Содержание
- 2. Early Methods The relative error in the activation energy as a function of the activation energy
- 3. Friedman methods Fig.1.2. The relative error in the activation energy as a function of the activation
- 4. Ozawa, and Flynn and Wall Fig. 1.3. The activation energies determined by Friedman for the thermal
- 5. Modern Methods (Vyazovkin) Fig 1.4 Relative error in the activation energy as a function of x=
- 7. Скачать презентацию
Слайд 2
Early Methods
The relative error in the activation energy as a
Early Methods
The relative error in the activation energy as a
function of the activation energy and the distance between the initial temperature ( T0 ) and temperature of a given conversion ( Tf1) at the slowest heating rate β1 . (Reproduced from Starink [18] with permission of Springer)
Слайд 3
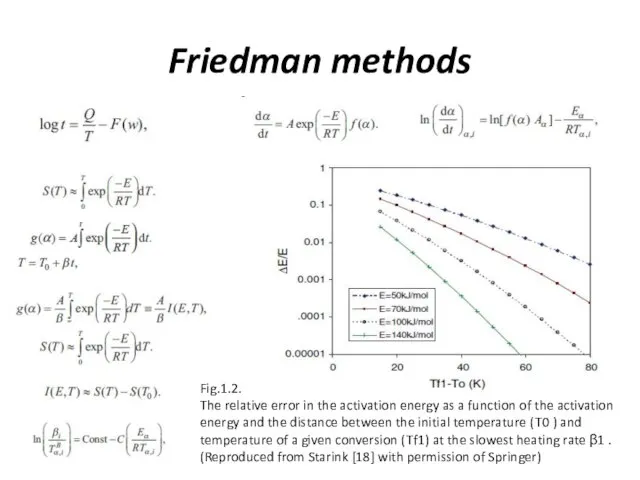
Friedman methods
Fig.1.2.
The relative error in the activation energy as a function
Friedman methods
Fig.1.2.
The relative error in the activation energy as a function
of the activation energy and the distance between the initial temperature ( T0 ) and temperature of a given conversion ( Tf1) at the slowest heating rate β1 . (Reproduced from Starink [18] with permission of Springer)
Слайд 4
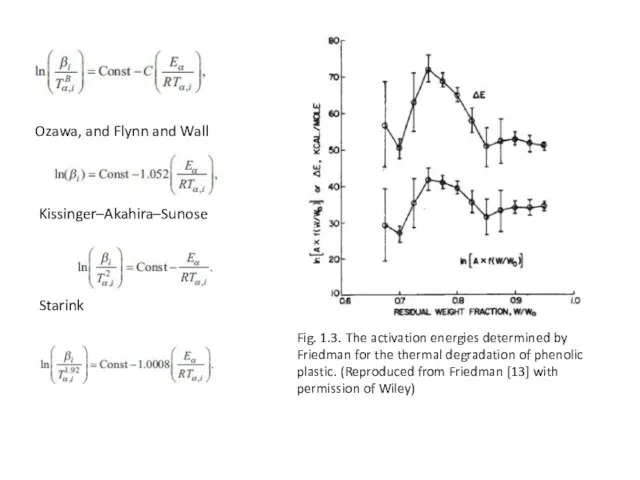
Ozawa, and Flynn and Wall
Fig. 1.3. The activation energies determined by
Ozawa, and Flynn and Wall
Fig. 1.3. The activation energies determined by
Friedman for the thermal degradation of phenolic plastic. (Reproduced from Friedman [13] with permission of Wiley)
Starink
Kissinger–Akahira–Sunose
Слайд 5
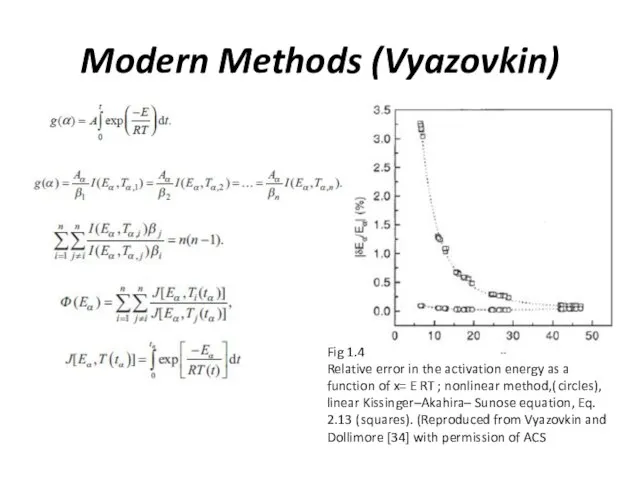
Modern Methods (Vyazovkin)
Fig 1.4
Relative error in the activation energy as a
Modern Methods (Vyazovkin)
Fig 1.4
Relative error in the activation energy as a
function of x= E RT ; nonlinear method,( circles), linear Kissinger–Akahira– Sunose equation, Eq. 2.13 ( squares). (Reproduced from Vyazovkin and Dollimore [34] with permission of ACS
- Предыдущая
Площадь. Равновеликие и равносоставленные фигурыСледующая -
Наука химия. Роль химии в промышленности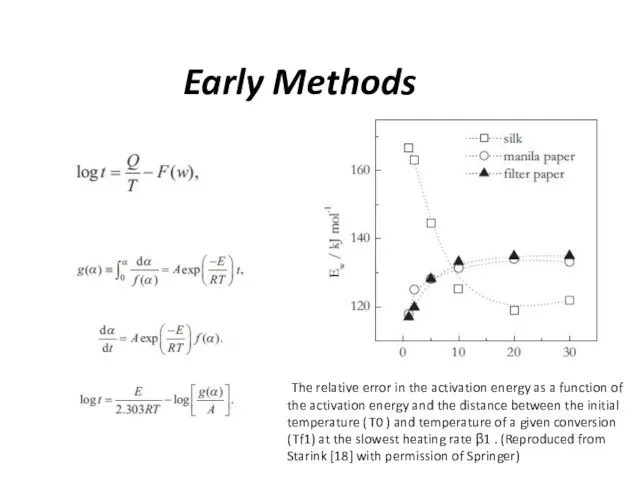
 Метаболизм этанола
Метаболизм этанола Аттестационная работа. Методическая разработка индивидуальной исследовательской работы по химии педагогического колледжа
Аттестационная работа. Методическая разработка индивидуальной исследовательской работы по химии педагогического колледжа Радиоспектроскопические методы исследования, часть 3
Радиоспектроскопические методы исследования, часть 3 Aditivii alimentari: inofensivi sau poluanţi
Aditivii alimentari: inofensivi sau poluanţi Теория химического строения Бутлерова. Второе положение теории Бутлерова. Изомерия. Структурная изомерия
Теория химического строения Бутлерова. Второе положение теории Бутлерова. Изомерия. Структурная изомерия Суды залалсыздандыру әдістері
Суды залалсыздандыру әдістері Презентация по Химии "Бриллианты" - скачать смотреть
Презентация по Химии "Бриллианты" - скачать смотреть  Состав, строение и свойства натурального каучука
Состав, строение и свойства натурального каучука Молочний цукор. Склад і властивості. Роль його в технології молочних продуктів
Молочний цукор. Склад і властивості. Роль його в технології молочних продуктів Теоретическая электрохимия, часть 1
Теоретическая электрохимия, часть 1 Нітрати та нітриди та їх вплив на навколишнє середовище
Нітрати та нітриди та їх вплив на навколишнє середовище Вода Презентацию подготовила ученица 8-б класса Гордеева Юлия
Вода Презентацию подготовила ученица 8-б класса Гордеева Юлия Проектная деятельность школьников в процессе обучения химии
Проектная деятельность школьников в процессе обучения химии Процессы технологии природных энергоносителей и углеродных материалов
Процессы технологии природных энергоносителей и углеродных материалов Основные классы неорганических соединений
Основные классы неорганических соединений Химические элементы и организм человека
Химические элементы и организм человека Электроотрицательность (ЭО). Ковалентная связь
Электроотрицательность (ЭО). Ковалентная связь Алкендер – қаныпаған көмірсутектер. Алынуы, химиялық қасиеттері және қолданылуы
Алкендер – қаныпаған көмірсутектер. Алынуы, химиялық қасиеттері және қолданылуы Презентация по Химии "Производство каучука" - скачать смотреть
Презентация по Химии "Производство каучука" - скачать смотреть  1.При растворении в воде гидроксид-ионы образует вещество, формула которого: 1.При растворении в воде гидроксид-ионы образует ве
1.При растворении в воде гидроксид-ионы образует вещество, формула которого: 1.При растворении в воде гидроксид-ионы образует ве Изомерия
Изомерия Периодическая система элементов Д.И. Менделеева
Периодическая система элементов Д.И. Менделеева Пластмассы в машиностроении
Пластмассы в машиностроении Попутные нефтяные газы
Попутные нефтяные газы Приминение серной кислоты в хозяйстве
Приминение серной кислоты в хозяйстве Презентация по Химии "Презентація з хімії" - скачать смотреть бесплатно
Презентация по Химии "Презентація з хімії" - скачать смотреть бесплатно Фармацевтические суспензии и эмульсии
Фармацевтические суспензии и эмульсии Презентация по Химии "Металлы. Общая характеристика металлов (нахождение в природе и физические свойства)" - скачать смотреть
Презентация по Химии "Металлы. Общая характеристика металлов (нахождение в природе и физические свойства)" - скачать смотреть