Содержание
- 2. EDUCATIONAL GOALS 1) Compare and contrast: mixtures and pure substances. solutions, suspensions, and colloids. 2) Understand,
- 3. Disperse called the mixture in which one substance in the form of very small particles (in
- 4. Classification of Disperse systems and Solution Disperse system Solution Suspensions Colloidal system Emulsions Suspensions Aerosols Gel
- 5. Classification of Disperse Systems by Physical State
- 6. TYNDALL EFFECT IS OPTICAL PROPERTY OF SOLUTION This phenomenon was studied for the first time by
- 7. TYPES OF DISPERES SYSTEMS BY PARTICLE SIZE TRUE SOLUTION D COLLOIDAL SYSTEM D = 10-7 –
- 9. QUIZ ME NEXT 1 What is it a real solution? a pure substances in water compound
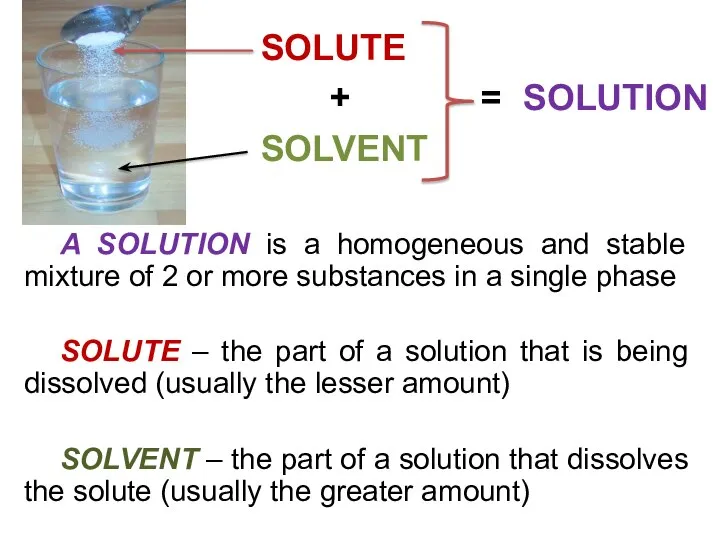
- 10. A SOLUTION is a homogeneous and stable mixture of 2 or more substances in a single
- 11. QUIZ ME NEXT 2 A solution consists of two parts. One part is the substance that
- 12. TRUE SOLUTION CLASSIFICATION OF SOLUTION BY NATURE OF SOLUTE Molecular solution: non –electrolytes, e.g. organic substances
- 13. Solvation (dissolution) – the process of surrounding solute particles with solvent particles to form a solution
- 15. CLASSIFICATION OF SOLUTION BY NATURE OF SOLVENT
- 16. SOLUBILITY refers to the maximum amount of solute, expressed in grams, that can be dissolved in
- 17. The Diluted is a solution in which small amount of solute dispersed in the solvent The
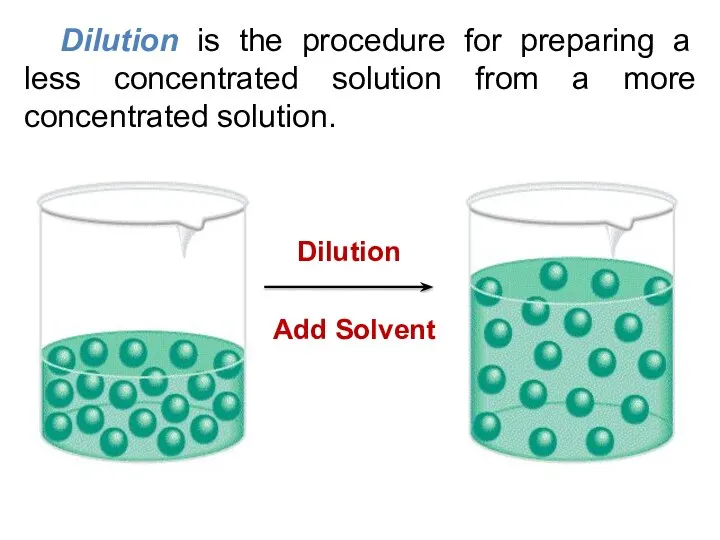
- 18. Dilution is the procedure for preparing a less concentrated solution from a more concentrated solution.
- 19. Miscible liquids dissolve in all proportions, e.g. ethanol and water (both H-bonded polar liquids). Immiscible liquids
- 20. Factors affecting solubility The nature of the solute and solvent: Polar substances tend to dissolve in
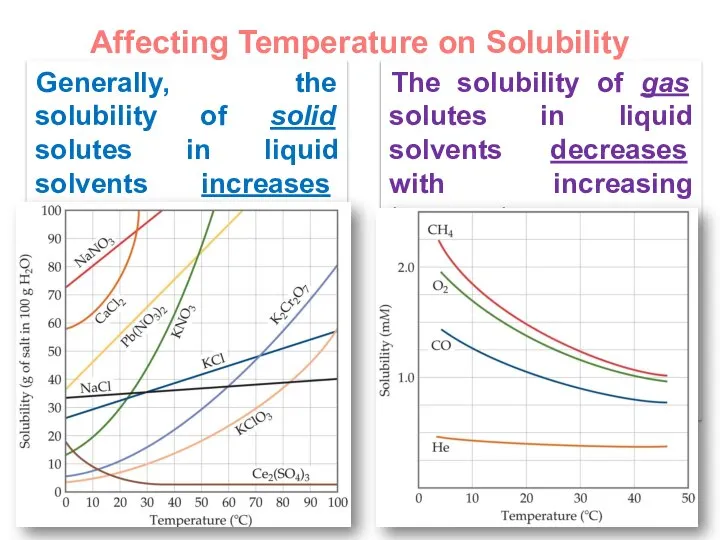
- 21. Affecting Temperature on Solubility Generally, the solubility of solid solutes in liquid solvents increases with increasing
- 22. Gases in Solution Increasing pressure above solution forces more gas to dissolve. The solubility of liquids
- 23. สารละลาย ผู้สอน: อ.ศราวุทธ แสงอุไร Pressure and Solubility of Gases The solubility of a gas in a
- 24. The solubility product constant, Ksp, is the equilibrium constant for a solid substance dissolving in an
- 25. QUIZ ME NEXT 3 The amount of a solute dissolved in a given amount of solvent
- 26. The concentration of a solution is the amount of solute present in a given quantity of
- 27. 1) Percent composition by mass is the mass of the solute divided by the mass of
- 28. 3) Normality is equal to the gram equivalent weight of a solute per 1 liter of
- 29. 5) Titer is equal to the gram of a solute per 1 milliliter of solution (g/ml):
- 31. Скачать презентацию






















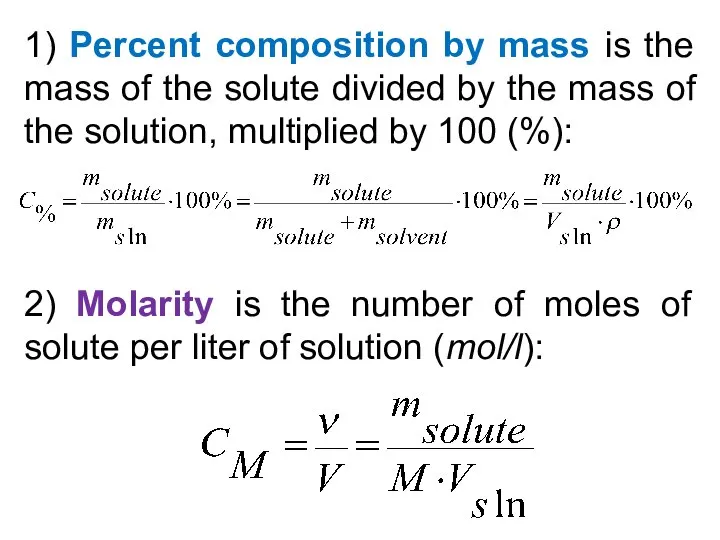


 ТОМИЛОВА НАТАЛЬЯ ВЛАДИМИРОВНА УЧИТЕЛЬ ХИМИИ И ЕСТЕСТВОЗНАНИЯ МОУ СОШ № 47
ТОМИЛОВА НАТАЛЬЯ ВЛАДИМИРОВНА УЧИТЕЛЬ ХИМИИ И ЕСТЕСТВОЗНАНИЯ МОУ СОШ № 47 Периодический закон Менделеева и периодическая система химических элементов
Периодический закон Менделеева и периодическая система химических элементов  Презентация по Химии "Нафта і нафтопродукти" - скачать смотреть бесплатно
Презентация по Химии "Нафта і нафтопродукти" - скачать смотреть бесплатно Горение водорода
Горение водорода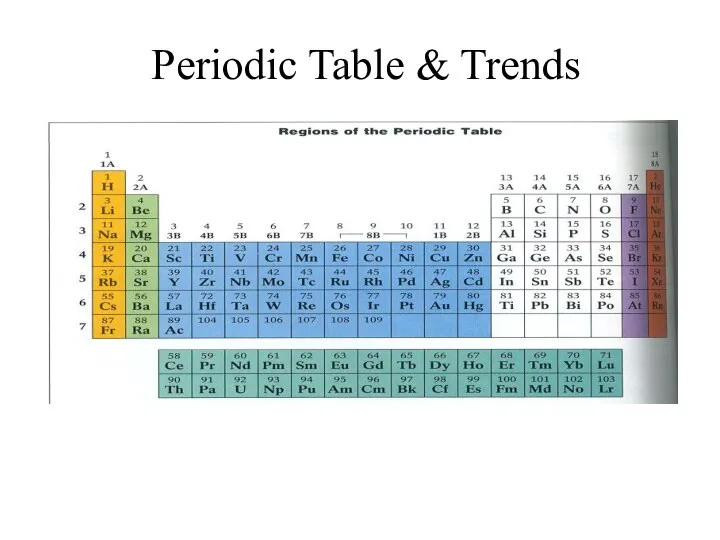 Periodic Table and Trends
Periodic Table and Trends Презентация по Химии "Галогены и их соединения" - скачать смотреть
Презентация по Химии "Галогены и их соединения" - скачать смотреть  Нуклеиновые кислоты Выполнил : Росовский Алексей
Нуклеиновые кислоты Выполнил : Росовский Алексей Получение этилена и опыты с ним. (Практическая работа 2)
Получение этилена и опыты с ним. (Практическая работа 2) Чистые вещества и смеси. 5 класс
Чистые вещества и смеси. 5 класс Презентация Драгоценные камни
Презентация Драгоценные камни  Жирорастворимые витамины. (Лекция 12)
Жирорастворимые витамины. (Лекция 12) Тема: Витамин Е Выполнила: студентка Шагиева А.А. ГОУ СПО ТОМУ № 1 г. Узловая 2012
Тема: Витамин Е Выполнила: студентка Шагиева А.А. ГОУ СПО ТОМУ № 1 г. Узловая 2012 Basic concepts and laws of chemistry
Basic concepts and laws of chemistry Природні гази
Природні гази Василий Баженов
Василий Баженов  Промежуточные фазы
Промежуточные фазы Окислительно-восстановительные реакции (ОВР). ОВР с участием органических веществ. Окисление углеводородов
Окислительно-восстановительные реакции (ОВР). ОВР с участием органических веществ. Окисление углеводородов Периодический закон Д.И. Менделеева
Периодический закон Д.И. Менделеева Сера. Важнейшие соединения серы
Сера. Важнейшие соединения серы Керамика на основе Al2O3
Керамика на основе Al2O3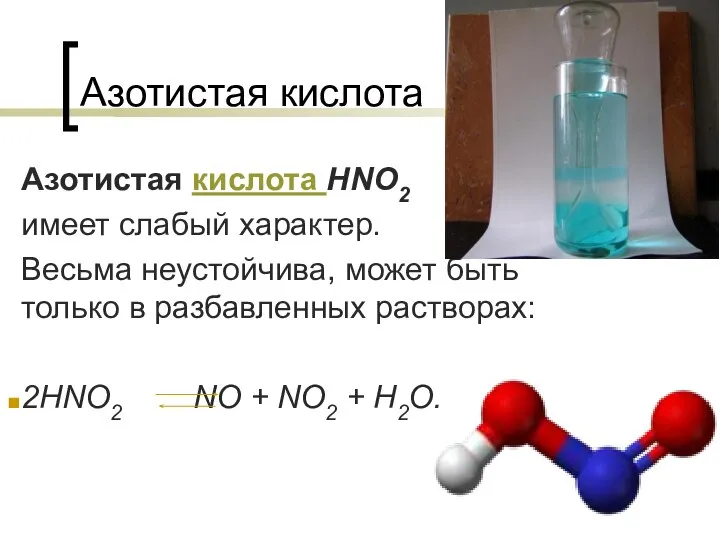 Азотистая кислота
Азотистая кислота Строение газообразных, жидких и твердых тел
Строение газообразных, жидких и твердых тел Каучук
Каучук Реакции иммунного лизиса. Реакция связывания комплемента (РСК)
Реакции иммунного лизиса. Реакция связывания комплемента (РСК) Золото Aurum
Золото Aurum Элементы VIIА-группы (галогены). Группа самых электроотрицательных элементов
Элементы VIIА-группы (галогены). Группа самых электроотрицательных элементов Крахмал
Крахмал Геология полезных ископаемых
Геология полезных ископаемых