Содержание
- 2. INTRODUCTION This Powerpoint show is one of several produced to help students understand selected topics at
- 3. RATES OF REACTION CONTENTS Prior knowledge Collision Theory Methods for increasing rate Surface area Temperature Catalysts
- 4. Before you start it would be helpful to… know how the energy changes during a chemical
- 5. CHEMICAL KINETICS Introduction Chemical kinetics is concerned with the dynamics of chemical reactions such as the
- 6. COLLISION THEORY Collision theory states that... particles must COLLIDE before a reaction can take place not
- 7. COLLISION THEORY Collision theory states that... particles must COLLIDE before a reaction can take place not
- 8. INCREASING THE RATE INCREASE THE SURFACE AREA OF SOLIDS INCREASE TEMPERATURE SHINE LIGHT ADD A CATALYST
- 9. INCREASING SURFACE AREA Increases chances of a collision - more particles are exposed Powdered solids react
- 10. Effect increasing the temperature increases the rate of a reaction particles get more energy so can
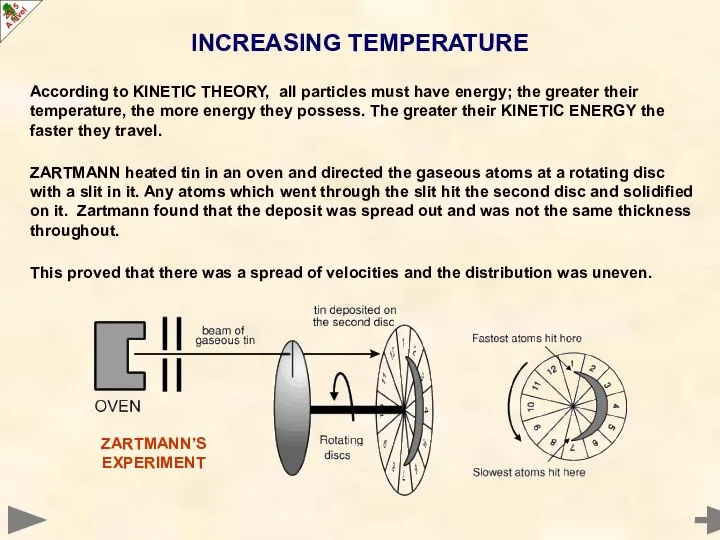
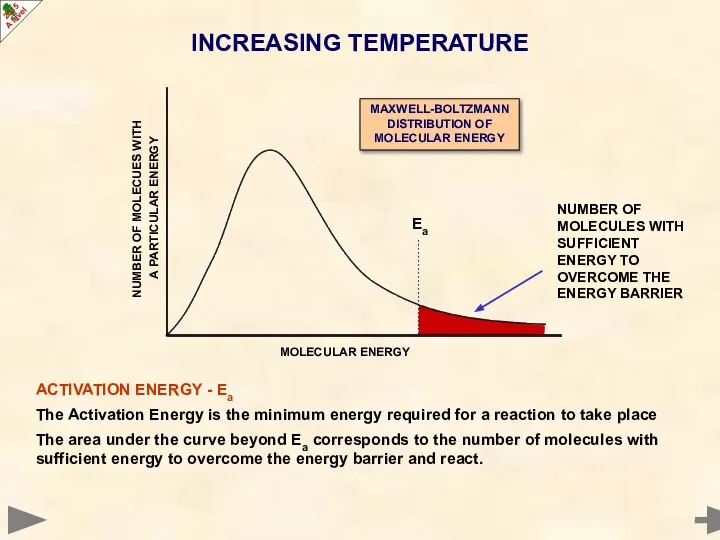
- 11. INCREASING TEMPERATURE According to KINETIC THEORY, all particles must have energy; the greater their temperature, the
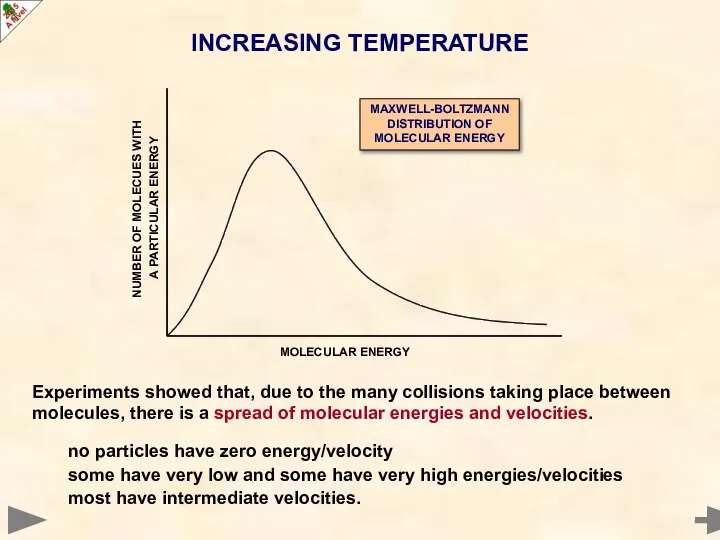
- 12. Experiments showed that, due to the many collisions taking place between molecules, there is a spread
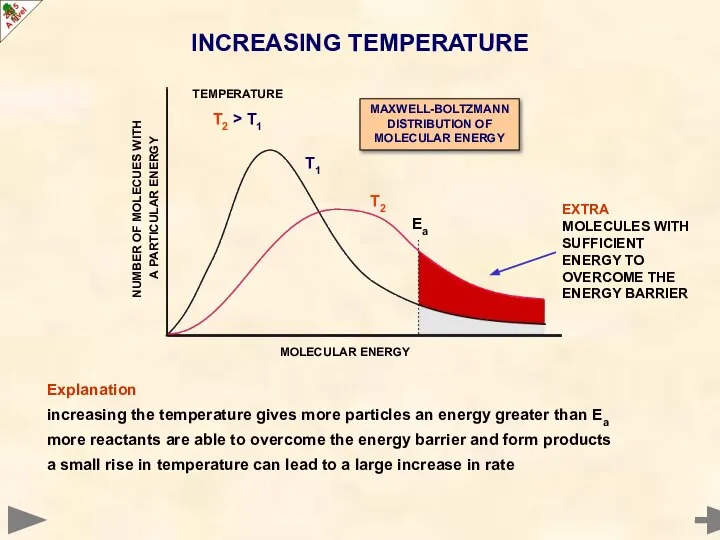
- 13. Increasing the temperature alters the distribution get a shift to higher energies/velocities curve gets broader and
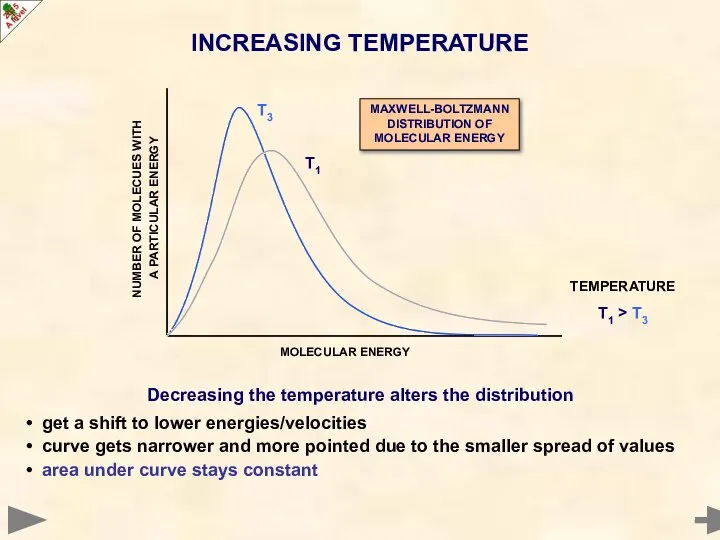
- 14. Decreasing the temperature alters the distribution get a shift to lower energies/velocities curve gets narrower and
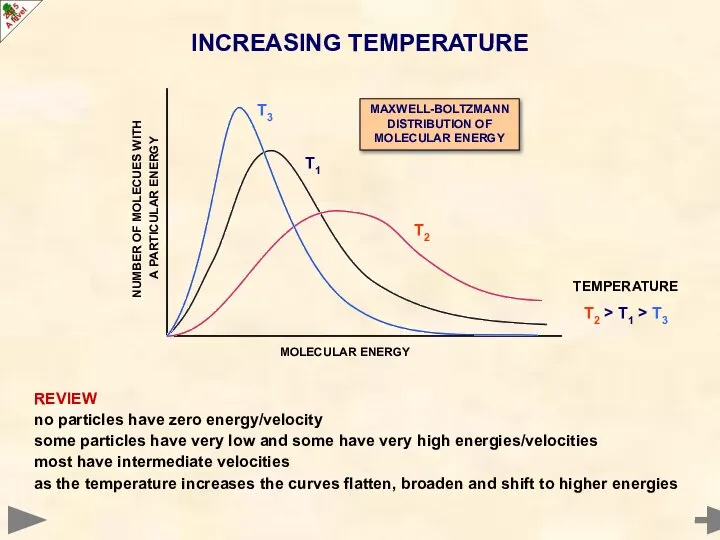
- 15. REVIEW no particles have zero energy/velocity some particles have very low and some have very high
- 16. Ea ACTIVATION ENERGY - Ea The Activation Energy is the minimum energy required for a reaction
- 17. Explanation increasing the temperature gives more particles an energy greater than Ea more reactants are able
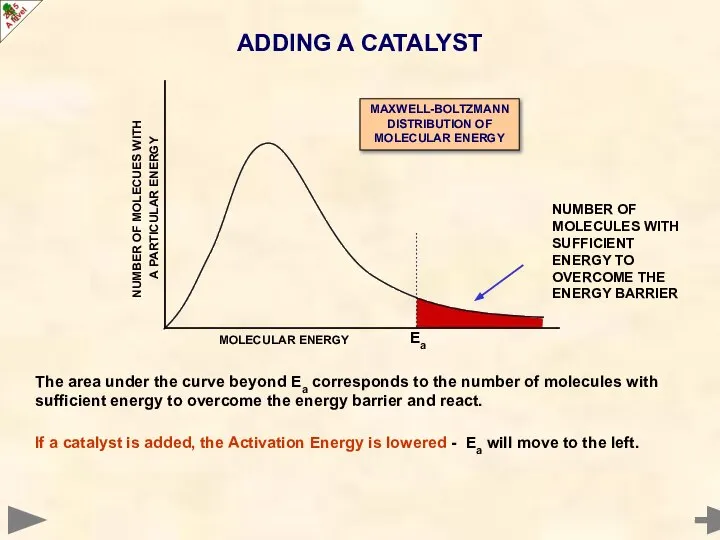
- 18. Catalysts provide an alternative reaction pathway with a lower Activation Energy (Ea) Decreasing the Activation Energy
- 19. The area under the curve beyond Ea corresponds to the number of molecules with sufficient energy
- 20. The area under the curve beyond Ea corresponds to the number of molecules with sufficient energy
- 21. work by providing an alternative reaction pathway with a lower Activation Energy using catalysts avoids the
- 22. work by providing an alternative reaction pathway with a lower Activation Energy using catalysts avoids the
- 23. Catalysts are widely used in industry because they… CATALYSTS - WHY USE THEM?
- 24. Catalysts are widely used in industry because they… allow reactions to take place at lower temperatures
- 25. Catalysts are widely used in industry because they… allow reactions to take place at lower temperatures
- 26. Catalysts are widely used in industry because they… allow reactions to take place at lower temperatures
- 27. Catalysts are widely used in industry because they… allow reactions to take place at lower temperatures
- 28. Catalysts are widely used in industry because they… allow reactions to take place at lower temperatures
- 29. Catalysts are widely used in industry because they… allow reactions to take place at lower temperatures
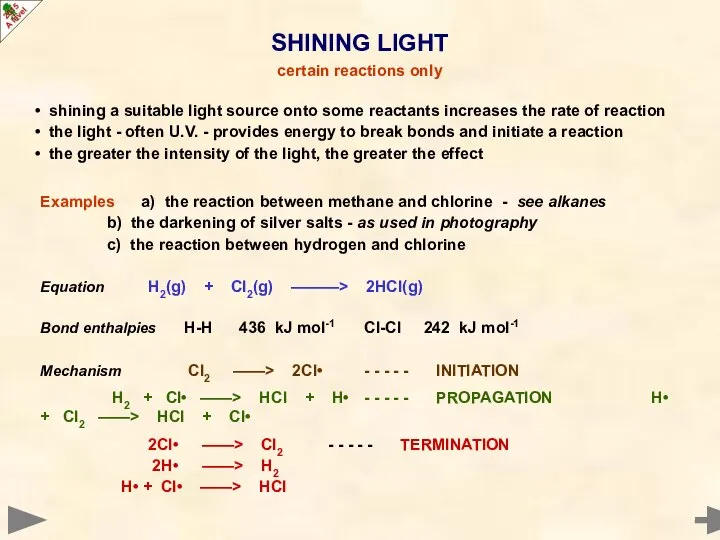
- 30. shining a suitable light source onto some reactants increases the rate of reaction the light -
- 31. increasing the pressure forces gas particles closer together this increases the frequency of collisions so the
- 32. Increasing concentration = more frequent collisions = increased rate of reaction INCREASING CONCENTRATION However, increasing the
- 33. Reactions are fastest at the start and get slower as the reactants concentration drops. In a
- 34. Experimental Investigation the variation in concentration of a reactant or product is followed with time the
- 35. RATE How much concentration changes with time. It is the equivalent of velocity. MEASURING THE RATE
- 36. REVISION CHECK What should you be able to do? Recall and understand the statements in Collision
- 37. You need to go over the relevant topic(s) again Click on the button to return to
- 38. WELL DONE! Try some past paper questions
- 40. Скачать презентацию





























 Химический турнир «Химия –наука чудес и превращений»
Химический турнир «Химия –наука чудес и превращений» Химическая кинетика
Химическая кинетика Полезные геологические химические соединения. Получение меди из малахита
Полезные геологические химические соединения. Получение меди из малахита Теоретические основы органической химии
Теоретические основы органической химии Предмет химии
Предмет химии № 14.
№ 14.  Kristālisku vielu uzbūve
Kristālisku vielu uzbūve Структура и свойства чистого титана
Структура и свойства чистого титана Биохимия печени. (Лекция 19)
Биохимия печени. (Лекция 19) Валентність хімічних елементів
Валентність хімічних елементів Prezentatsia
Prezentatsia ЕГЭ по химии Технология подготовки учащихся
ЕГЭ по химии Технология подготовки учащихся Кристаллы вокруг нас
Кристаллы вокруг нас Предельные углеводороды. Проверочная работа № 4. Вариант 4
Предельные углеводороды. Проверочная работа № 4. Вариант 4 Енергія води та вітру . Як ми її використовуємо в житті.
Енергія води та вітру . Як ми її використовуємо в житті. Chemophobia
Chemophobia Синтез упорядоченных мезопористых материалов (УММ) с использованием матриц из жидких кристаллов. (Лекция 15)
Синтез упорядоченных мезопористых материалов (УММ) с использованием матриц из жидких кристаллов. (Лекция 15) Щелочноземельные металлы Учитель МОУ СОШ № 130 Разнополова О.П.
Щелочноземельные металлы Учитель МОУ СОШ № 130 Разнополова О.П. Классификация химических элементов Й. Я. Берцелиус – выдающийся шведский ученый разделил все элементы на металлы и неметаллы.
Классификация химических элементов Й. Я. Берцелиус – выдающийся шведский ученый разделил все элементы на металлы и неметаллы. Nucleic acids
Nucleic acids Строение атомов. Периодическая система. Лекция 2
Строение атомов. Периодическая система. Лекция 2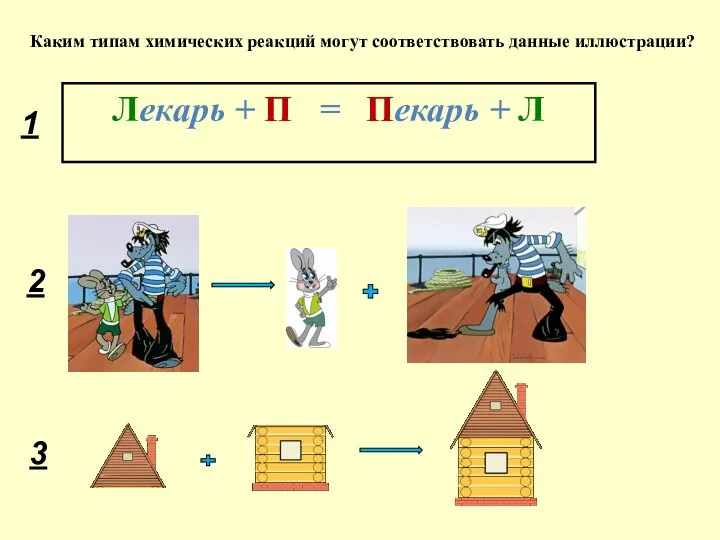 Реакции обмена
Реакции обмена Установка гидрокрекинга в составе завода глубокой переработки нефти ООО «Кинеф»
Установка гидрокрекинга в составе завода глубокой переработки нефти ООО «Кинеф» 04. Наблюдение за горящей свечой. Устройство и работа спиртовки
04. Наблюдение за горящей свечой. Устройство и работа спиртовки Строение и обмен углеводов
Строение и обмен углеводов Зарождение научной химии в России в XVI XVIII веках. История и методология химии. Лекция 9
Зарождение научной химии в России в XVI XVIII веках. История и методология химии. Лекция 9 Элементный, фракционный и химический состав нефти. Классификация нефтей
Элементный, фракционный и химический состав нефти. Классификация нефтей Формирование ответственного отношения школьников к окружающей среде родного края на уроках химии и во внеурочное время.
Формирование ответственного отношения школьников к окружающей среде родного края на уроках химии и во внеурочное время.