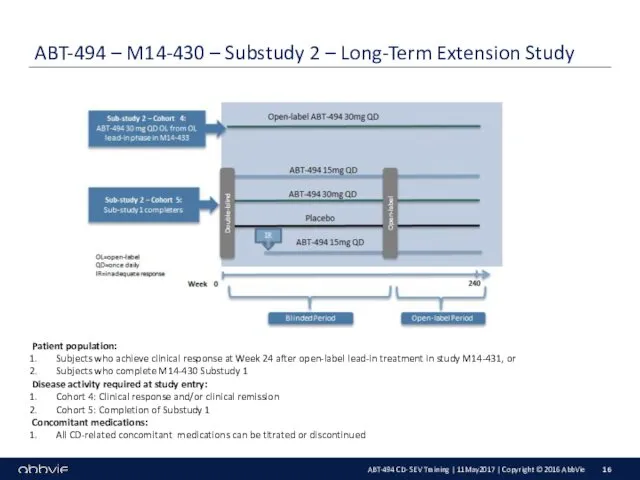
Key Inclusion Criteria:
Male or female aged 18 – 75 years old.
Confirmed
diagnosis of CD for at least 3 months prior to Baseline.
Simplified endoscopic score for CD (SES-CD) excluding the presence of narrowing component, ≥ 6 (or ≥ 4 for subjects with isolated ileal disease), confirmed by a central reader.
Average daily very soft/liquid stool frequency (SF) ≥ 4 and/or average daily abdominal pain (AP) score ≥ 2 at Baseline.
Demonstrated intolerance or inadequate response to one or more of the following categories of drugs :
M14-431: biologic therapy (adalimumab, certolizumab, infliximab, natalizumab, ustekinumab and/or vedolizumab)
M14-433: aminosalicylates, oral locally acting steroids, systemic steroids, immunomodulators (prior biologic use without failure or intolerance is allowed in up to 30% of subjects)
ABT-494 CD- SEV Training | 11May2017 | Copyright © 2016 AbbVie










 Экспериментальная психология
Экспериментальная психология Қорытынды нұсқаулықты құрастыру
Қорытынды нұсқаулықты құрастыру Дамымаған түсік
Дамымаған түсік Асептика и антисептика. Стерилизация перевязочного материала и хирургического белья автоклавированием. Подготовка рук
Асептика и антисептика. Стерилизация перевязочного материала и хирургического белья автоклавированием. Подготовка рук Онкогенные вирусы. Особенности противоопухолевого иммунитета
Онкогенные вирусы. Особенности противоопухолевого иммунитета Діагностика протозоонозів
Діагностика протозоонозів Структура личности по Платонову
Структура личности по Платонову LADA-диабет
LADA-диабет Інфекція та інфекційний процес. Основи епідеміології
Інфекція та інфекційний процес. Основи епідеміології Медицинская стерилизация
Медицинская стерилизация Последовательность действий спасателя
Последовательность действий спасателя Изменения в организме женщины при беременности
Изменения в организме женщины при беременности Реабилитация пациентов с заболеваниями внутренних органов: дыхания и пищеварения
Реабилитация пациентов с заболеваниями внутренних органов: дыхания и пищеварения Клиническая фармакология ЛП, применяемых при АГ
Клиническая фармакология ЛП, применяемых при АГ Лекарственные препараты, используемые в анестезиологии и интенсивной терапии
Лекарственные препараты, используемые в анестезиологии и интенсивной терапии Психологические центры РФ
Психологические центры РФ Чума. Эпидемия
Чума. Эпидемия Повреждение живота
Повреждение живота Первая помощь при отравлении
Первая помощь при отравлении Психика и теория отражения. Формы отражения
Психика и теория отражения. Формы отражения Новая коронавирусная инфекция
Новая коронавирусная инфекция Цинк. Роль цинка в организме человека
Цинк. Роль цинка в организме человека Острый панкреатит
Острый панкреатит Анатомические и физиологические основы дуоденального зондирования
Анатомические и физиологические основы дуоденального зондирования Черепно-мозговая травма у детей
Черепно-мозговая травма у детей Введение в инфекции. Бронхиты. Пневмонии. Рак легкого. Грипп
Введение в инфекции. Бронхиты. Пневмонии. Рак легкого. Грипп Пренатальная диагностика врождённых пороков развития и наследственных заболеваний
Пренатальная диагностика врождённых пороков развития и наследственных заболеваний Роль медицинской сестры в организации ЦСО
Роль медицинской сестры в организации ЦСО