Содержание
- 2. Q1 Select the best choice : Most energy necessary to remove one electron: Cu Cu+ Cu2+
- 3. R1 Select the best choice : Most energy necessary to remove one electron: Cu Cu+ Cu2+
- 4. Q2 Explain why Ag+ is the most common ion for silver. Which is the more likely
- 5. R2 Ag+ is the most common ion for silver because it has [Kr]4d10 . With filled
- 6. R1C The preferred configuration of Mn2+ is [Ar]3d5 The 3d orbital are lower in energy than
- 7. 3.2 Units A) Electromagnetic Radiation 3.2.1: Electromagnetic Radiation 3.2.2: Quantization 3.2.3: The Atomic Spectrum of Hydrogen
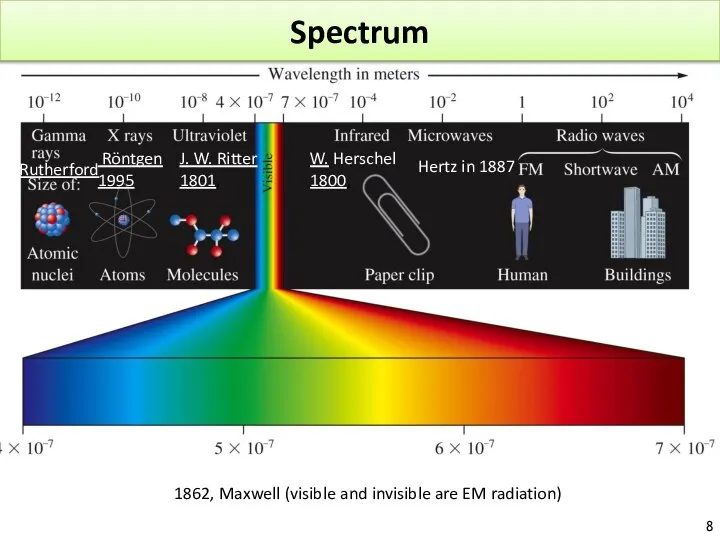
- 8. Spectrum 1862, Maxwell (visible and invisible are EM radiation) J. W. Ritter 1801, W. Herschel 1800,
- 9. ELECTROMAGNETIC RADIATION
- 10. 3.2 EM Radiation A) Electromagnetic Radiation The frequency of radiation used in a typical microwave oven
- 11. 3.2 Units A) Electromagnetic Radiation The frequency of radiation used in a typical microwave oven is
- 12. Solution 1 E = hν, Multiply this value by the Avogadro constant to find the energy
- 13. Solution a. 39.9 J mol–1 b. 3.99 J mol–1 c. 399 J mol–1 d. 0.39 J
- 14. 3.2 Atomic Spectra A) What is the ionization energy (kJ/mol) for an excited state of hydrogen
- 15. 3.2 Response A
- 16. 3.2 Response A
- 17. Exercise A line from the Pfund series has the frequency 8.02 × 1013 Hz. What value
- 18. Useful Table 3.4. The atomic spectrum of hydrogen
- 19. Exercise A line from the Pfund series has the frequency 8.02 × 1013 Hz. What value
- 20. Exercise A line from the Pfund series has the frequency 8.02 × 1013 Hz. What value
- 21. Exercise Rearranging, by taking 0.04 from each side, gives Dividing both sides by –0.01563 and multiplying
- 22. 3.2 Light Interference In Thomas Young’s experiment when he passed light through two closely placed slits,
- 23. 1.2 Light Interference A set of maxima and minima in an interference pattern suggests a totally
- 24. Q4 The wave function of an electron is related to the probability for finding a particle
- 25. Radial Probability Distribution https://www.youtube.com/watch?v=Prf_jzbD_bM Ψ(r, θ, φ)=R(r) Y(θ, φ)
- 26. Radial Distance
- 27. Exercise Sketch radial wavefunctions, radial distribution functions and boundary diagrams for 6s and 5p electrons
- 28. Radial nodes for S = n-1 Radial nodes for p = n-2
- 29. Particle in a Box
- 33. Show that if Ψ = Asinrx, the boundary conditions require that (Ψ = 0 when x
- 35. Show that if , the energy levels of the particle are given by
- 37. Show that substituting the value of r given in question C into ψ=Asinrx and applying the
- 39. Integration
- 40. Schrodinger Equation What is the normalization constant for the wave function exp(-ax) over the range from
- 41. What is the normalization constant for the wave function exp(-ax) over the range from 0 to
- 42. Match the type of orbital defined by the quantum numbers given in questions (a) to (d)
- 43. Use Slater’s rules to determine the relative sizes of N, O and F atoms. Slater Rules
- 44. Step-1 Slater Rules N (Z = 7) is 1s22s22p3 O (Z = 8) is 1s22s22p4 F
- 45. Use Slater’s rules to determine the relative sizes of N, O and F atoms. Slater Rules
- 46. Electronic Configuration
- 47. Q3 What are the values and quantum numbers l and n of a 5d electron. n
- 48. Answer 2 for any d orbitals the l = 2, n = 5
- 49. R1c Explain factors that cause lanthanide contraction. (0.25pts) Explain why Ag+ is the most common ion
- 50. ρsinφ Δρ Angle = Arc Length radius of the circle Arc Length × Arc Length ×
- 52. Скачать презентацию




![R1C The preferred configuration of Mn2+ is [Ar]3d5 The 3d orbital](/_ipx/f_webp&q_80&fit_contain&s_1440x1080/imagesDir/jpg/1407923/slide-5.jpg)











































 Смазывающие вещества
Смазывающие вещества Физические и химические явления
Физические и химические явления Кислоты и соли
Кислоты и соли Виртуозы химического эксперимента
Виртуозы химического эксперимента Расчёты по уравнению реакции
Расчёты по уравнению реакции Типы кристаллических структур минералов. Формулы. Классификация
Типы кристаллических структур минералов. Формулы. Классификация Классификация химических реакций
Классификация химических реакций Идеал газ. Молекулалы-кинетикалық теорияның негізгі теңдеуі. Молекулалық орташа квадраттық жылдамдығына есептер шығару
Идеал газ. Молекулалы-кинетикалық теорияның негізгі теңдеуі. Молекулалық орташа квадраттық жылдамдығына есептер шығару Периодический закон и периодическая система элементов Д.И. Менделеева
Периодический закон и периодическая система элементов Д.И. Менделеева Кислородосодержащие органические соединения
Кислородосодержащие органические соединения История развития химии
История развития химии Застосування амоніаку і солей амонію
Застосування амоніаку і солей амонію  Благородные металлы
Благородные металлы Карбонильные соединения
Карбонильные соединения Электрический ток в растворах и расплавах электролитов. Электролиз
Электрический ток в растворах и расплавах электролитов. Электролиз Спирты, фенолы, тиолы
Спирты, фенолы, тиолы Чистые вещества и смеси. Способы разделения смесей
Чистые вещества и смеси. Способы разделения смесей Химическая связь. Типы кристаллических решеток. Урок №6,7
Химическая связь. Типы кристаллических решеток. Урок №6,7 Молярный объем Задачи урока: 1. Выяснить, что такое молярный объем. 2. Научиться решать задачи с использованием понятия «молярный
Молярный объем Задачи урока: 1. Выяснить, что такое молярный объем. 2. Научиться решать задачи с использованием понятия «молярный Қышқылдық және негіздік теориясы. Қанның рН анықтау жолдары. Қанның буферлік үйесі
Қышқылдық және негіздік теориясы. Қанның рН анықтау жолдары. Қанның буферлік үйесі Основные классы неорганических соединений
Основные классы неорганических соединений Презентация по Химии "Экологические проблемы химии гидросферы" - скачать смотреть
Презентация по Химии "Экологические проблемы химии гидросферы" - скачать смотреть  Fuziunea nucleara
Fuziunea nucleara Взаимное влияние атомов в молекулах
Взаимное влияние атомов в молекулах Составление химических формул по валентности
Составление химических формул по валентности 1oe_zanyatie (1)
1oe_zanyatie (1) Химическая термодинамика
Химическая термодинамика Инновационный проект производства фуллеренов
Инновационный проект производства фуллеренов