Содержание
- 2. CH4 + Cl2 CH3Cl + HCl Overall reaction equation Conditions ultra violet light excess methane i.e.
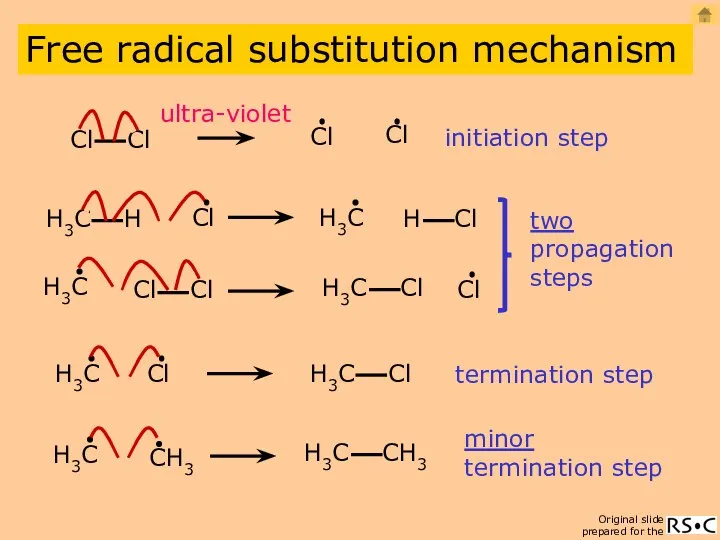
- 3. initiation step two propagation steps termination step ultra-violet minor termination step Free radical substitution mechanism
- 4. CH3Cl + Cl2 CH2Cl2 + HCl Overall reaction equations Conditions ultra-violet light CH2Cl2 + Cl2 CHCl3
- 5. n H2C=CH2 Overall reaction equation polyethene free radical source i.e. homolytic breaking of covalent bonds (a
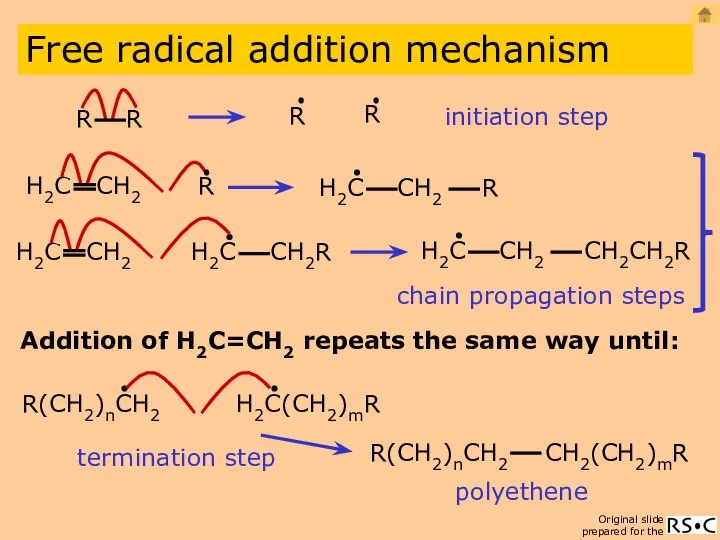
- 6. initiation step Addition of H2C=CH2 repeats the same way until: polyethene Free radical addition mechanism chain
- 7. Electrophilic addition CH3CH=CH2 + Br2 CH3CHBrCH2Br bromine with propene hydrogen bromide with but-2-ene CH3CH=CHCH3 + HBr
- 8. Electrophilic addition mechanism carbocation 1,2-dibromopropane bromine with propene reaction equation
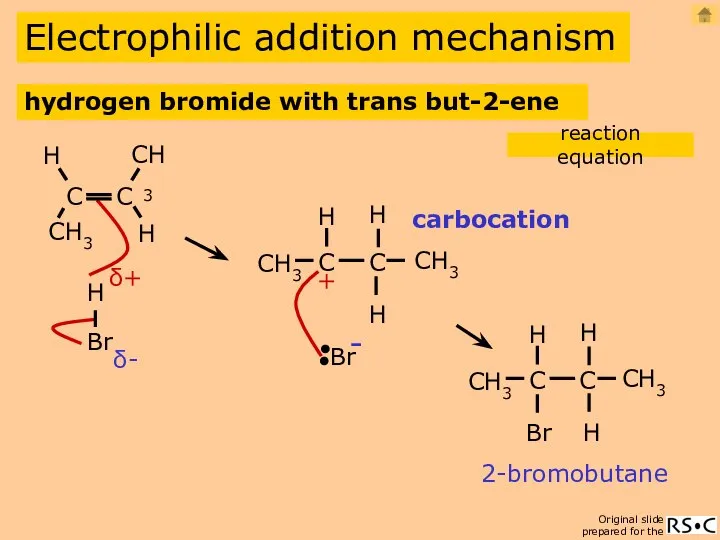
- 9. Electrophilic addition mechanism carbocation 2-bromobutane hydrogen bromide with trans but-2-ene reaction equation
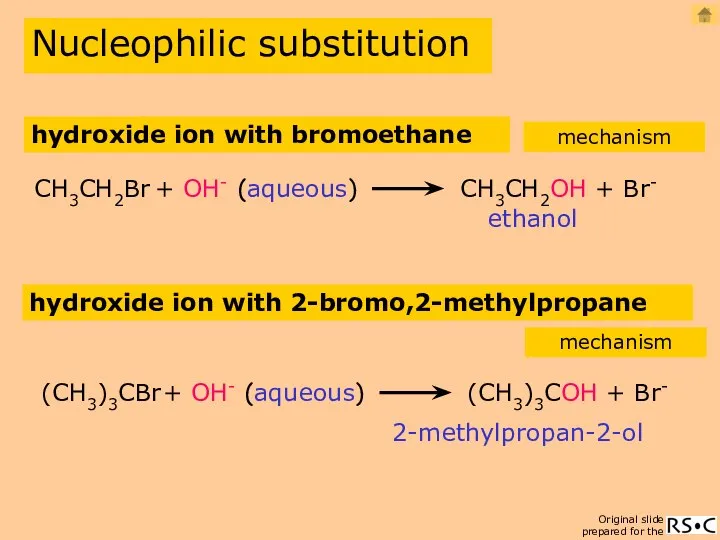
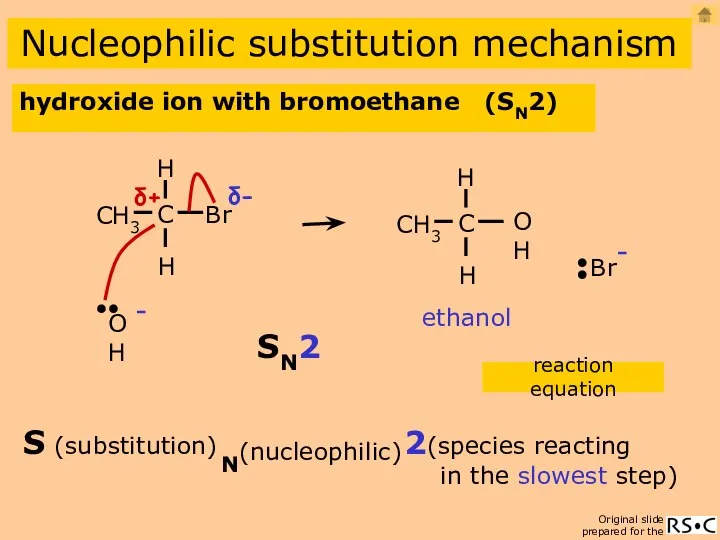
- 10. hydroxide ion with bromoethane ethanol CH3CH2Br + OH- CH3CH2OH + Br- (aqueous) Nucleophilic substitution mechanism hydroxide
- 11. hydroxide ion with bromoethane (SN2) Nucleophilic substitution mechanism ethanol reaction equation 2(species reacting in the slowest
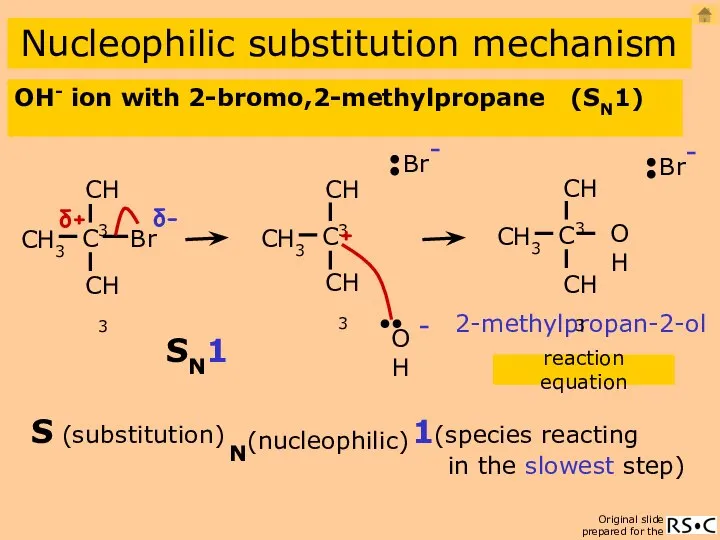
- 12. OH- ion with 2-bromo,2-methylpropane (SN1) Nucleophilic substitution mechanism 2-methylpropan-2-ol reaction equation 1(species reacting in the slowest
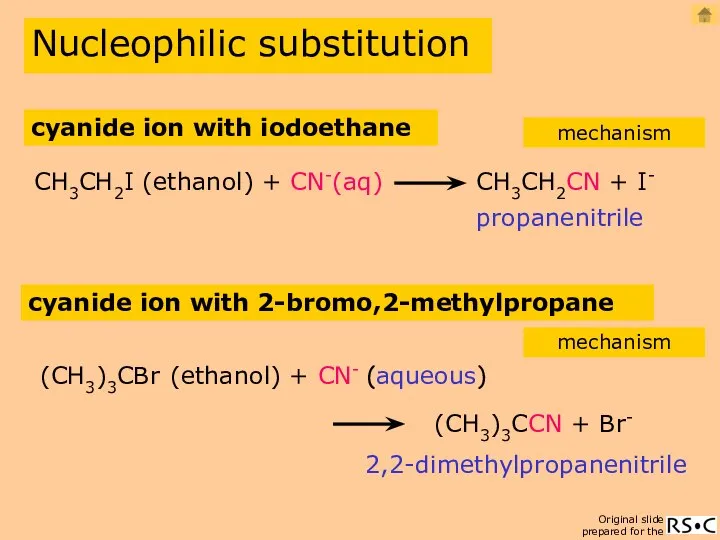
- 13. Nucleophilic substitution propanenitrile CH3CH2I (ethanol) + CN-(aq) CH3CH2CN + I- cyanide ion with iodoethane mechanism cyanide
- 14. cyanide ion with iodoethane (SN2) Nucleophilic substitution mechanism propanenitrile reaction equation 2(species reacting in the slowest
- 15. CN- ion with 2-bromo,2-methylpropane (SN1) Nucleophilic substitution mechanism 2,2-dimethyl propanenitrile 1(species reacting in the slowest step)
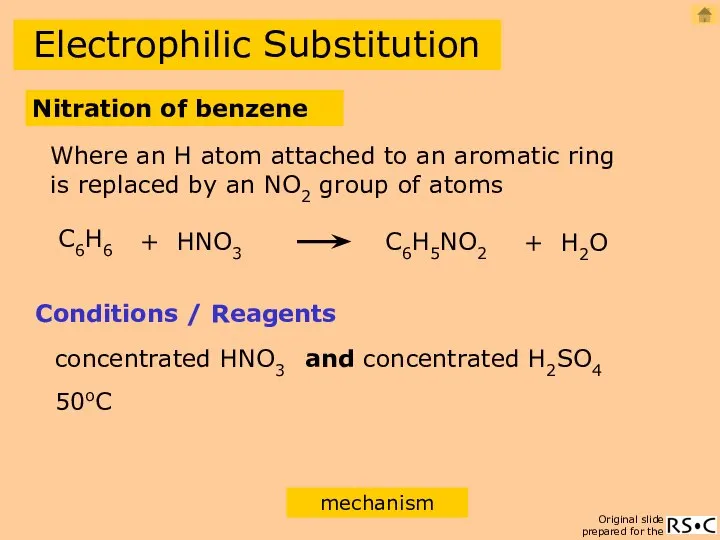
- 16. Electrophilic Substitution Nitration of benzene C6H6 + HNO3 C6H5NO2 + H2O Conditions / Reagents concentrated HNO3
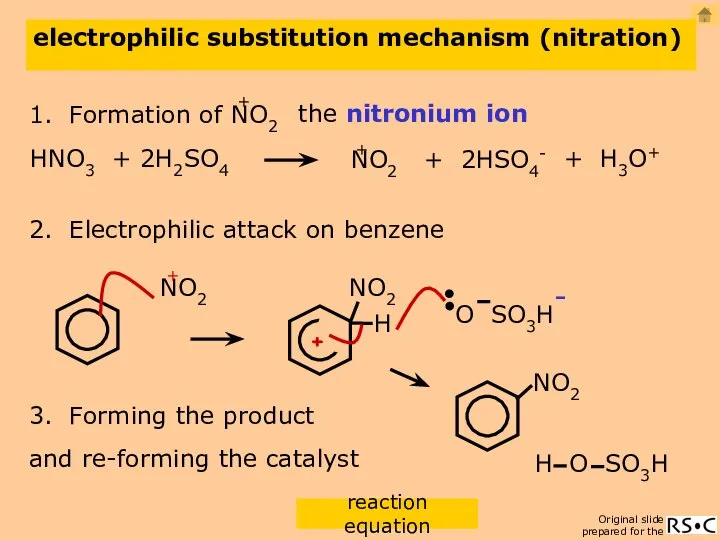
- 17. electrophilic substitution mechanism (nitration) 2. Electrophilic attack on benzene 3. Forming the product the nitronium ion
- 18. Bromination of benzene C6H6 + Br2 C6H5Br + HBr Conditions / Reagents Br2 and anhydrous AlBr3
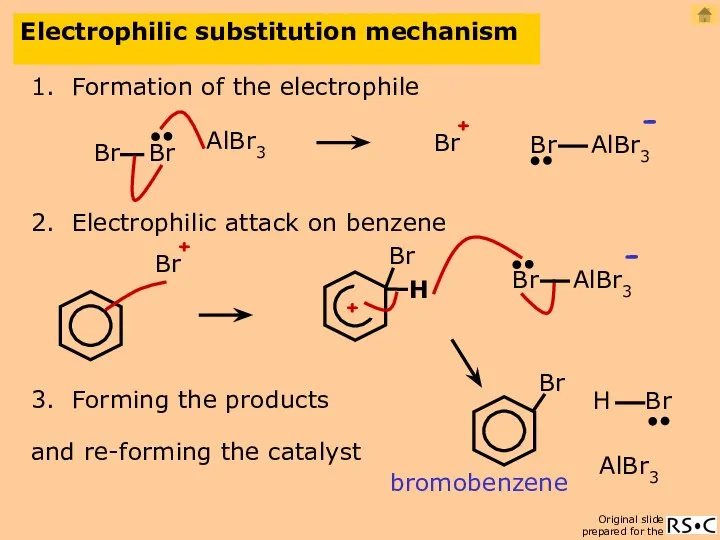
- 19. Electrophilic substitution mechanism 1. Formation of the electrophile AlBr3 2. Electrophilic attack on benzene 3. Forming
- 20. Alkylation of benzene C6H6 + RCl C6H5R + HCl Conditions / Reagents RCl (haloakane) and anhydrous
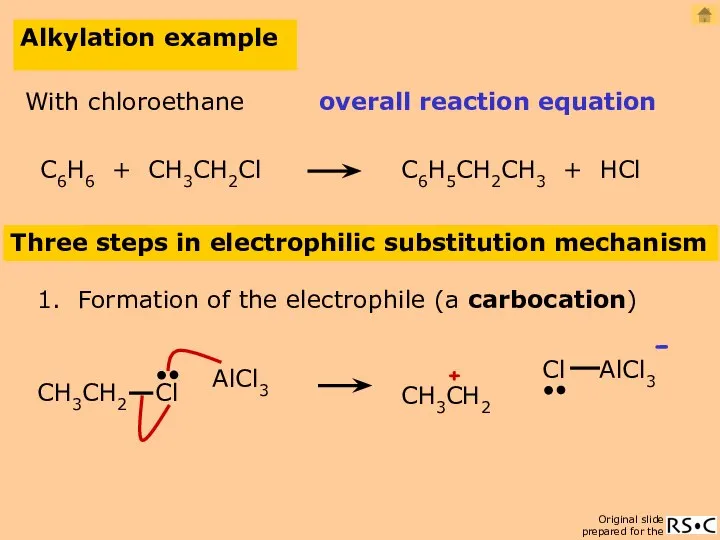
- 21. Alkylation example Three steps in electrophilic substitution mechanism 1. Formation of the electrophile (a carbocation) AlCl3
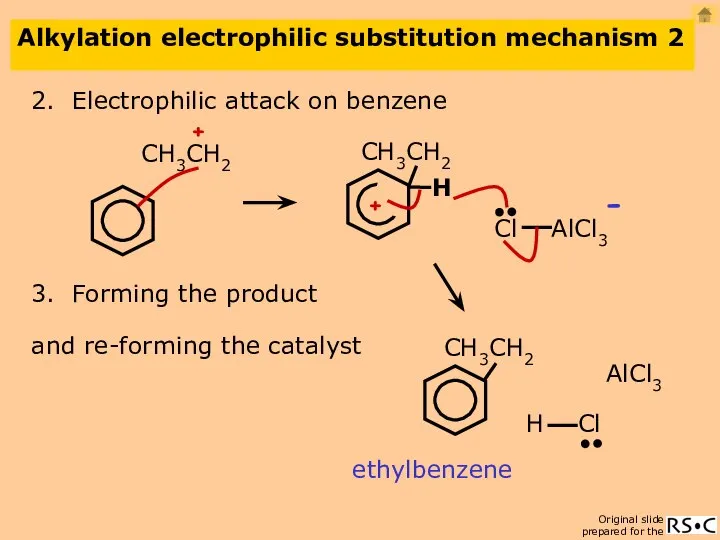
- 22. Alkylation electrophilic substitution mechanism 2 2. Electrophilic attack on benzene ethylbenzene 3. Forming the product AlCl3
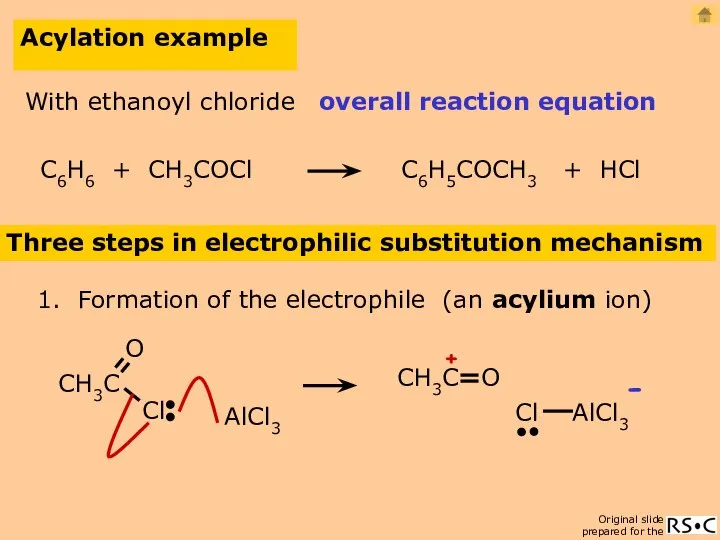
- 23. Acylation of benzene C6H6 + RCOCl C6H5COR + HCl Conditions / Reagents RCOCl (acyl chloride) and
- 24. Acylation example Three steps in electrophilic substitution mechanism 1. Formation of the electrophile (an acylium ion)
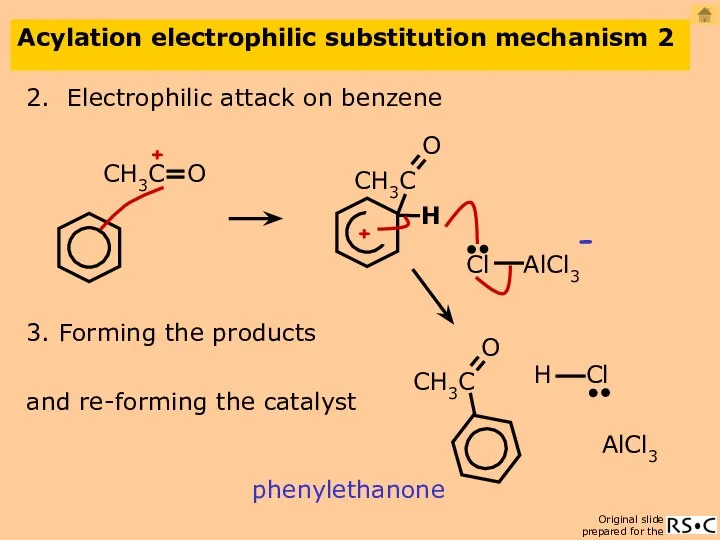
- 25. Acylation electrophilic substitution mechanism 2 2. Electrophilic attack on benzene phenylethanone AlCl3 3. Forming the products
- 26. Nucleophilic Addition RCHO + HCN RCH(OH)CN Conditions / Reagents NaCN (aq) and H2SO4(aq) Room temperature and
- 27. Nucleophilic Addition Mechanism hydrogen cyanide with propanone H+ NaCN (aq) is a source of cyanide ions
- 28. Advice
- 30. Скачать презентацию













 Выращивание кристаллов
Выращивание кристаллов Кристаллы необычной красоты
Кристаллы необычной красоты Интернет-семинар (вебинар). Методика подготовки к олимпиадам и интеллектуальным турнирам по химии. Часть 1
Интернет-семинар (вебинар). Методика подготовки к олимпиадам и интеллектуальным турнирам по химии. Часть 1 Предалхимический период
Предалхимический период Сверхразветвленные полимеры: типы, получение, применение
Сверхразветвленные полимеры: типы, получение, применение Уравнения химических реакций
Уравнения химических реакций Polivinilpirrolidon-limon turşusunun Na duzu-su ikifazalı sisteminin fizikikimyəvi xassələri
Polivinilpirrolidon-limon turşusunun Na duzu-su ikifazalı sisteminin fizikikimyəvi xassələri в організмі
в організмі  Симметрия в химии
Симметрия в химии Алюминий және оның қосылыстары
Алюминий және оның қосылыстары Липиды омыляемые и неомыляемые
Липиды омыляемые и неомыляемые Молекулярно-кінетичні явища в дисперсних системах
Молекулярно-кінетичні явища в дисперсних системах Презентация по химии Нуклеофильное замещение карбоновых кислот
Презентация по химии Нуклеофильное замещение карбоновых кислот Аминокислоты. Определение молекулярной формулы органического вещества
Аминокислоты. Определение молекулярной формулы органического вещества Липиды омыляемые и неомыляемые
Липиды омыляемые и неомыляемые Водород.Способы получения. Физические свойства
Водород.Способы получения. Физические свойства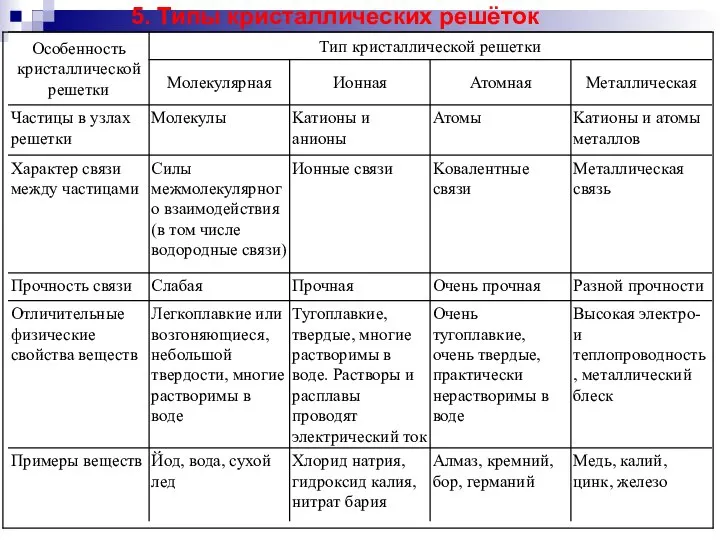 Типы кристаллических решёток. (Лекция 3)
Типы кристаллических решёток. (Лекция 3) Витами́н A
Витами́н A Фосфорное сырье. Применение и обогащение фосфорного сырья
Фосфорное сырье. Применение и обогащение фосфорного сырья Современная химия Химия- это наука, изучающая свойства и превращения веществ, сопровождающееся изменением их состава и строе
Современная химия Химия- это наука, изучающая свойства и превращения веществ, сопровождающееся изменением их состава и строе Обратимость химических реакций. Химическое равновесие
Обратимость химических реакций. Химическое равновесие Общая характеристика металлов
Общая характеристика металлов Каталіз: каталітичне гідрування
Каталіз: каталітичне гідрування Нефть. Чёрное золото
Нефть. Чёрное золото Тепловой эффект химических реакций
Тепловой эффект химических реакций Угольная кислота H2CO3
Угольная кислота H2CO3 Минералогия. Силикаты
Минералогия. Силикаты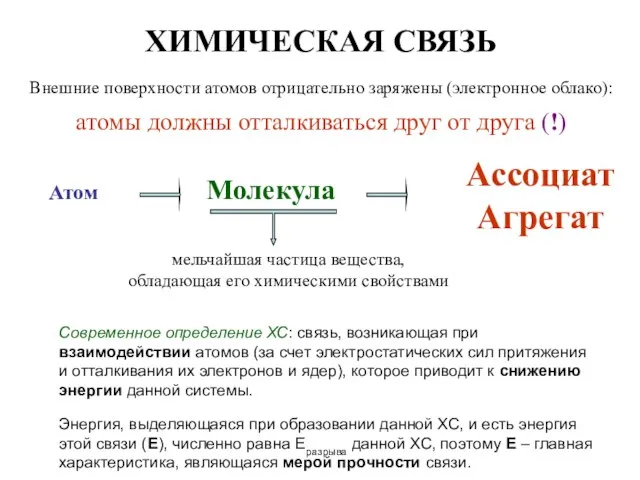 Химическая связь
Химическая связь