Содержание
- 2. The concept of redox reactions Redox reactions - chemical reactions that occur with a change in
- 3. Oxidation - the process of recoil electrons an atom, molecule or ion. Atoms are converted into
- 4. Recovery - the process of accession of electrons an atom, molecule or ion. Atom converted to
- 5. Recovery - atoms, molecules, or ions donate electrons. They are in the process redox reaction oxidized
- 6. Oxidants - atoms, molecules or ions, electrons join. They are in the process of oxidation-reduction reactions
- 7. On the display of the redox properties of the effect of such factors as the stability
- 8. The degree of oxidation of sulfur: -2,0,+4,+6 Н2S-2 - reductant 2Н2S+3O2=2H2O+2SO2 S0,S+4O2 – oxidant and reductant
- 9. Определение степеней окисления атомов химических элементов The oxidation state of atoms of chemical elements in the
- 10. Classification of redox reactions Intermolecular oxidation reactions 2Al0 + 3Cl20 → 2Al+3 Cl3-1 Intramolecular oxidation 2KCl+5O3-2
- 11. The value of redox reactions Redox reactions are very common. They linked the metabolic processes in
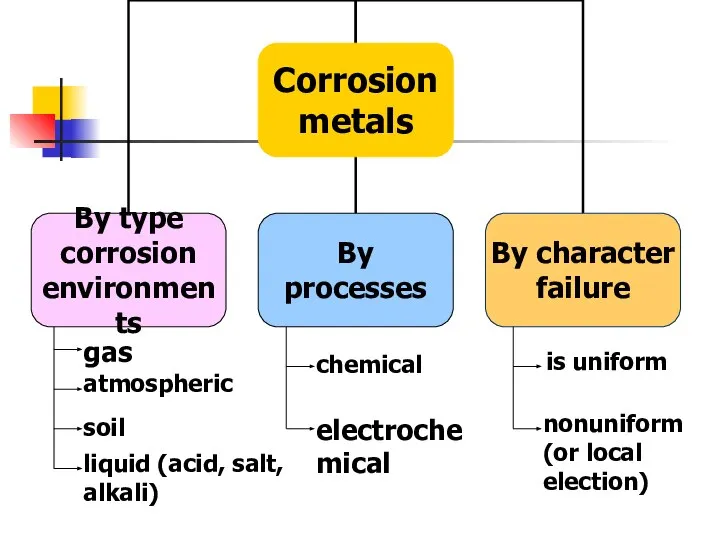
- 12. Corrosion of metals Methods corrosion protection
- 13. CORROSION - spontaneous destruction of metals and alloys as a result of chemical and electrochemical interactions
- 14. Factors that may cause corrosion Oxygen and atmospheric moisture Carbon and sulfur gases contained in the
- 15. gas atmospheric soil liquid (acid, salt, alkali) chemical electrochemical is uniform nonuniform (or local election)
- 16. CHEMICAL - a failure of metals and alloys as a result of their chemical interactions with
- 17. Electrochemical - a failure of metals, which is accompanied by the appearance of an electric current
- 18. CONDITIONS of electrochemical corrosion The position of the metal in a series of activity of metal:
- 19. METHODS corrosion protection The application of protective coatings (paints, varnishes, enamels); Covering other metals (gold-plated, silver,
- 21. Скачать презентацию

















 Относительные молекулярные массы некоторых веществ
Относительные молекулярные массы некоторых веществ Kristālisku vielu uzbūve
Kristālisku vielu uzbūve Растворы. Часть 1
Растворы. Часть 1 Предельные одноатомные спирты
Предельные одноатомные спирты  Структура и функции аминокислот с полярным (заряженным отрицательно) радикалом
Структура и функции аминокислот с полярным (заряженным отрицательно) радикалом Индикаторы
Индикаторы Классификация нефтей и нефтепродуктов
Классификация нефтей и нефтепродуктов Алмаз. Алмаз дегеніміз не
Алмаз. Алмаз дегеніміз не Підготувала Бендюк Вікторія,10-Б
Підготувала Бендюк Вікторія,10-Б  Қышқылдық және негіздік теориясы. Қанның рН анықтау жолдары. Қанның буферлік үйесі
Қышқылдық және негіздік теориясы. Қанның рН анықтау жолдары. Қанның буферлік үйесі Каталитический риформинг
Каталитический риформинг Уран – периодты жүйедегі атомдық номері 92 болатын химиялық элемент
Уран – периодты жүйедегі атомдық номері 92 болатын химиялық элемент Содержание ртути и её соединений в воздухе при использовании осветительных ламп
Содержание ртути и её соединений в воздухе при использовании осветительных ламп Окислительно-восстановительные реакции
Окислительно-восстановительные реакции Химическая посуда и её назначение (3)
Химическая посуда и её назначение (3) Химиядағы «жылдамдық» түсінігі. Химиялық реакция жылдамдығына әсер ететін факторлар
Химиядағы «жылдамдық» түсінігі. Химиялық реакция жылдамдығына әсер ететін факторлар Презентация по Химии "Мыло: польза или вред?" - скачать смотреть
Презентация по Химии "Мыло: польза или вред?" - скачать смотреть  Гидрохимические определения. Методы определения растворенного кислорода в воде
Гидрохимические определения. Методы определения растворенного кислорода в воде Кислоты и соли
Кислоты и соли Муниципальное общеобразовательное учреждение «Итатская СОШ №2 с.Томское» Томский район Как больше и дешевле получать кисл
Муниципальное общеобразовательное учреждение «Итатская СОШ №2 с.Томское» Томский район Как больше и дешевле получать кисл Клонирование генов, создание геномных и кДНК-библиотек, искусственный синтез генов
Клонирование генов, создание геномных и кДНК-библиотек, искусственный синтез генов Аттестационная работа. Программа регионального компонента образовательной области «Химия»
Аттестационная работа. Программа регионального компонента образовательной области «Химия» Исследование фруктовых и овощных соков на наличие в них синтетических красителей и кислотность среды
Исследование фруктовых и овощных соков на наличие в них синтетических красителей и кислотность среды Химический состав водотоков архипилага Новая земля и их воздействие на прибрежную зону
Химический состав водотоков архипилага Новая земля и их воздействие на прибрежную зону Нитраты в овощах и фруктах
Нитраты в овощах и фруктах Презентация по Химии "Правила образования названий разветвленных алканов по международной номенклатуре (ИЮПАК)" - скачать см
Презентация по Химии "Правила образования названий разветвленных алканов по международной номенклатуре (ИЮПАК)" - скачать см Полиэтилен
Полиэтилен Общая характеристика углерода
Общая характеристика углерода