Содержание
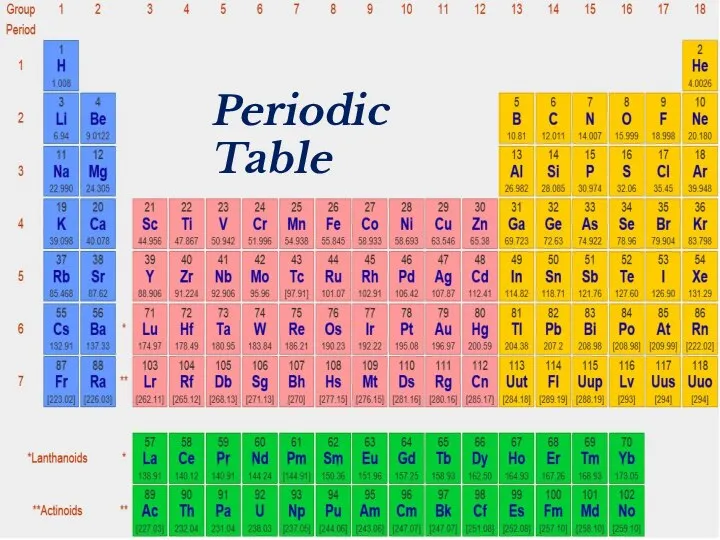
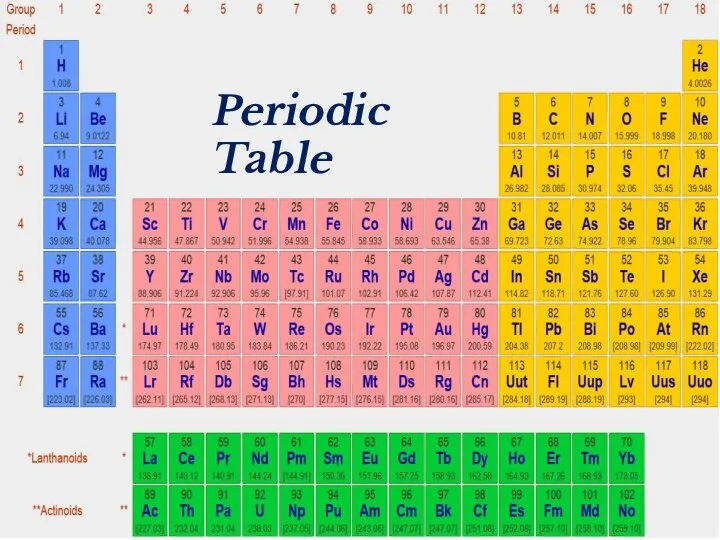
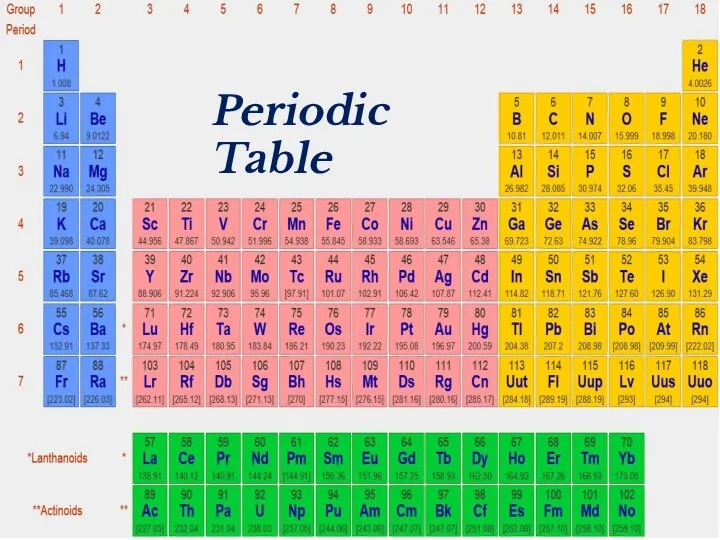
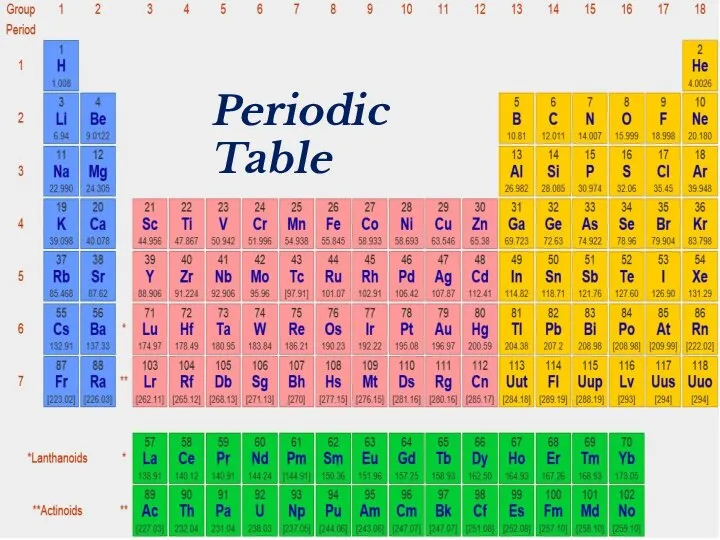
- 2. Periodic Table
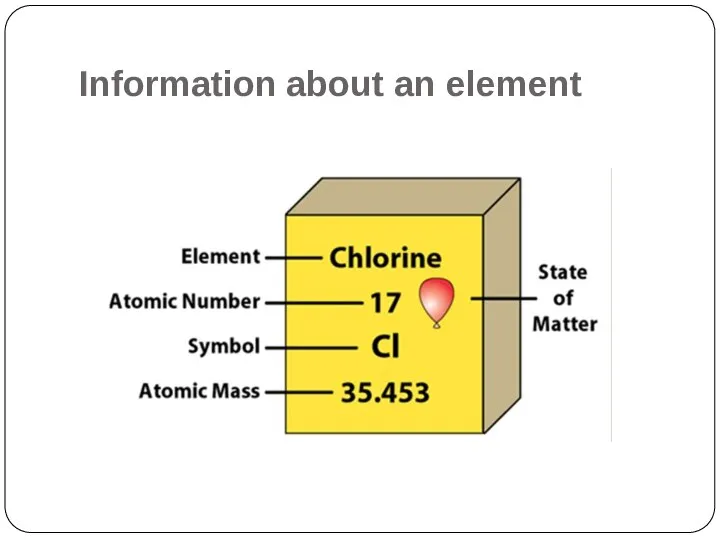
- 3. Information about an element
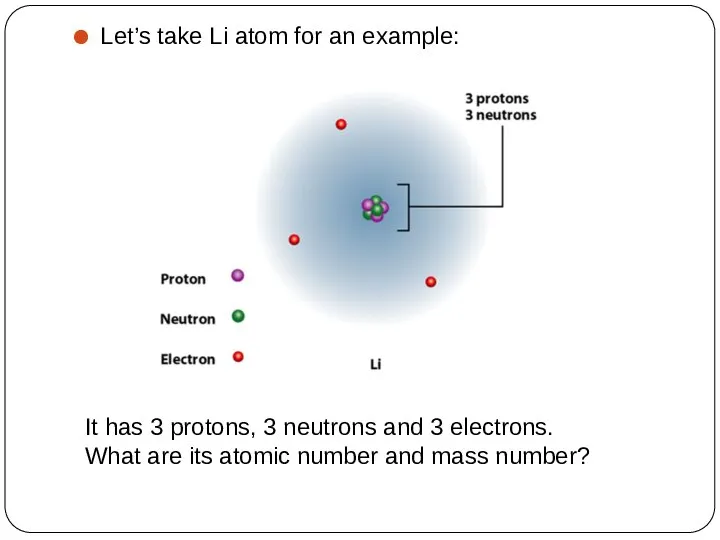
- 4. Let’s take Li atom for an example: It has 3 protons, 3 neutrons and 3 electrons.
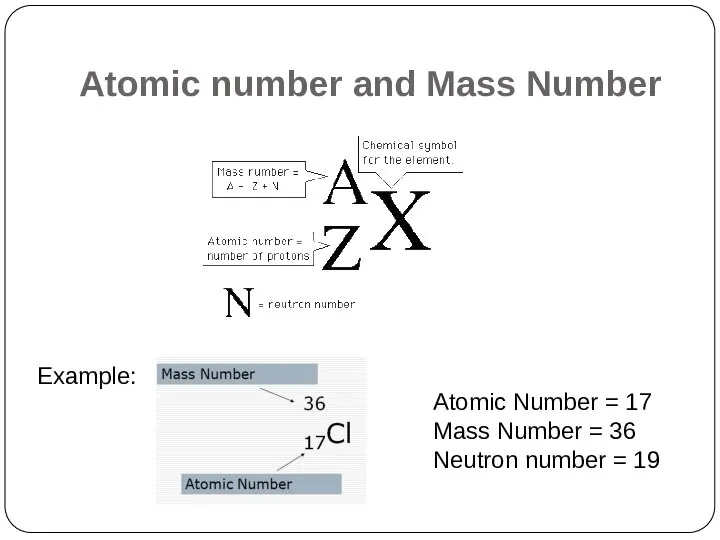
- 5. Atomic number and Mass Number The number of protons in a given atom is called the
- 6. Atomic number and Mass Number Example: Atomic Number = 17 Mass Number = 36 Neutron number
- 7. Number of electrons An atom is neutral. The net charge is zero. Therefore, Number of protons
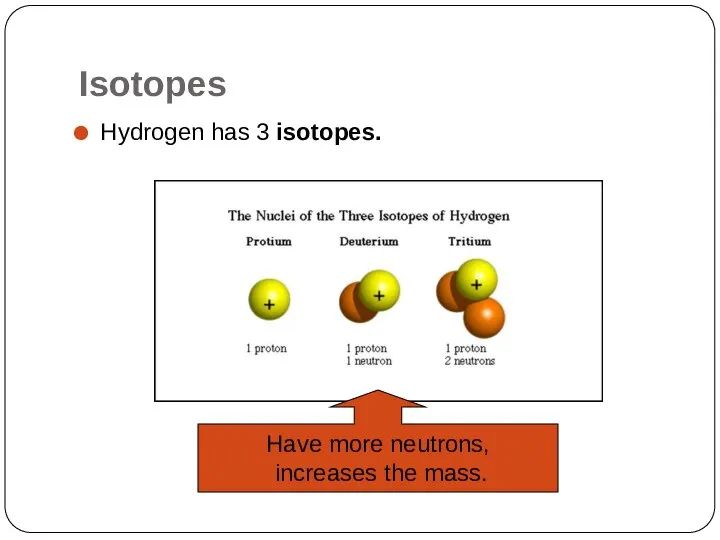
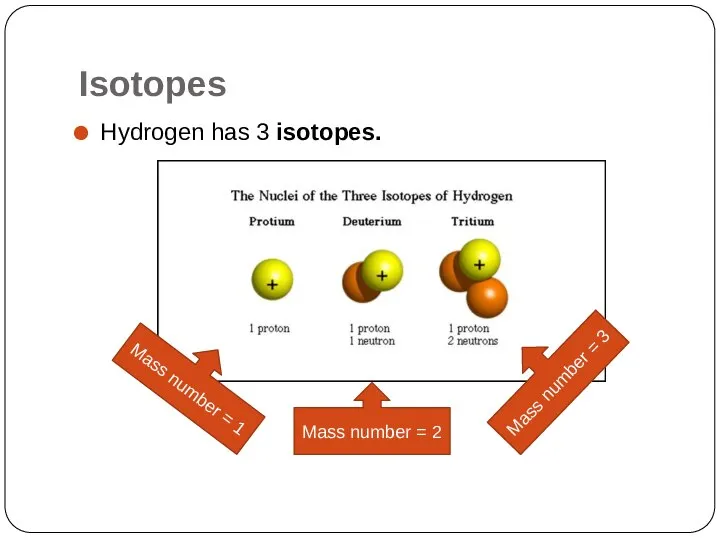
- 13. Isotopes同位素 They are atoms of the same element that have different numbers of neutrons. Hydrogen has
- 14. Isotopes Hydrogen has 3 isotopes. Have more neutrons, increases the mass.
- 15. Isotopes Hydrogen has 3 isotopes. Mass number = 1 Mass number = 2 Mass number =
- 16. 6 protons, 8 (14 - 6) neutrons, 6 electrons 6 protons, 5 (11 - 6) neutrons,
- 18. Periodic Table
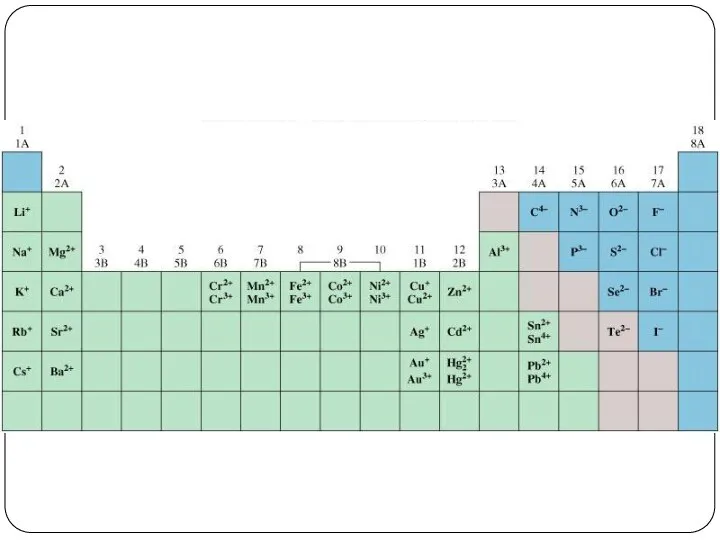
- 23. Cation and Anion An ion is an atom, or group of atoms, that has a net
- 24. Cation and Anion A monatomic ion contains only one atom eg. Na+, Cl-, Ca2+, O2-, Al3+,
- 25. 13 protons, 10 (13 – 3) electrons 34 protons, 36 (34 + 2) electrons Do You
- 28. ***
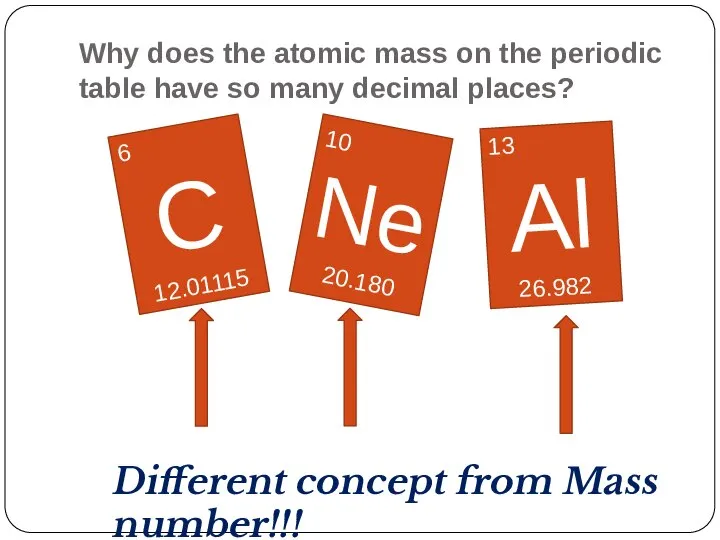
- 30. Why does the atomic mass on the periodic table have so many decimal places? 6 C
- 31. Atomic mass 1 H 1.00797 This atomic mass is the one number that best represents the
- 32. Calculating a “weighted” average 1. First, you have to know the mass of each isotope. 2.
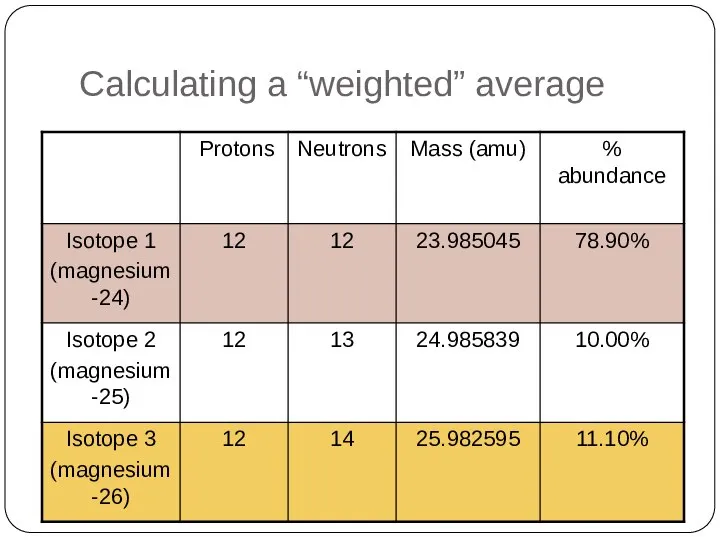
- 33. Calculating a “weighted” average A sample of magnesium collected anywhere in the universe will contain three
- 34. Calculating a “weighted” average
- 35. Calculating a “weighted” average Atomic mass of magnesium = 23.985045 x 78.90% + 24.985839 x 10.00%
- 36. For your reference
- 37. Cl-35 is about 75.5 % and Cl-37 about 24.5% of natural chlorine. Another example This atomic
- 38. Exercise Copper has two isotopes 63Cu (62.9 amu) and 65Cu (64.9 amu). What is the %
- 39. Periodic Table
- 41. Periodic Table
- 43. Radioactive isotope Radioactive isotope, also called radioisotope, any of several species of the same chemical element
- 45. Скачать презентацию





























 Аттестационная работа. Музей минералов и горных пород
Аттестационная работа. Музей минералов и горных пород Алканы. Свойства. Строение и применение
Алканы. Свойства. Строение и применение Презентация исследовательской работы на тему: «Витамин С и его значение» Ученика 8а класса Чипизубова Дениса ЖМСОШ№1
Презентация исследовательской работы на тему: «Витамин С и его значение» Ученика 8а класса Чипизубова Дениса ЖМСОШ№1 Сера и её соединения
Сера и её соединения Изомерия. Типы изомерии
Изомерия. Типы изомерии Использование проектного метода при изучении химии
Использование проектного метода при изучении химии Природные биоактивные соединения (БИС)
Природные биоактивные соединения (БИС) Структура електронної оболонки атома
Структура електронної оболонки атома 10 смыслов картины Черный квадрат
10 смыслов картины Черный квадрат  Липидтердің метаболизмі
Липидтердің метаболизмі Оборотные средства предприятия по переработке пластических масс и эластомеров
Оборотные средства предприятия по переработке пластических масс и эластомеров Углеводы. Строение углеводов
Углеводы. Строение углеводов Звездный час по теме «Карбоновые кислоты»
Звездный час по теме «Карбоновые кислоты» Гидрогенизационные процессы переработки нефти и газа
Гидрогенизационные процессы переработки нефти и газа Поверхневі явища. Адсорбція
Поверхневі явища. Адсорбція Путешествие по континенту Химия познавательная игра по химии, 8 класс Автор: Лаврентьева Снежана Павловна, учитель химии и би
Путешествие по континенту Химия познавательная игра по химии, 8 класс Автор: Лаврентьева Снежана Павловна, учитель химии и би Солюбилизация в прямых мицеллах ПАВ
Солюбилизация в прямых мицеллах ПАВ Резина. Свойства резины
Резина. Свойства резины Химическая связь
Химическая связь Степень окисления. Бинарные соединения
Степень окисления. Бинарные соединения Тема проекту: “ЕКОЛОГІЧНІ ПРОБЛЕМИ ЛЮДСТВА - радіоактивне забруднення” «Природа не терпить жартів. Вона завжди спокійна, завжди серйозна, завжди має рацію. Помилок припускається лише людина...» (Й.В.Гете).
Тема проекту: “ЕКОЛОГІЧНІ ПРОБЛЕМИ ЛЮДСТВА - радіоактивне забруднення” «Природа не терпить жартів. Вона завжди спокійна, завжди серйозна, завжди має рацію. Помилок припускається лише людина...» (Й.В.Гете). Определение гомологического ряда алканов
Определение гомологического ряда алканов Растворы. Процесс растворения
Растворы. Процесс растворения Неметалічні елементи
Неметалічні елементи Химия в системе современных естественнонаучных представлений
Химия в системе современных естественнонаучных представлений Кремний и его родственники
Кремний и его родственники Направление окислительно-восстановительного процесса
Направление окислительно-восстановительного процесса Аминокислоты. Физические свойства
Аминокислоты. Физические свойства