Содержание
- 2. Picture of the Atom Electromagnetic radiation and Atomic Spectra The Nature of Electron and Atomic Orbitals
- 3. 3.1.1 Atomic concept, 3.1.2 Subatomic particles, 3.1.3 Atomic structure: first ideas Part I
- 4. Dalton Atomic Theory Elements are made of tiny particles called atoms 2. The atoms of a
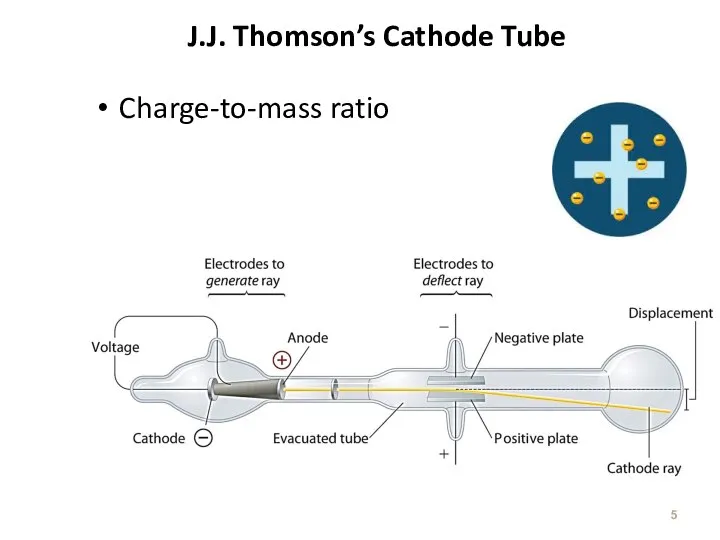
- 5. J.J. Thomson’s Cathode Tube Charge-to-mass ratio
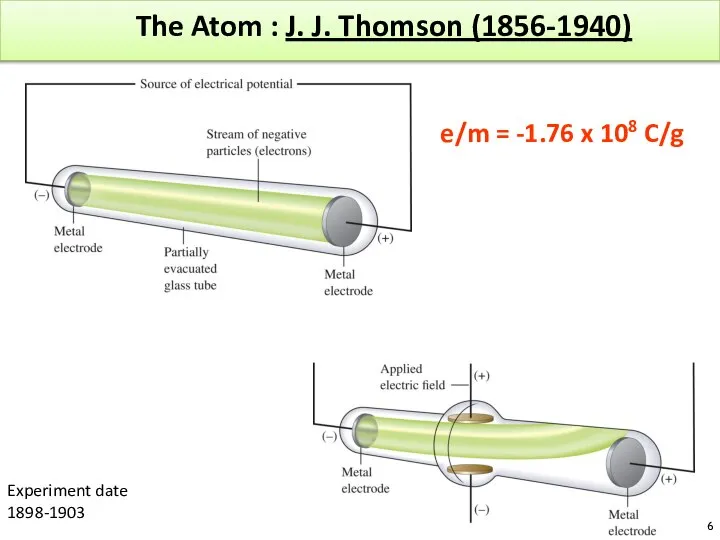
- 6. The Atom : J. J. Thomson (1856-1940) e/m = -1.76 x 108 C/g Experiment date 1898-1903
- 7. The Atom based on Thomson’s experiment A ray of particles is produced between two metallic electrodes.
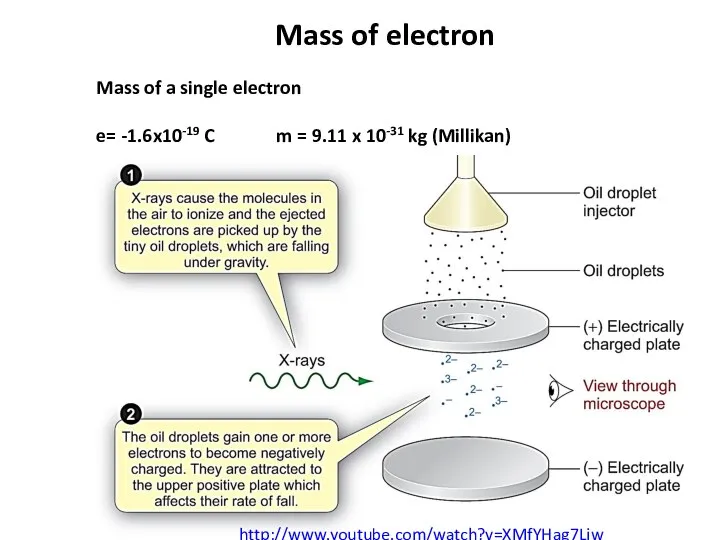
- 8. Mass of electron Mass of a single electron e= -1.6x10-19 C m = 9.11 x 10-31
- 9. Rutherford Experiment Ernest Rutherford – 1911 With Thomson Model : a particles should travel through the
- 10. Rutherford Experiment
- 11. The Nucleus Ernest Rutherford – 1911 Conclusion : Dense positive center with electrons far from the
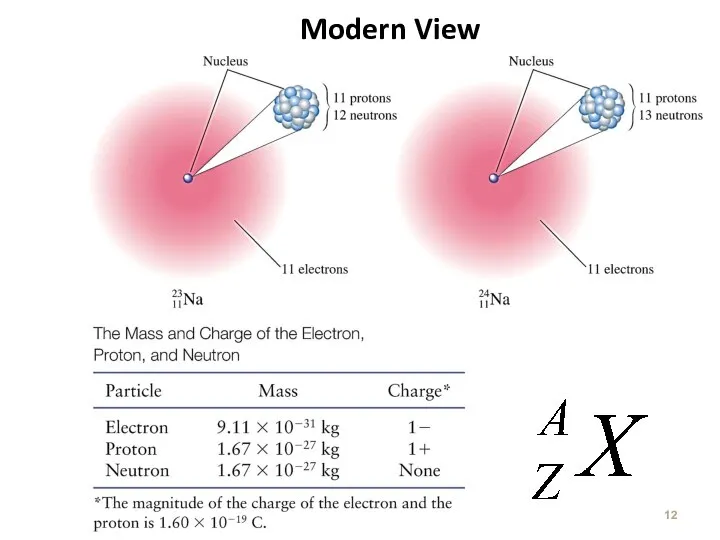
- 12. Modern View
- 13. 3.2. Electromagnetic Radiation and Quantization 3.2.1: Electromagnetic Radiation 3.2.2: Quantization 3.2.3: The Atomic Spectrum of Hydrogen
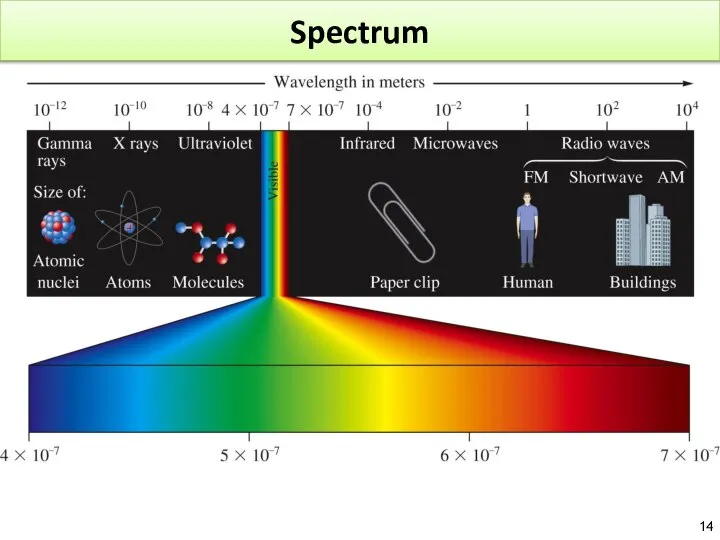
- 14. Spectrum
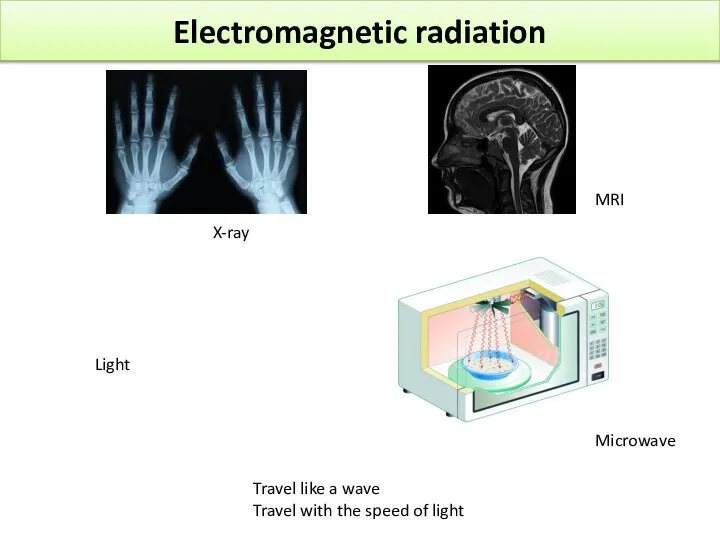
- 15. Electromagnetic radiation Light X-ray MRI Microwave Travel like a wave Travel with the speed of light
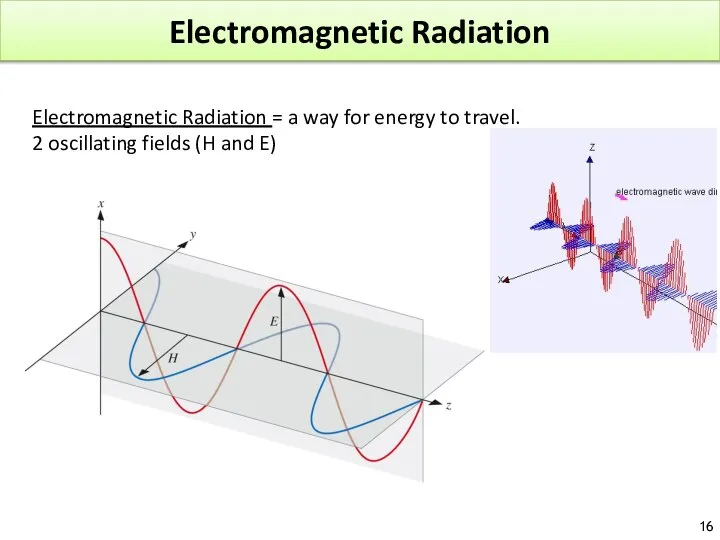
- 16. Electromagnetic Radiation Electromagnetic Radiation = a way for energy to travel. 2 oscillating fields (H and
- 17. ELECTROMAGNETIC RADIATION
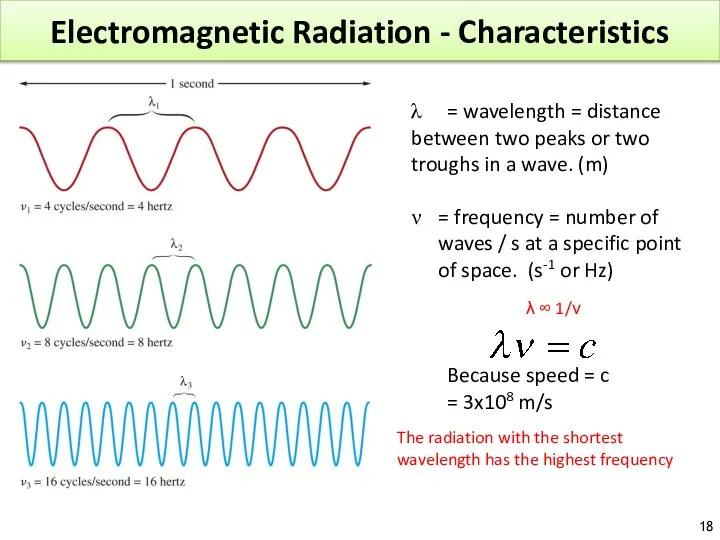
- 18. Electromagnetic Radiation - Characteristics λ = wavelength = distance between two peaks or two troughs in
- 19. Radio in the 909kHz. What wavelength does it correspond to? λ = c/ν = 330 m
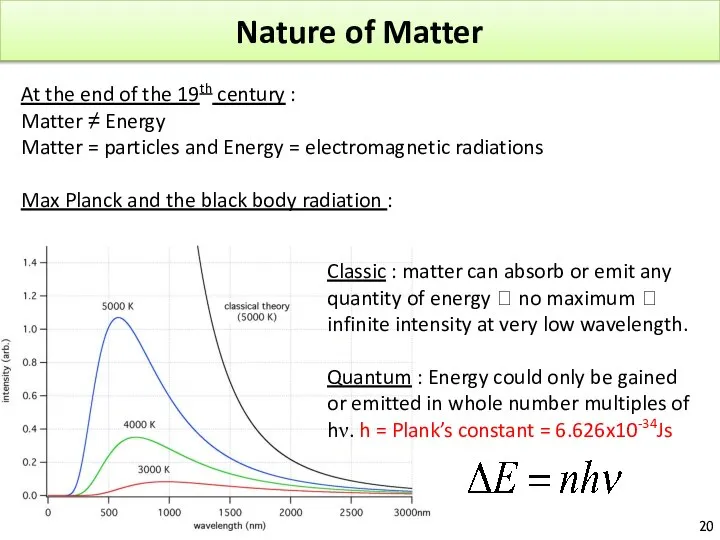
- 20. Nature of Matter At the end of the 19th century : Matter ≠ Energy Matter =
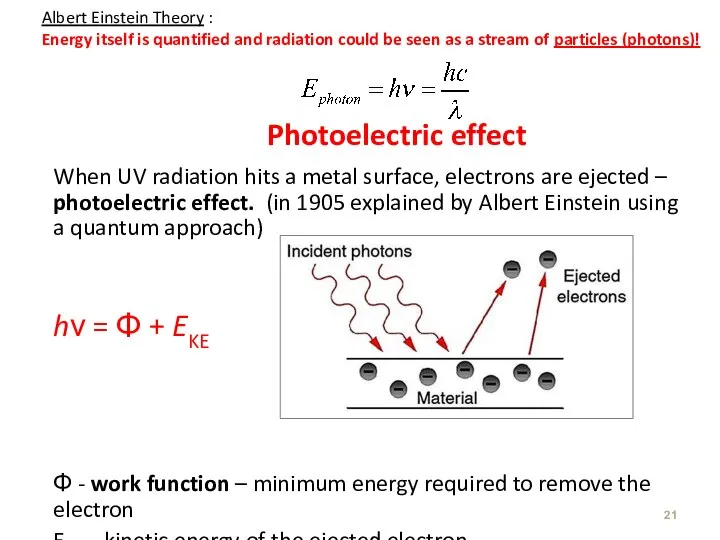
- 21. Photoelectric effect When UV radiation hits a metal surface, electrons are ejected – photoelectric effect. (in
- 22. E = h x ν E = 6.63 x 10-34 (J•s) x 3.00 x 10 8
- 23. Dual Nature of Light Energy – Mass relationship : A particle but also a wave :
- 24. De Broglie 1924 λ = h/mν λ Proportional to h/mν H :Planck Constant M : masse
- 25. Diffraction What is the wavelength for an electron? Me = 9.11x10-31 kg Ve = 1.0x107 m/s
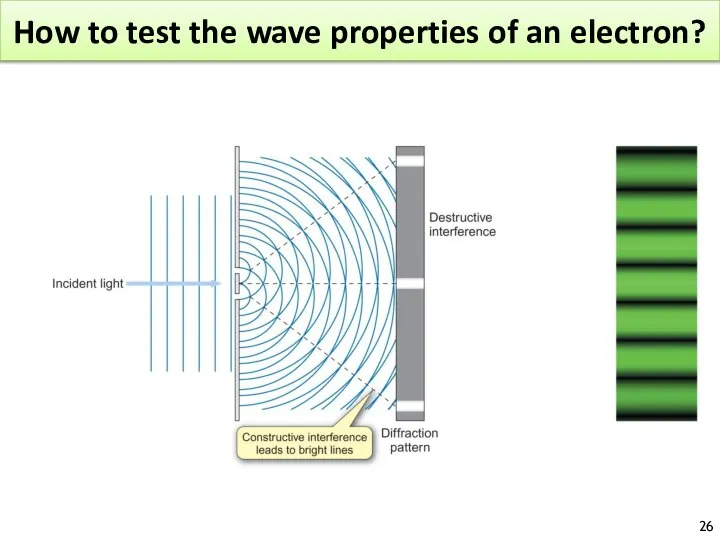
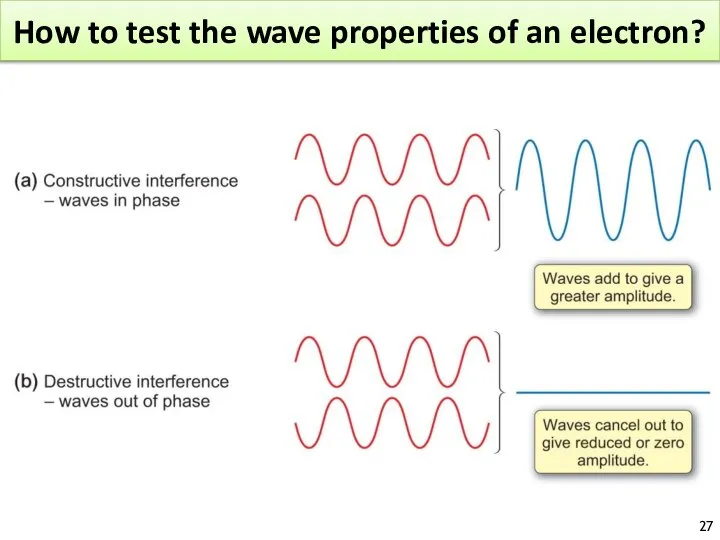
- 26. How to test the wave properties of an electron?
- 27. How to test the wave properties of an electron?
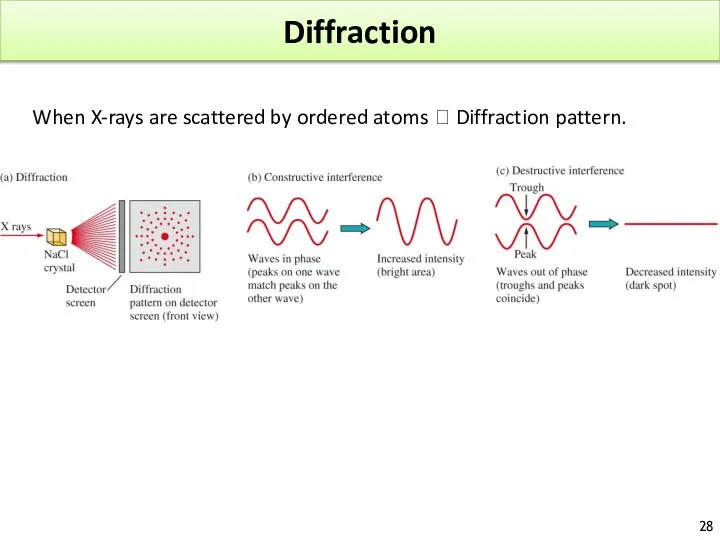
- 28. Diffraction When X-rays are scattered by ordered atoms ? Diffraction pattern.
- 29. Conclusion All matter exhibits both particulate and wave properties. Large particles : mainly particle Small particles
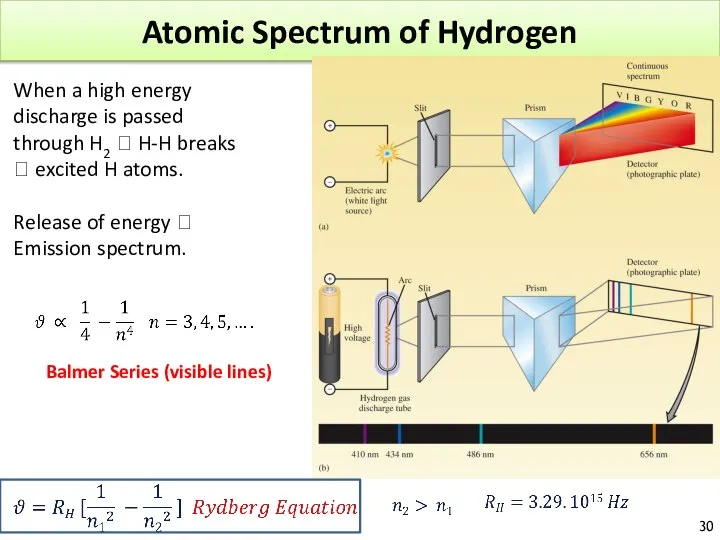
- 30. Atomic Spectrum of Hydrogen When a high energy discharge is passed through H2 ? H-H breaks
- 31. Table 3.4. The atomic spectrum of hydrogen
- 33. Atomic Spectrum of Hydrogen Why do we have a line spectrum for H ? Only certain
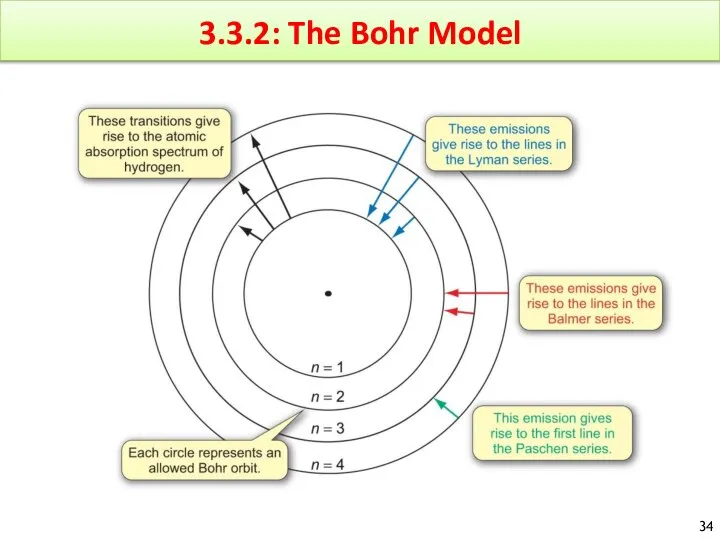
- 34. 3.3.2: The Bohr Model
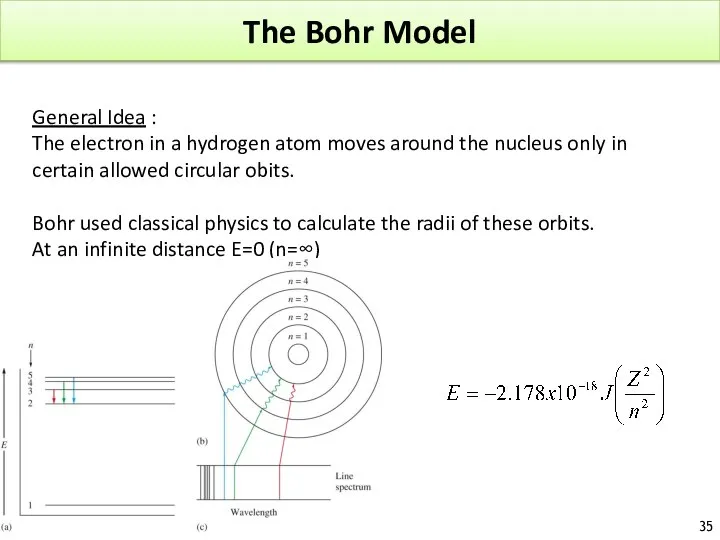
- 35. The Bohr Model General Idea : The electron in a hydrogen atom moves around the nucleus
- 36. The Bohr Model Example : Energy emitted from n=6 to ground state : The negative sign
- 37. Wave Function and Atomic Orbitals 3.5.1 Wave properties of matter, Heisenberg uncertainty principle 3.5.2 Wave-functions and
- 38. De Broglie All moving particles have wave properties λ= h mu = Wavelength h = Planck
- 39. 2.2 SCHRONDINGER EQUATION Enter
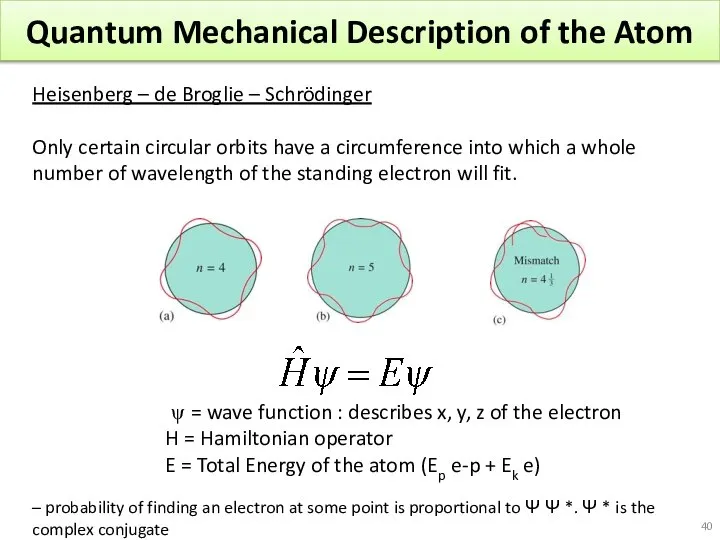
- 40. Quantum Mechanical Description of the Atom Heisenberg – de Broglie – Schrödinger Only certain circular orbits
- 41. The Schrödinger equation The probability distributions and allowed energy levels for electrons in atoms and molecules
- 42. Schrodinger Wave Equation
- 43. Kinetic Energy of the Electron Motion Potential Energy of the Electron. The result of electrostatic attraction
- 44. Kinetic Energy Potential Energy
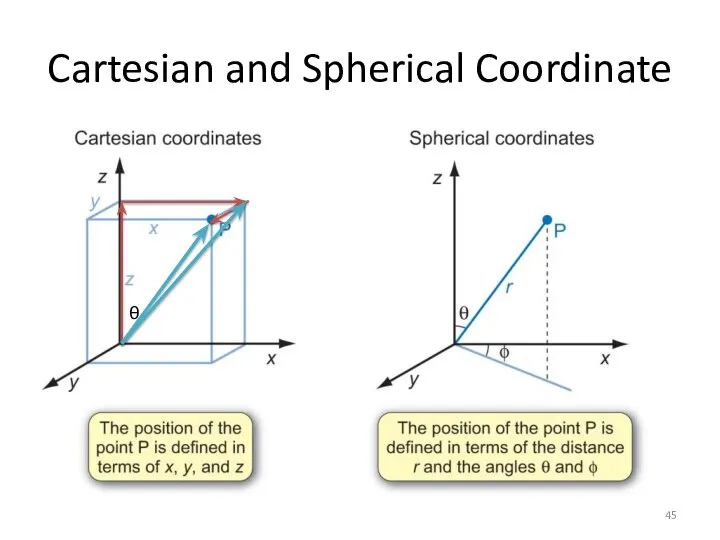
- 45. Cartesian and Spherical Coordinate
- 46. The wavefunction Atomic wavefunctions are usually expressed in spherical polar coordinates – give value of Ψ
- 47. Homework-2 Please solve problems ; Chapter 3 6, 9, 10, 12, 14, 16 and 17 Due
- 48. Wave Equation for the Hydrogen Atom – R(r) is radial part of wavefunction Describes electrons density
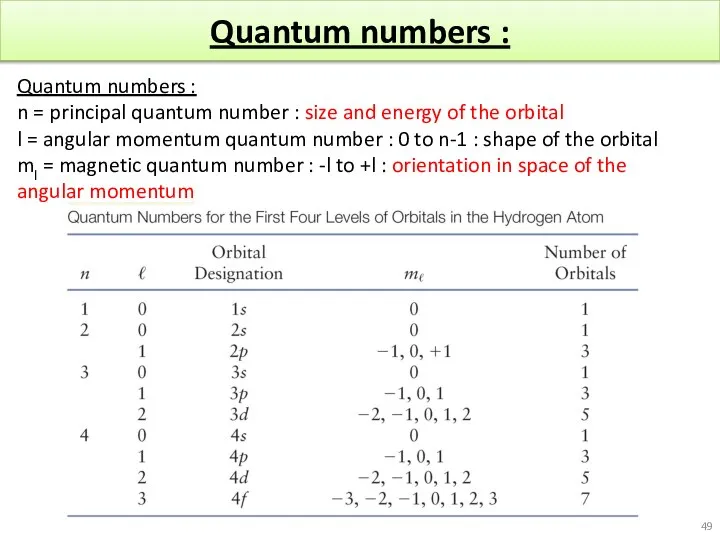
- 49. Quantum numbers : Quantum numbers : n = principal quantum number : size and energy of
- 50. Radial and Angular Wave Function for 1s derived from Schrodinger Equation
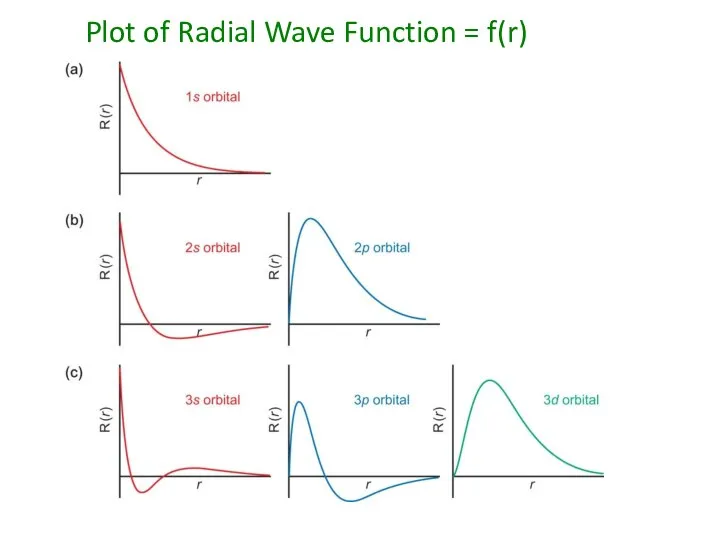
- 51. Plot of Radial Wave Function = f(r)
- 52. s orbitals Size : 1s Energy : 1s Surface of 0 probability = nodal surface /
- 53. Physical Meaning of Orbitals The wave function has no easy physical meaning. The square of the
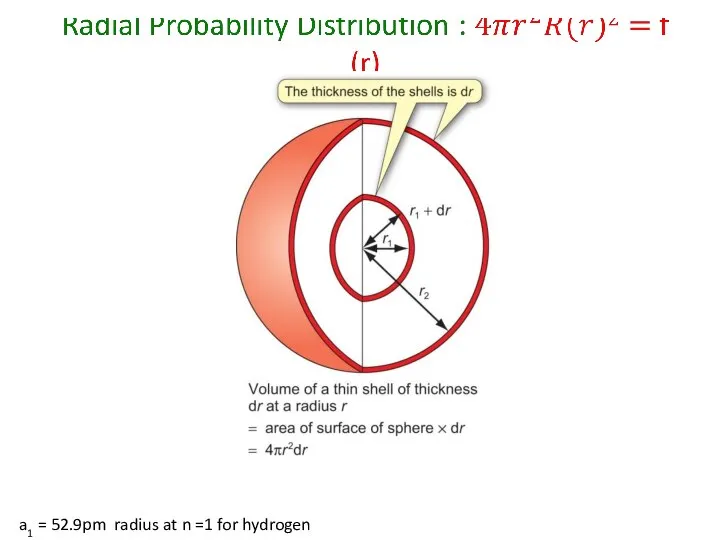
- 54. a1 = 52.9pm radius at n =1 for hydrogen
- 55. a1 = 52.9pm radius at n =1 for hydrogen
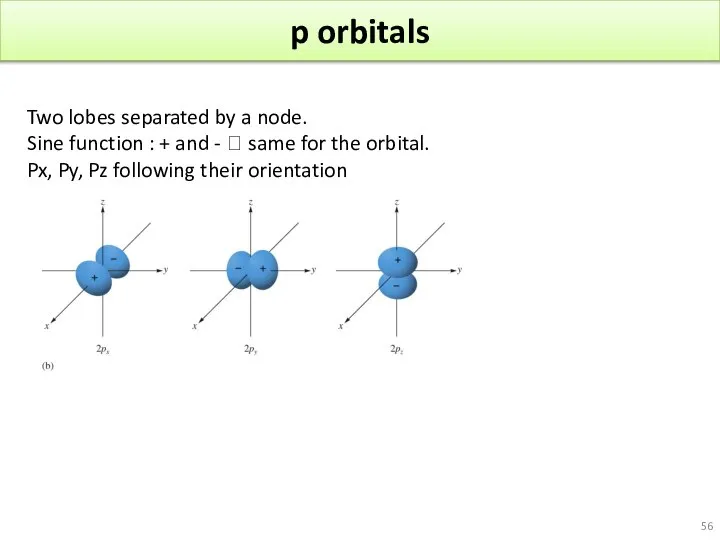
- 56. p orbitals Two lobes separated by a node. Sine function : + and - ? same
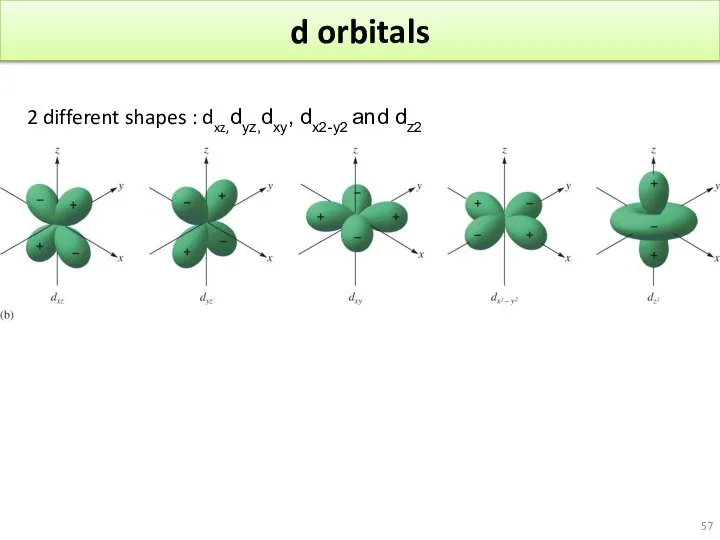
- 57. d orbitals 2 different shapes : dxz,dyz,dxy, dx2-y2 and dz2
- 58. f orbitals Very complex shapes
- 59. Schrödinger Equation Each solution ψ of the Schrödinger equation has a specific value for E. A
- 60. Heisenberg uncertainty principle There is a fundamental limitation to just how precisely we can know both
- 61. The Hydrogen Atom : summary The quantum mechanical model : electron = wave Series of wave
- 62. Polyelectronic Model Schrödinger equation can be solved exactly only for hydrogen. Schrödinger equation cannot be solved
- 63. Self-Consistent Field Method http://www.youtube.com/watch?v=UVkTuOwfOh0
- 64. https://www.youtube.com/watch?v=A6DiVspoZ1E Review this link at home
- 65. Many Electron Atoms Electron spin, Aufbau principle, Anomalies in electronic configuration, Structure of Periodic table Part
- 66. Electron Spin and Pauli Principle A 4th quantum number describe the electron : ms : electron
- 67. History of the Periodic Table Dmitri Mendeleev : ми́трий Менделе́ев One of first to arrange known
- 68. The Aufbau Principle Principle to populate orbitals.
- 69. Valence electrons Valence electrons = electrons from the outermost principal quantum level of an atom. Group
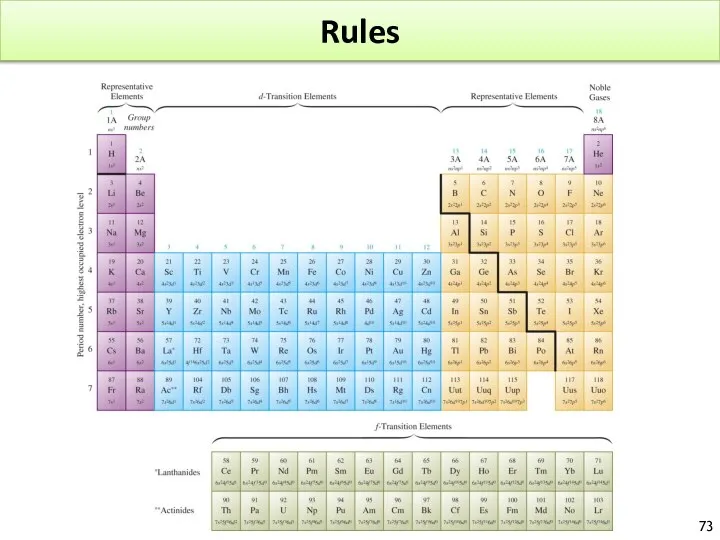
- 70. Rules
- 71. Rules After 4s2, we fill 3d. Mn : [Ar]4s23d5 – Fe [Ar]4s23d6 Additional Rules: The (n+1)
- 72. Rules Element above 118 are generally unstable G contain 9 orbitals l = n-1 = 4
- 73. Rules
- 74. Hund’s Rule The lowest energy configuration for an atom is the one having the maximum number
- 75. Pauli Exclusion Principle Pauli Exclusion principle ; no two electrons in an atom can have the
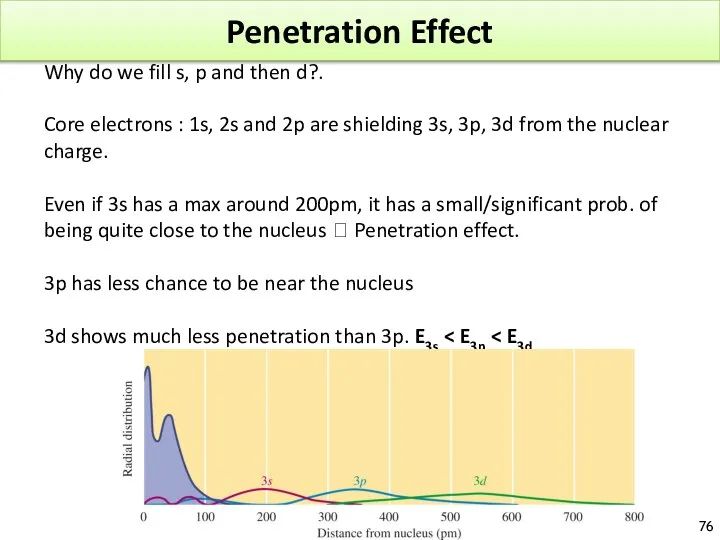
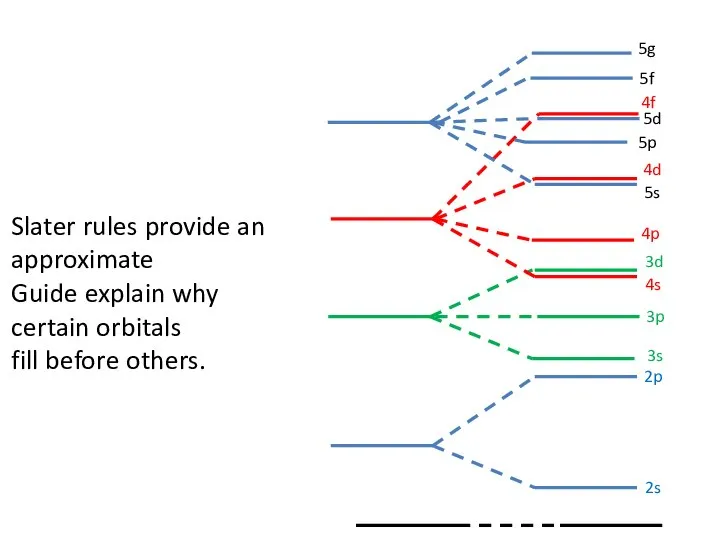
- 76. Penetration Effect Why do we fill s, p and then d?. Core electrons : 1s, 2s
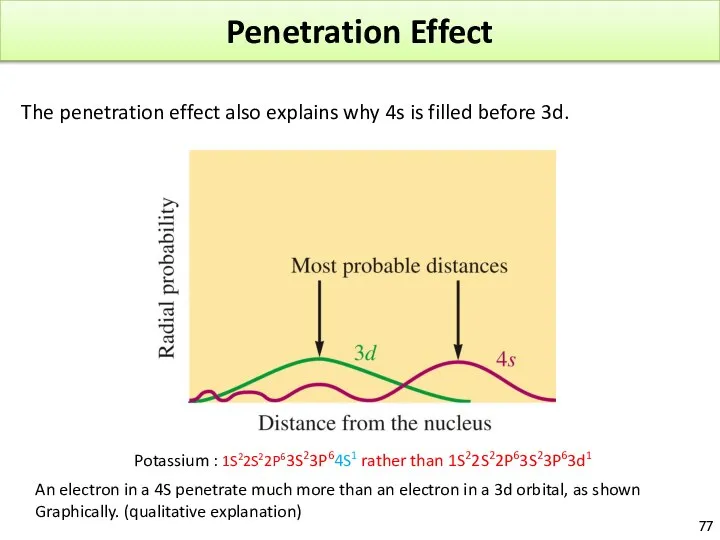
- 77. Penetration Effect The penetration effect also explains why 4s is filled before 3d. Potassium : 1S22S22P63S23P64S1
- 78. 4s 5g 5s 3p 3s 2p 2s Slater rules provide an approximate Guide explain why certain
- 79. https://en.wikipedia.org/wiki/Effective_nuclear_charge
- 80. Slater’s Rules The rules were devised semi-empirically by John C. Slater and published in 1930 Identify
- 81. Slater’s Rules The rules were devised semi-empirically by John C. Slater and published in 1930 Rules
- 82. Slater’s Rules for determining S for a specific electron The shielding constant (S) ns and np
- 83. Slater’s Rules for determining S for a specific electron Rule -3b: Each electron in n-1 group
- 84. Z* for Na = Z – S = 11 – 8.8 = 2.2
- 85. Slater’s Rules for determining S for a specific electron Rule -4a: Each electron in nd and
- 86. Nickel Use slater rules to calculate the shielding constant S and effective nuclear charge of 3d
- 87. Solution Rule-1 : the electron configuration is written using slater’s groupings: (1s2)(2s2, 2p6)(3s2, 3p6)(3d8)(4s2) To calculate
- 88. Solution Rule-1 : the electron configuration is written using slater’s groupings: (1s2)(2s2, 2p6)(3s2, 3p6)(3d8) (4s2) To
- 89. Comparison of The effective nuclear charge 3d electrons The effective nuclear charge Z*=28-20.45=7.55 4s electrons Z*=28-23.95=4.05
- 90. Periodic Properties of Atoms : Ionization Energy Ionization Energy : Energy required to remove an electron
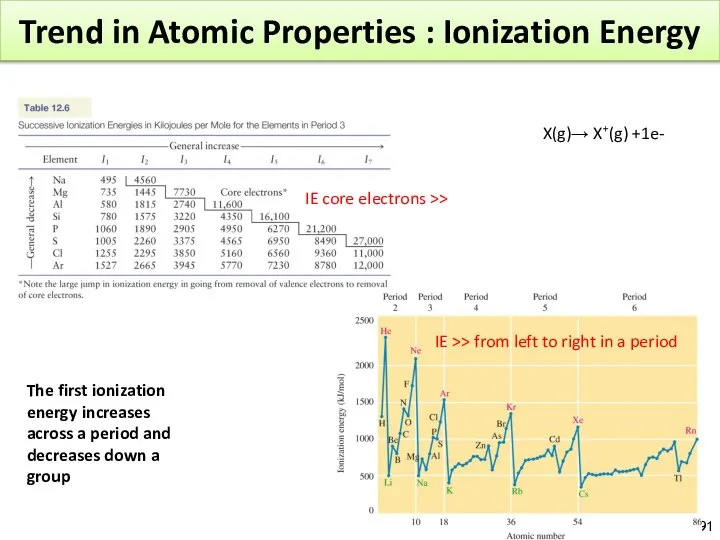
- 91. Trend in Atomic Properties : Ionization Energy IE core electrons >> IE >> from left to
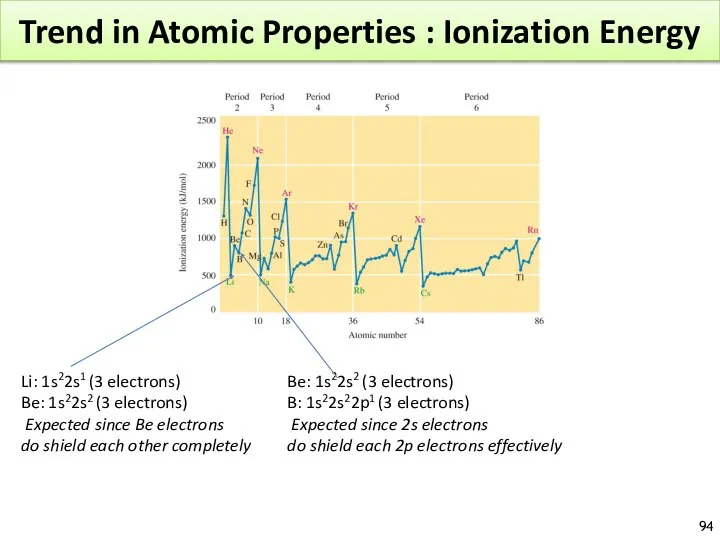
- 94. Trend in Atomic Properties : Ionization Energy Li: 1s22s1 (3 electrons) Be: 1s22s2 (3 electrons) Expected
- 95. Trend in Atomic Properties : Ionization Energy IE goes down along a group. The removed electron
- 96. Trend in Atomic Properties : Atomic Radius Atomic Radius: half the distance between the nuclei in
- 97. END END
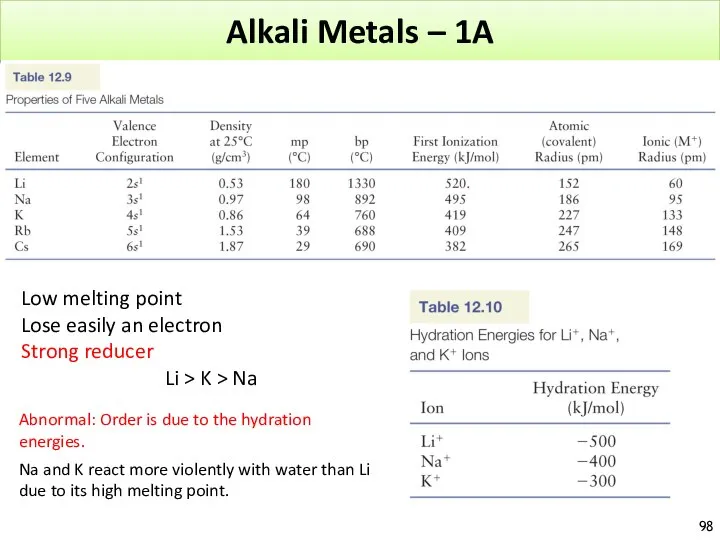
- 98. Alkali Metals – 1A Low melting point Lose easily an electron Strong reducer Li > K
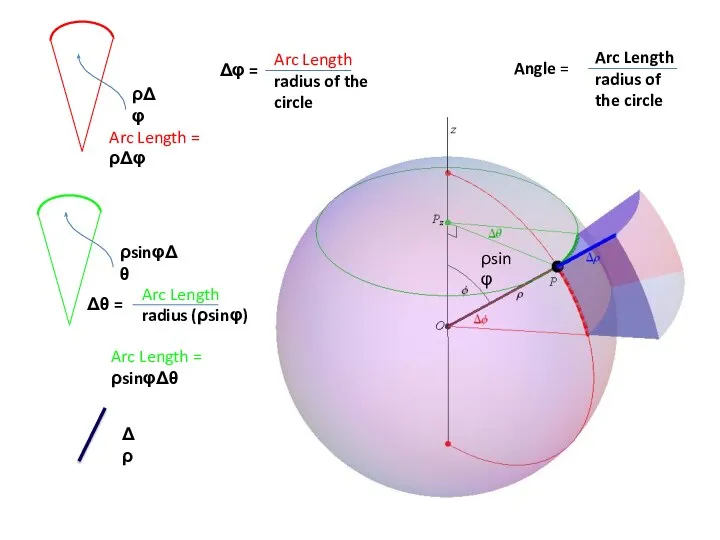
- 99. ρsinφ Δρ Angle = Arc Length radius of the circle
- 102. Скачать презентацию












































![Rules After 4s2, we fill 3d. Mn : [Ar]4s23d5 – Fe](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/620409/slide-70.jpg)





















 Получение армированных фотополимерных композиций
Получение армированных фотополимерных композиций Идеальные и реальные кристаллы
Идеальные и реальные кристаллы Методы экстрагирования ЛРС и оборудование
Методы экстрагирования ЛРС и оборудование Общая характеристика углерода
Общая характеристика углерода Портфолио Учителя химии
Портфолио Учителя химии Химическая кинетика
Химическая кинетика Генетическая связь веществ. Виды реакций
Генетическая связь веществ. Виды реакций Классификация химических реакций
Классификация химических реакций Минеральные удобрения. Для школьников
Минеральные удобрения. Для школьников Стеариновая кислота
Стеариновая кислота Игра-квест на уроке химии
Игра-квест на уроке химии Пластмассы (пластики)
Пластмассы (пластики) Биологически важные реакции монофункциональных соединений. Реакционная способность альдегидов, кетонов, карбоновых кислот
Биологически важные реакции монофункциональных соединений. Реакционная способность альдегидов, кетонов, карбоновых кислот Нафта, вугілля, природний газ як вуглеводнева сировина. Основні види палива та їх значення в енергетиці країни
Нафта, вугілля, природний газ як вуглеводнева сировина. Основні види палива та їх значення в енергетиці країни Карбон Колообіг в природі
Карбон Колообіг в природі  Сера
Сера Агрегатное состояние вещества. Газообразные вещества
Агрегатное состояние вещества. Газообразные вещества Стеклоткани. Виды стеклотканей
Стеклоткани. Виды стеклотканей Биологически важные ароматические и гетероциклические соединения
Биологически важные ароматические и гетероциклические соединения Введение в химию. 8 класс
Введение в химию. 8 класс Амфотерные неорганические и органические соединения МОУ «Гатчинская СОШ №2» Учитель химии: Г.Г.Павлова Учитель инфор
Амфотерные неорганические и органические соединения МОУ «Гатчинская СОШ №2» Учитель химии: Г.Г.Павлова Учитель инфор Бензол. Получение бензола. Химические свойства бензола. Применение бензола на основе его свойств
Бензол. Получение бензола. Химические свойства бензола. Применение бензола на основе его свойств Хром и его соединения
Хром и его соединения Значение коллодиной химии в производстве
Значение коллодиной химии в производстве Crystallography. Part 4: Crystal Forms Twinning
Crystallography. Part 4: Crystal Forms Twinning ПРЕДЕЛЬНЫЕ УГЛЕВОДОРОДЫ
ПРЕДЕЛЬНЫЕ УГЛЕВОДОРОДЫ  Биологически важные химические элементы. Неорганические соединения
Биологически важные химические элементы. Неорганические соединения Физические явления - основа разделения смесей в химии
Физические явления - основа разделения смесей в химии