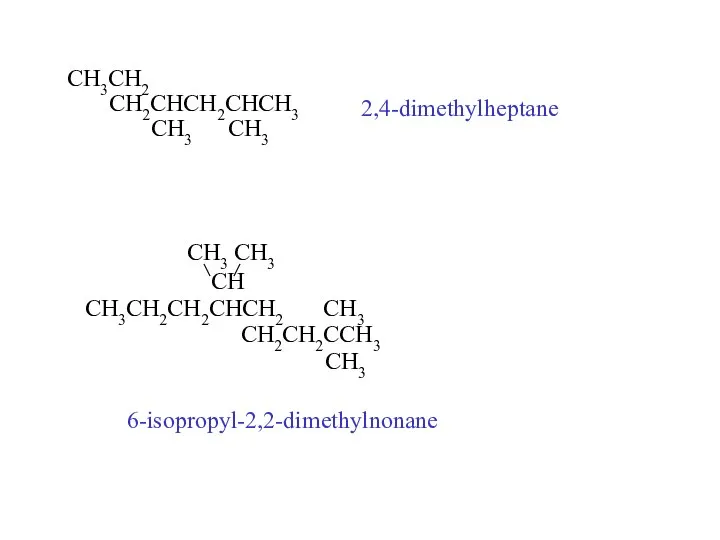
- 23. CH3CH2 CH2CHCH2CHCH3 CH3 CH3 2,4-dimethylheptane CH3 CH3 CH CH3CH2CH2CHCH2 CH3 CH2CH2CCH3 CH3 6-isopropyl-2,2-dimethylnonane
- 24. “classes of carbons” primary carbon (1o) – a carbon bonded to one carbon secondary carbon (2o)
- 25. classification of hydrogens, halides – hydrogens or halides are classified by the carbon to which they
- 26. alkanes, physical properties non-polar or only weakly polar, cannot hydrogen bond ? relatively weak intermolecular forces
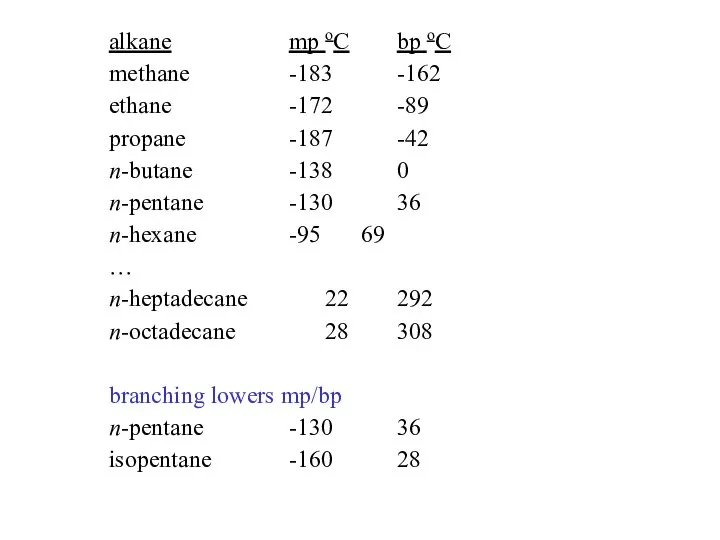
- 27. alkane mp oC bp oC methane -183 -162 ethane -172 -89 propane -187 -42 n-butane -138
- 28. fossil fuels: natural gas petroleum coal petroleum is a complex mixture of hydrocarbons 1. solvents 2.
- 29. products from fractional distillation of petroleum: fraction b. range carbons natural gas below 20o C1 –
- 30. syntheses Industrial Laboratory large amounts (tons) small amounts (grams) lowest cost non-profit mixtures often okay pure
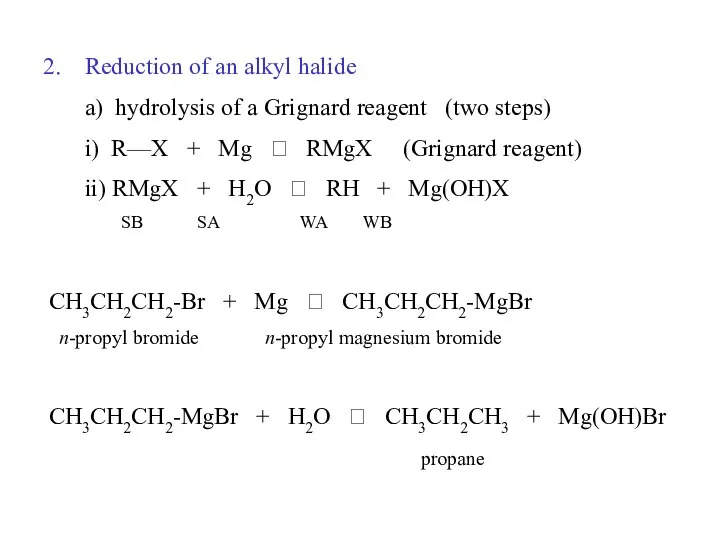
- 31. Alkanes, syntheses: (to be covered later) Reduction of an alkyl halide a) hydrolysis of a Grignard
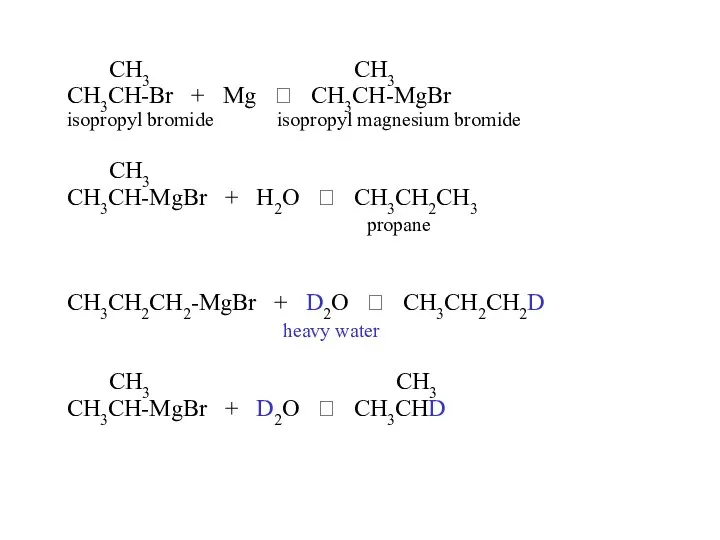
- 32. Reduction of an alkyl halide a) hydrolysis of a Grignard reagent (two steps) i) R—X +
- 33. CH3 CH3 CH3CH-Br + Mg ? CH3CH-MgBr isopropyl bromide isopropyl magnesium bromide CH3 CH3CH-MgBr + H2O
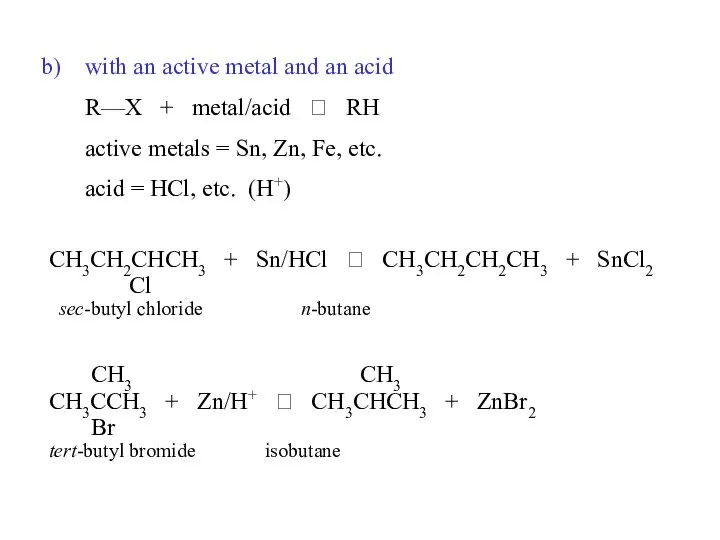
- 34. with an active metal and an acid R—X + metal/acid ? RH active metals = Sn,
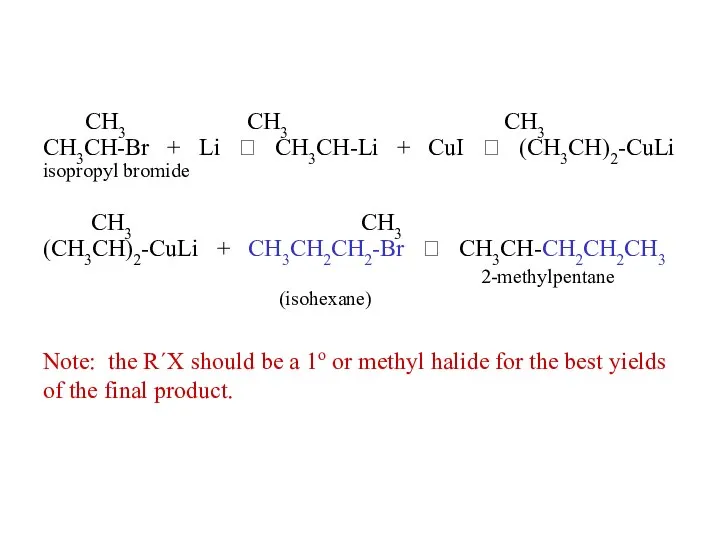
- 35. Corey-House synthesis R-X + Li ? R-Li + CuI ? R2CuLi R2CuLi + R´-X ? R—R´
- 36. note: the previous equations are not balanced: R-X + 2 Li ? R-Li + LiX 2
- 37. CH3 CH3 CH3 CH3CH-Br + Li ? CH3CH-Li + CuI ? (CH3CH)2-CuLi isopropyl bromide CH3 CH3
- 38. Alkanes, syntheses: (to be covered later) Reduction of an alkyl halide a) hydrolysis of a Grignard
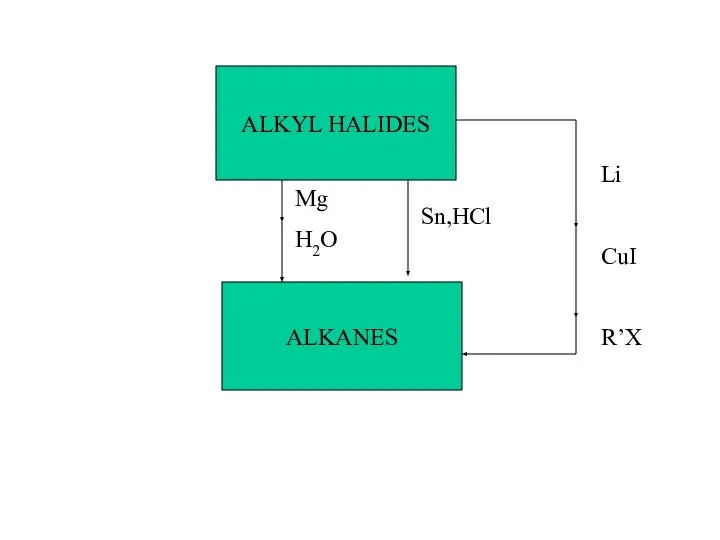
- 39. ALKANES ALKYL HALIDES Mg H2O Sn,HCl Li CuI R’X
- 40. Reactions of alkanes: alkane + H2SO4 ? no reaction (NR) alkane + NaOH ? NR alkane
- 41. Alkane, reactions: Halogenation 2. Combustion (oxidation) 3. Pyrolysis (cracking)
- 42. Combustion CnH2n+2 + (xs) O2, flame ? n CO2 + (n+1) H2O + heat gasoline, diesel,
- 43. Halogenation R-H + X2, heat or hv ? R-X + HX a) heat or light required
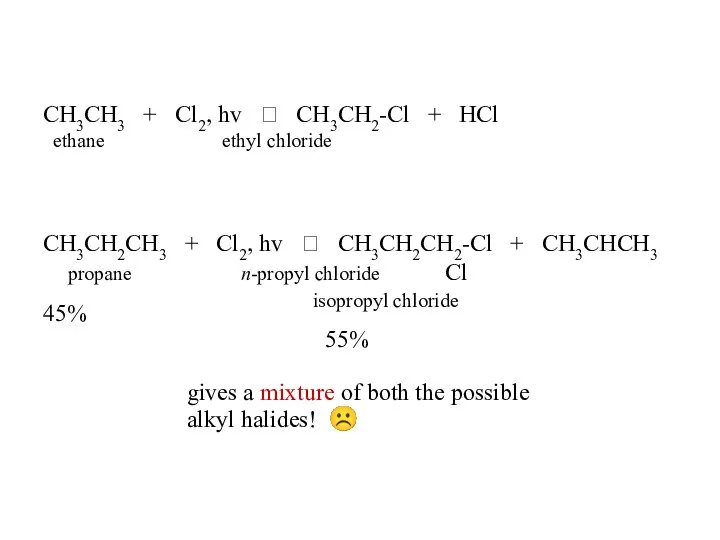
- 44. CH3CH3 + Cl2, hv ? CH3CH2-Cl + HCl ethane ethyl chloride CH3CH2CH3 + Cl2, hv ?
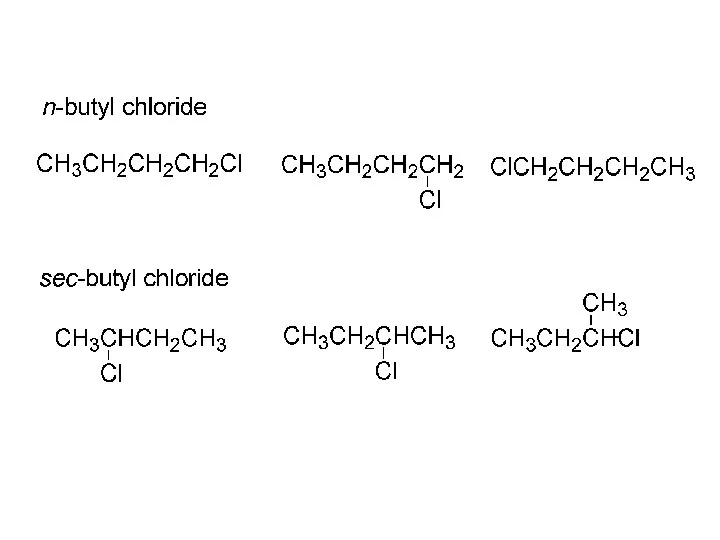
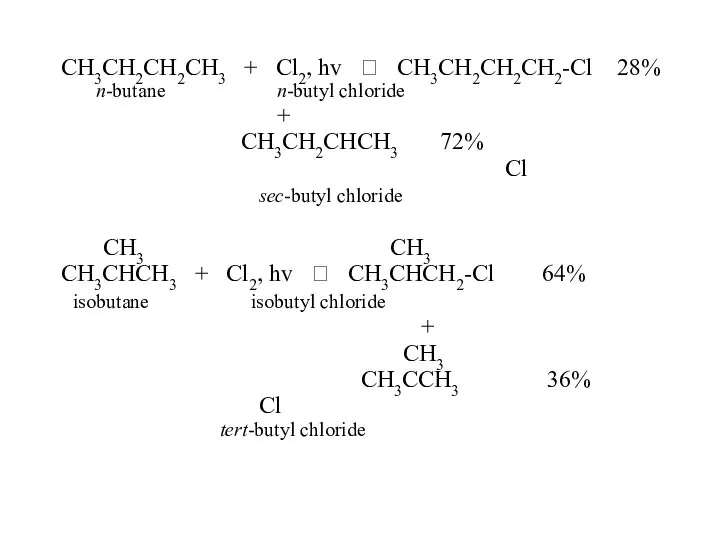
- 45. CH3CH2CH2CH3 + Cl2, hv ? CH3CH2CH2CH2-Cl 28% n-butane n-butyl chloride + CH3CH2CHCH3 72% Cl sec-butyl chloride
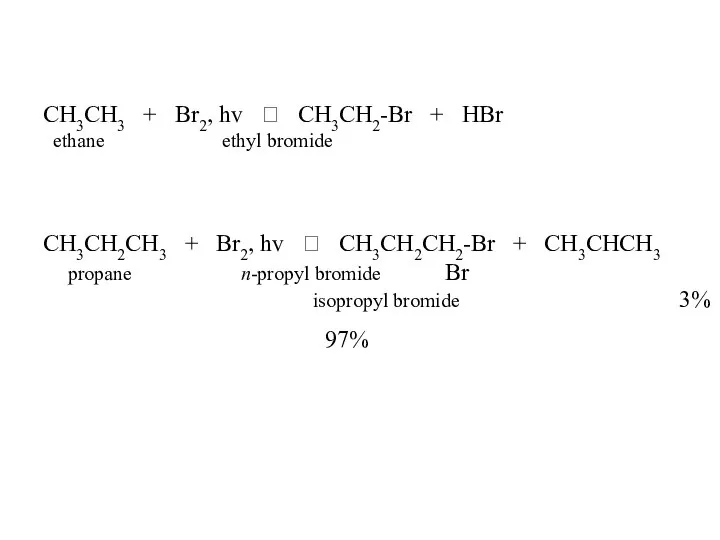
- 46. CH3CH3 + Br2, hv ? CH3CH2-Br + HBr ethane ethyl bromide CH3CH2CH3 + Br2, hv ?
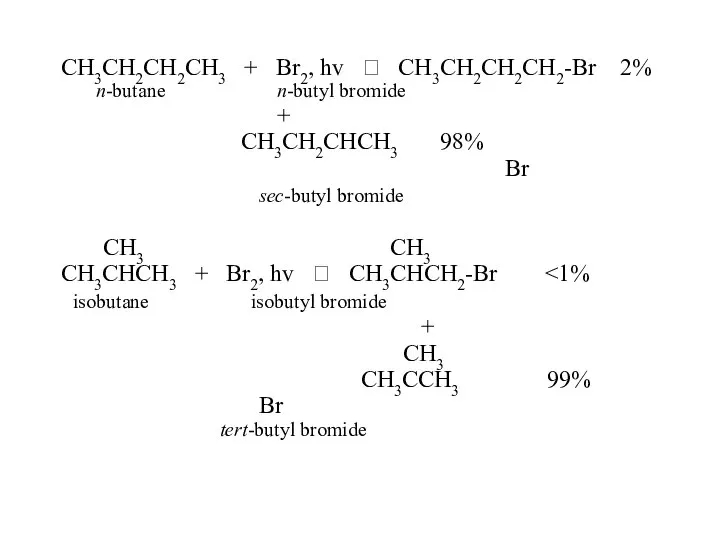
- 47. CH3CH2CH2CH3 + Br2, hv ? CH3CH2CH2CH2-Br 2% n-butane n-butyl bromide + CH3CH2CHCH3 98% Br sec-butyl bromide
- 48. In the reaction of alkanes with halogens, bromine is less reactive but more selective. Why? How?
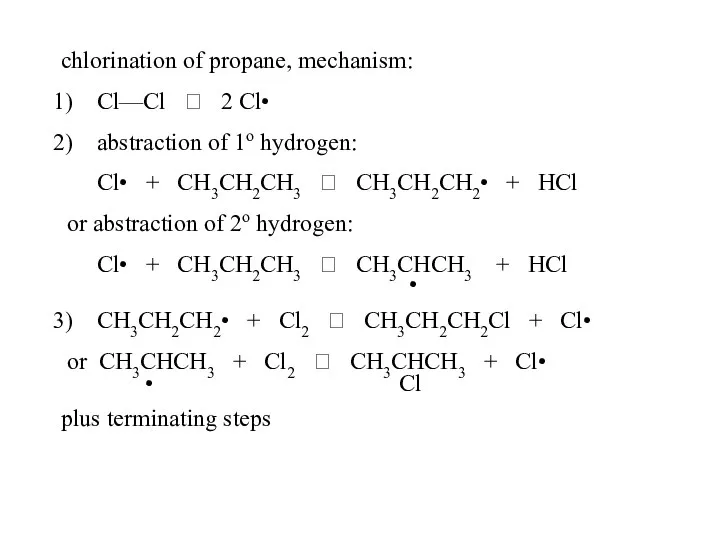
- 49. chlorination of propane, mechanism: Cl—Cl ? 2 Cl• abstraction of 1o hydrogen: Cl• + CH3CH2CH3 ?
- 50. 2) abstraction of 1o hydrogen: Cl• + CH3CH2CH3 ? CH3CH2CH2• + HCl or abstraction of 2o
- 51. The number of hydrogens (probability factor) may also be important. CH3CH2CH2CH3 + Cl2, hv ? CH3CH2CH2CH2-Cl
- 52. CH3 CH3 CH3 CH3CHCH3 + Cl2, hv ? CH3CHCH2-Cl + CH3CCH3 isobutane Cl isobutyl chloride =
- 53. Relative reactivity in bromination: 3o : 2o : 1o = 1600 : 82 : 1 In
- 54. Why is relative reactivity of H: 3o > 2o > 1o ? CH3—H ? CH3• +
- 55. Relative reactivity in halogenation: Stability of free radicals: Ease of formation of free radicals: Ease of
- 56. Halogenation R-H + X2, heat or hv ? R-X + HX a) heat or light required
- 58. Скачать презентацию





















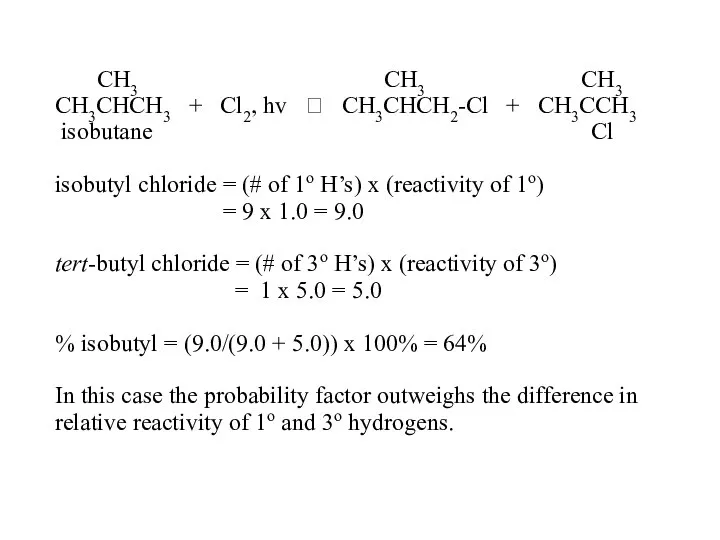




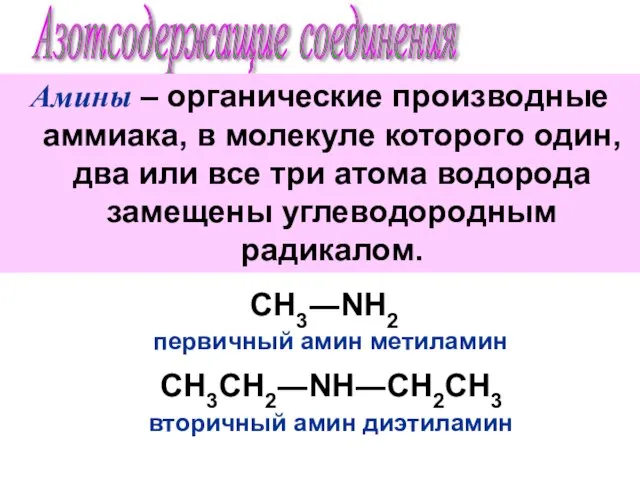 Азотсодержащие соединения. Амины
Азотсодержащие соединения. Амины Особенности строения, реакционной способности и методы синтеза азотсодержащих соединений
Особенности строения, реакционной способности и методы синтеза азотсодержащих соединений Аттестационная работа. Выращивание кристаллов. Выявить условия, позволяющие из раствора медного купороса выращивать кристаллы
Аттестационная работа. Выращивание кристаллов. Выявить условия, позволяющие из раствора медного купороса выращивать кристаллы Электродные потенциалы. Окислительно-восстановительные потенциалы. Потенциометрия в медицинской практике
Электродные потенциалы. Окислительно-восстановительные потенциалы. Потенциометрия в медицинской практике Адсорбционные равновесия и процессы на подвижных и неподвижных границах раздела фаз
Адсорбционные равновесия и процессы на подвижных и неподвижных границах раздела фаз Стероиды. Дицетин-холестерин-ацилтрансферазная реакция
Стероиды. Дицетин-холестерин-ацилтрансферазная реакция Каучуки. Гума
Каучуки. Гума Мое любимое химическое вещество: Серебро
Мое любимое химическое вещество: Серебро Специальный выпуск Слово эксперту. Антиоксидантная защита от Орифлэйм
Специальный выпуск Слово эксперту. Антиоксидантная защита от Орифлэйм Теоретические основы качественного анализа
Теоретические основы качественного анализа Хлор
Хлор Карбоновые кислоты. Модели молекул
Карбоновые кислоты. Модели молекул Химия вокруг нас
Химия вокруг нас Fuel cells
Fuel cells Алканы. Строение алканов
Алканы. Строение алканов Биополимеры Диктант
Биополимеры Диктант Предмет и задачи химии. Вещества и их свойства (продолжение)
Предмет и задачи химии. Вещества и их свойства (продолжение) Электрический ток в растворах и расплавах электролитов. Электролиз
Электрический ток в растворах и расплавах электролитов. Электролиз Химия неметаллов. Урок № 77
Химия неметаллов. Урок № 77 Химический состав клетки. Нуклеиновые кислоты. ДНК
Химический состав клетки. Нуклеиновые кислоты. ДНК Презентация по Химии "Соли в медицине" - скачать смотреть
Презентация по Химии "Соли в медицине" - скачать смотреть  Коллагеноздар биохимиясы
Коллагеноздар биохимиясы Изучение физико-химических свойств яблок разных сортов и влияние их на здоровье школьников
Изучение физико-химических свойств яблок разных сортов и влияние их на здоровье школьников Обмен липидов-1
Обмен липидов-1 Шампуни: классификация по составу и назначению
Шампуни: классификация по составу и назначению Электролитическая диссоциация_
Электролитическая диссоциация_ Ксенобиотики в окружающей среде и живых организмах. (Лекция 2)
Ксенобиотики в окружающей среде и живых организмах. (Лекция 2) Органикалық қосылыстар. Көмірсутектерден жасалған
Органикалық қосылыстар. Көмірсутектерден жасалған