Содержание
- 2. Crystalline solids have a very regular atomic structure: that is, the local positions of atoms with
- 3. Point Defects Point defects in ionic solids I Frenkel defect: anion vacancy-interstitial cation pair. Schottky defect:
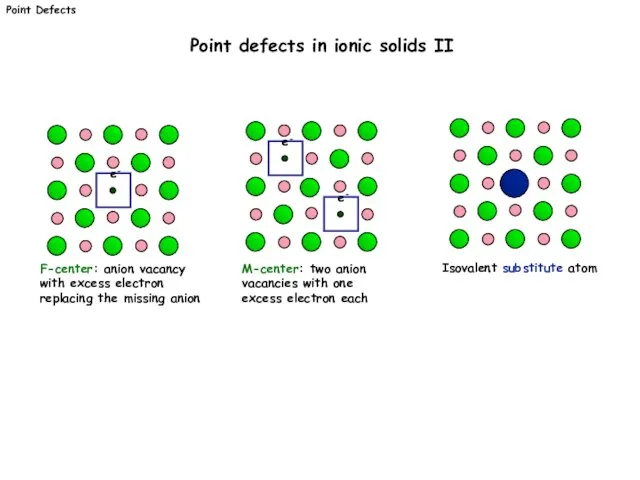
- 4. F-center: anion vacancy with excess electron replacing the missing anion e- M-center: two anion vacancies with
- 5. M corresonds to the species. These include: atoms - e.g. Si, Ni, O, Cl, vacancies -
- 6. Point Defects Kröger-Vink notation II = an aluminium ion sitting on an aluminium lattice site, with
- 7. Reaction involving defects must be: Example: Point Defects Defect chemical reaction Formation of a Schottky defect
- 8. Gperf: free energy of the perfect crystal hdef: enthalpy of formation of one defect sdvib: vibrational
- 9. Yanagida et al.: p. 60-61 Number of ways to arrange nv vacancies within a crystal with
- 10. Entropy Configurational Entropy Entropy originating from the many possibilities of arranging many vacancies Formation ("vibrational") Entropy
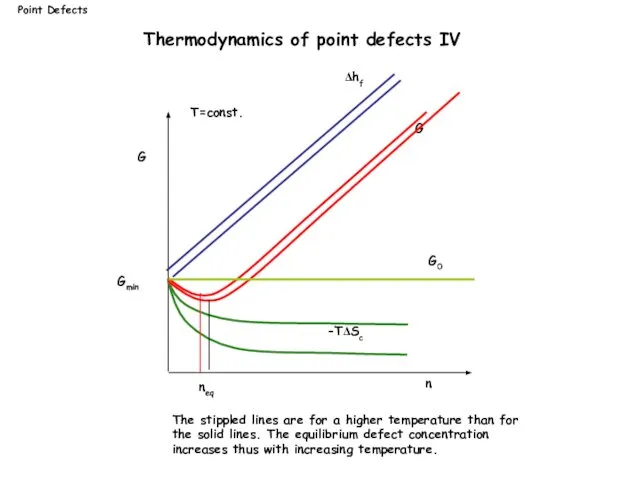
- 11. G n G0 Δhf G neq -TΔSc Gmin T=const. The stippled lines are for a higher
- 12. Point Defects Equilibrium Schottky defect concentration - Number of Schottky pairs: - Formation of a Schottky
- 13. Extrinsic defect concentration I - Total number of cation vacancies: - Substitution of a divalent cation
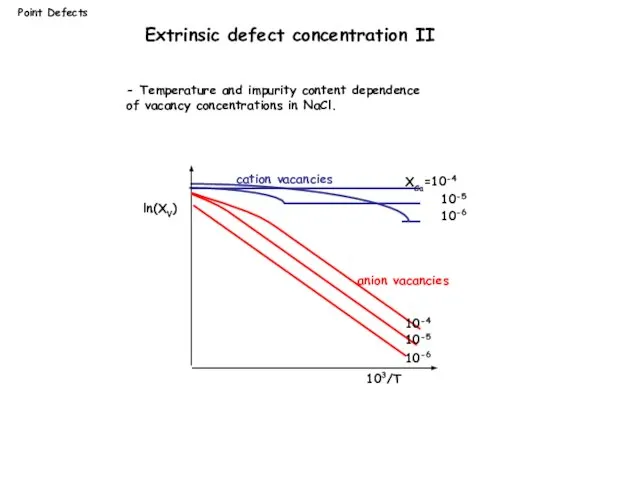
- 14. Point Defects ln(XV) 103/T XCa=10-4 10-5 10-6 10-4 10-5 10-6 cation vacancies anion vacancies - Temperature
- 15. Point Defects Nonstoichiometric defects In nonstoichiometric defect reactions the composition of the cystal changes as a
- 16. Atomic diffusion is a process whereby the random thermally-activated hopping of atoms in a solid results
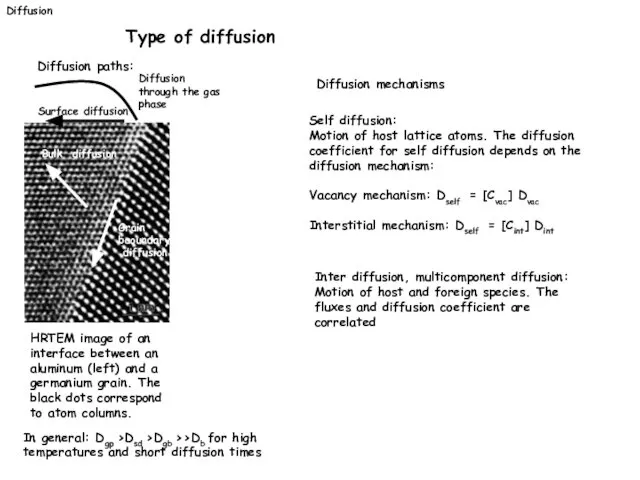
- 17. Diffusion Type of diffusion Diffusion paths: HRTEM image of an interface between an aluminum (left) and
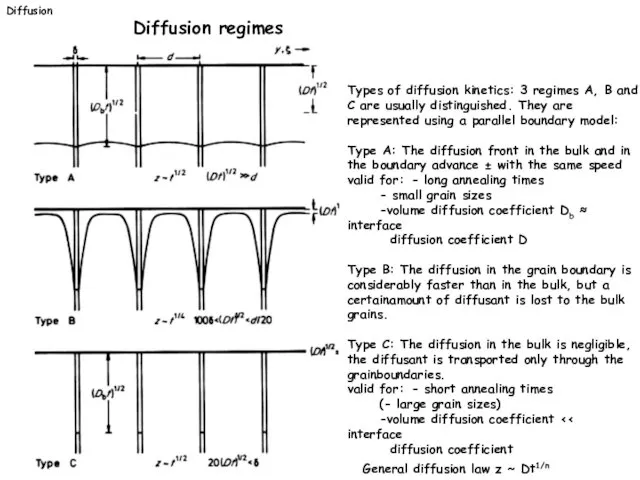
- 18. Types of diffusion kinetics: 3 regimes A, B and C are usually distinguished. They are represented
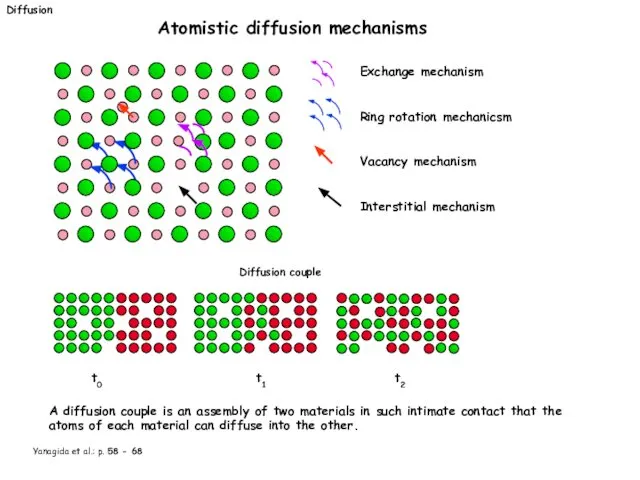
- 19. Diffusion Atomistic diffusion mechanisms Exchange mechanism Ring rotation mechanicsm Vacancy mechanism Interstitial mechanism Diffusion couple t0
- 20. Diffusion Fick’s 1. law dC dx C x The flux J in direction x of the
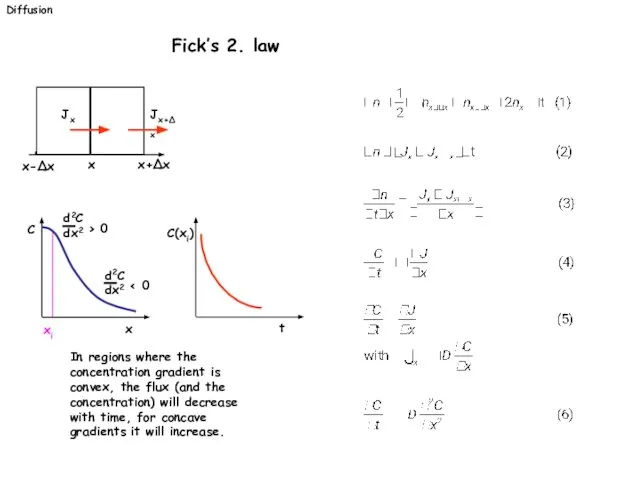
- 21. Diffusion Fick’s 2. law In regions where the concentration gradient is convex, the flux (and the
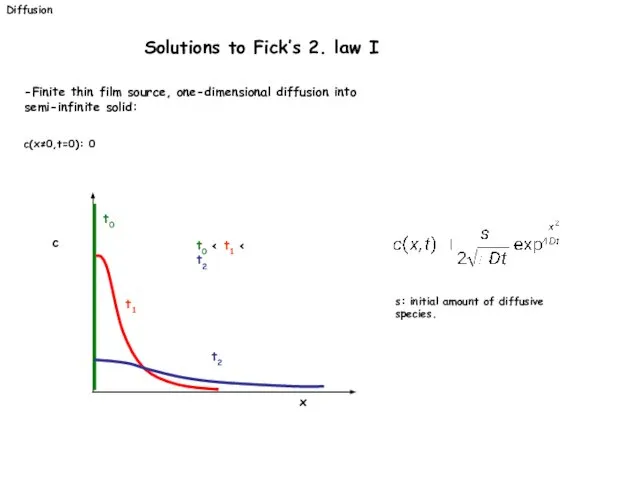
- 22. Diffusion Solutions to Fick’s 2. law I -Finite thin film source, one-dimensional diffusion into semi-infinite solid:
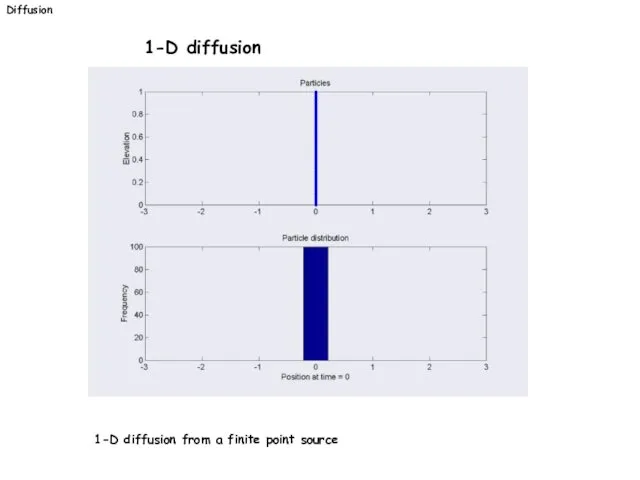
- 23. Diffusion 1-D diffusion 1-D diffusion from a finite point source
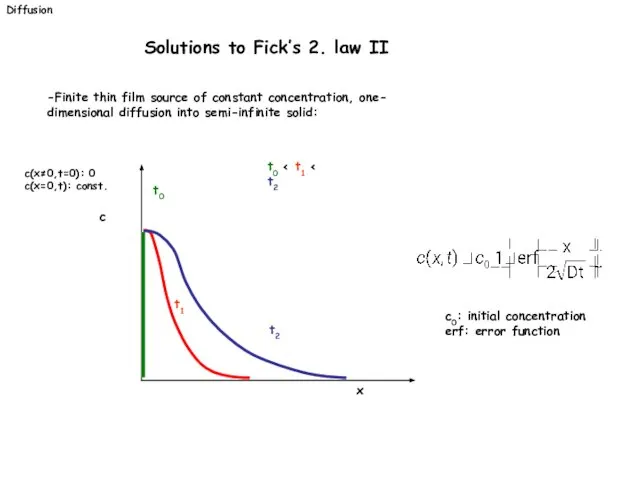
- 24. -Finite thin film source of constant concentration, one- dimensional diffusion into semi-infinite solid: c(x≠0,t=0): 0 c(x=0,t):
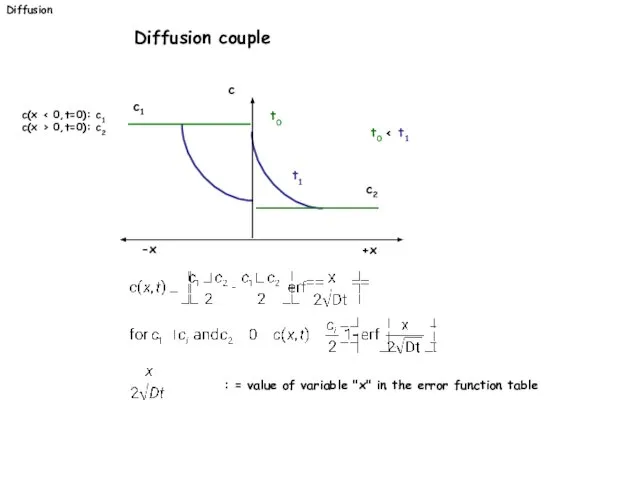
- 25. Diffusion Diffusion couple c(x c(x > 0,t=0): c2 +x -x c t1 t0 t0 c1 c2
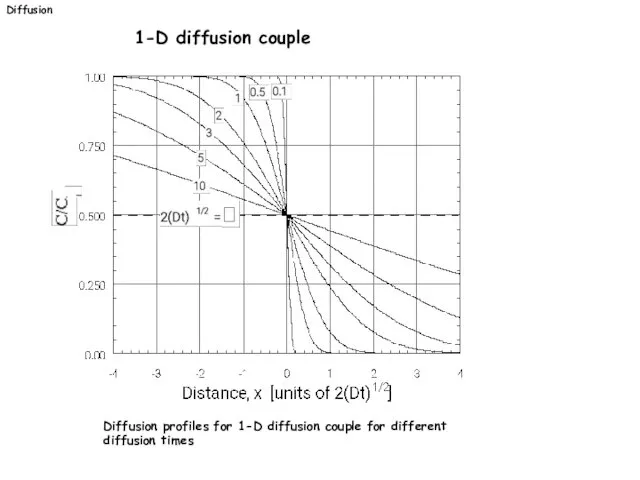
- 26. Diffusion 1-D diffusion couple Diffusion profiles for 1-D diffusion couple for different diffusion times
- 27. Diffusion
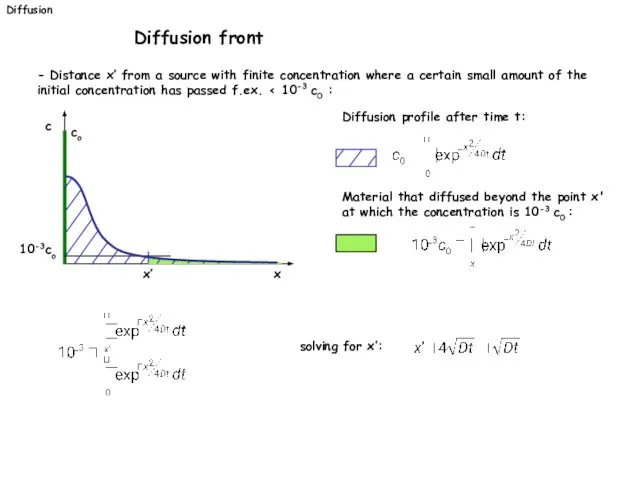
- 28. - Distance x’ from a source with finite concentration where a certain small amount of the
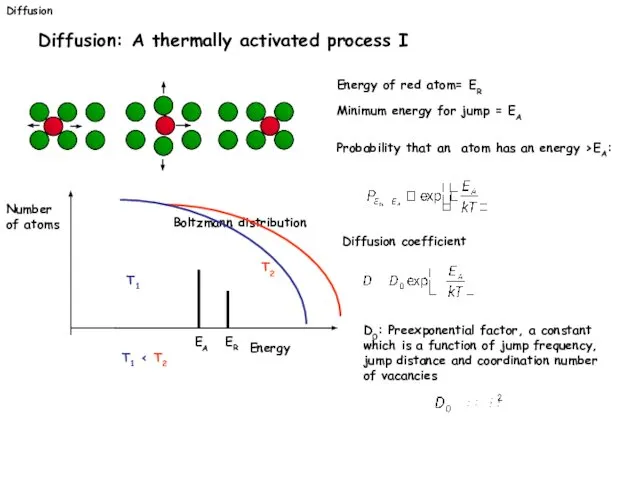
- 29. Diffusion Diffusion: A thermally activated process I Energy of red atom= ER Minimum energy for jump
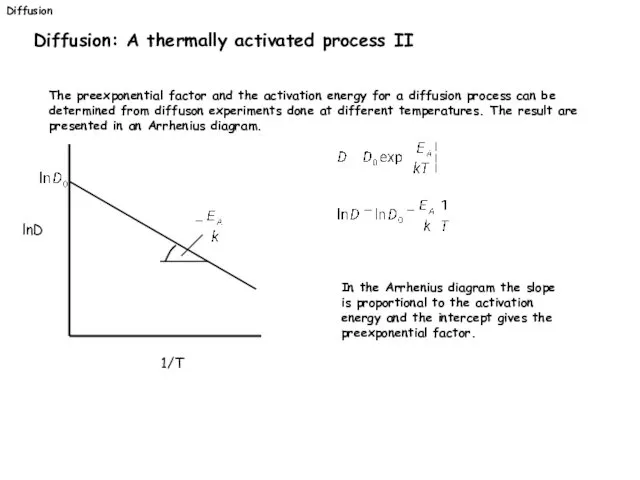
- 30. Diffusion Diffusion: A thermally activated process II The preexponential factor and the activation energy for a
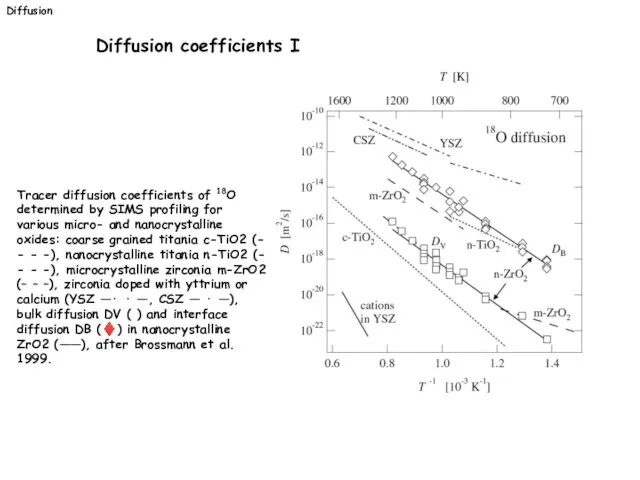
- 31. Tracer diffusion coefficients of 18O determined by SIMS profiling for various micro- and nanocrystalline oxides: coarse
- 33. Скачать презентацию













 Выращивание кристаллов Выполнил: ученик 8 класса А Алексеев Ден
Выращивание кристаллов Выполнил: ученик 8 класса А Алексеев Ден Хром және оның қосылыстары
Хром және оның қосылыстары Способы выращивания кристаллов (лекция 3)
Способы выращивания кристаллов (лекция 3) Морфология тел полезных ископаемых
Морфология тел полезных ископаемых Аминокислоты. АТФ
Аминокислоты. АТФ Защита от коррозии изменением состава агрессивной среды и введением ингибиторов
Защита от коррозии изменением состава агрессивной среды и введением ингибиторов Макроэлементы и микроэлементы
Макроэлементы и микроэлементы Химия элементов IVA группы
Химия элементов IVA группы Качественные реакции
Качественные реакции Хром. Соединения хрома
Хром. Соединения хрома Коррозия
Коррозия Строение электронных оболочек. Объяснение периодической системы элементов Д.И.Менделеева
Строение электронных оболочек. Объяснение периодической системы элементов Д.И.Менделеева Презентация по Химии "Неметаллы" - скачать смотреть бесплатно
Презентация по Химии "Неметаллы" - скачать смотреть бесплатно Презентация по Химии "Применение электролиза" - скачать смотреть
Презентация по Химии "Применение электролиза" - скачать смотреть  G12 - класс антифризов
G12 - класс антифризов Atomic number, Mass number and Isotopes
Atomic number, Mass number and Isotopes Электрохимия. Электродные потенциалы
Электрохимия. Электродные потенциалы Обмен сложных липидов
Обмен сложных липидов Шестичленные гетероциклические соединения с одним и двумя гетероатомами. Конденсированные системы гетероциклов
Шестичленные гетероциклические соединения с одним и двумя гетероатомами. Конденсированные системы гетероциклов Ֆլավանոիդներ պարունակող դեղաբույսեր եվ հումք
Ֆլավանոիդներ պարունակող դեղաբույսեր եվ հումք Презентація учня 11-Б класу гімназії №117 ім. Лесі Українки Мусієнка Андрія
Презентація учня 11-Б класу гімназії №117 ім. Лесі Українки Мусієнка Андрія  Клітинна біохімія. Метаболізм вуглеводів
Клітинна біохімія. Метаболізм вуглеводів Техника безопасности на уроках химии
Техника безопасности на уроках химии Окислительно-восстановительное титрование. Индикаторы
Окислительно-восстановительное титрование. Индикаторы Шкідливий вплив тютюнопаління
Шкідливий вплив тютюнопаління Оксид кремния - это.......
Оксид кремния - это....... Свойства белков как биологических полимеров. 10 класс
Свойства белков как биологических полимеров. 10 класс Углеводы. Глюкоза-представитель моносахаридов
Углеводы. Глюкоза-представитель моносахаридов